Q: Calculate the solubility at 25 °C of CuBr in pure water and in 0.33 M NaI. You'll probably find some…
A: Solution -
Q: Calculate the solubility at 25 °C of PbCo, in pure water and in a 0.0050 M Pb(NO,) solution. You'l…
A:
Q: Calculate the solubility at 25 °C of Co (OH), in pure water and in a 0.0010M COC1, solution. You'll…
A:
Q: Caluulate me pH of a solution containing 0.784m NHz and 1.332m NH4N02- kp for Nits is 1.76X105
A: Given that: [NH3] = 0.784M [NH4NO3] = 1.332M Kb = 1.76×10-5 pKb = -logKb = -log(1.76×10-5) = 4.75
Q: A…
A:
Q: The molar solubility of Zn:(PO4)2 is 5.6 × 10-5 M at a certain temperature. Determine the value of…
A:
Q: Calculate the solubility at 25 °C of Ni(OH), in pure water and in a 0.0110 MNAOH solution. You'll…
A: Solution
Q: Calculate the solubility at 25 "C of PhCO, in pure water and in a 0.0020 M Pb(NO,), solution. You'l…
A:
Q: One litre of a saturated aqueous solution of Ag2SO4 (MW = 311.79 g mol- 1) at 25 °C is evaporated to…
A:
Q: Calculate the solubility of Ba(IO3)2 in pure water, Ksp=1.5x10^-9. Then calculate the solubility of…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Determine the molar solubility for Agl (Ksp = 8.3 x 10"). < PREV 1 2 3 Based on your ICE table and…
A:
Q: 1. The Ksp of Ca3(PO4)2 is 1.3 × 10−26. Estimate the solubility of this salt in units of g. L−1
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: What is the molar solubility (in mols/L)of silver chloride in 2.00 M NH3(aq) given that Ksp of AgCI…
A:
Q: Calculate the solubility at 25 °C of BaCro, in pure water and in a 0.0030 MBaCl, solution. You'll…
A: The solubility product Ksp of BaCrO4 is 2.4 X 10-10 And the solubility reaction is BaCrO4 (s)…
Q: Calculate the Solebury at 25 Celsius of PbCrO4. In pure water and in a .0200 M Na2CrO4 solution. Ksp…
A: The solubility of PbCrO4 in pure water can be calculated as follows:
Q: Calculate the solubility at 25 °C of Zn(OH), in pure water and in a 0.0180M ZnSO, solution. You'll…
A: Solubility of a substance is the amount of that substance dissolved in a given amount of solvent.…
Q: The solubility of PbSO, in water at 25 °C is 7.9 × 10-4 M. What is Ksp for PBSO,?
A:
Q: Consider the following solubility data for various chromates at 25°C. Ksp Ag2 CrO4 9.0 x 10-12…
A:
Q: Suppose an unknown sample of calcium is to be analysed by gravimetry technique. Which of the…
A: To get pure crystalline precipitate we have to follow crystalline process.
Q: calculate the ksp for PbBr2 if it's molar solubility is 2.14x10^-2 M at 25 degrees celsius. show…
A:
Q: Determine the molar solubility for Ag.CrO. (Ksp = 1.2 x 10 12). PREV 2 3 sed on your ICE table and…
A:
Q: calculate the sulablity Iat 25 Celsius of Ni(OH)2 pure water and then .01 80 M NaOH solution. Ksp…
A: Given:Concentration of NaOH = 0.0180M.Ksp of Ni(OH)2 =5.48×10−16
Q: The Ksp of AgBr (188g/mol) is 5.4 x 10-13 mol2/L2 at 25C if the solubility of it is----- g/L *
A: We know that Ksp is given by the product of individual solubility terms
Q: 4 5, Calculate the solubility of PbCO, in water at 25 °C. You'll find K, data in the ALEKS Data tab.…
A:
Q: The Ksp of CuCO3ls) (copper carbonate, a salt) is reported as 2.21x10-11. Determine the solubility…
A:
Q: Does the NaOH raise or lower the solution pH? What is the amount of NaOH added in lbs/ton? What is…
A: Volume of solution = 200 gallons 1 gallon = 0.0315 ton So, Volume = 6.3 ton We have added NaOH to…
Q: (a) Derive an expression for the ratio of solubilities of AgCI at two different temperatures; assume…
A: Let the value of the solubility product at temperature T1 be Ksp1 and the solubility product at a…
Q: Calculate the solubility at 25 °C of PbCrO4 in pure water and in a 0.0150M Na₂CrO4 solution. You'll…
A: Given: Concentration of Na2CrO4 solution = 0.0150 M Ksp of PbCrO4 = 2.8×10-13 Molar mass of PbCrO4 =…
Q: -7 -3 , of CuCl in pure water at this Given the Ksp temperature. of CuCl = 1.70 x 10 at 25 C,…
A: Here multiple questions are given.
Q: Determine the equilibrium constant (solubility constant) when a total of 0.025 m of M2SO4 wa…
A: Given reaction: M2SO4(s)↔2M++SO42- (Note that the reaction given in the question…
Q: Calculate the solubility at 25 °C of BaCrO, in pure water and in a 0.0140M BaCl, solution. You'll…
A:
Q: Calculate the solubility at 25 °C of P6CO3 in pure water and in a 0.0120M Pb(NO3), solution. You'll…
A: For PbCO3 , Ksp = 1.5 x 10-13 Solubility in pure water: PbCO3…
Q: Ametal sak vih he femal MCI, aystalies fom water to fom a soid with fhe compotion MCl, - GiH,0 The…
A: The reaction is – MCl2.6H2O s ⇔MCl2s +6H2Og
Q: I would like to calculate the molar solubility and Ksp from the following data: I have calculated…
A:
Q: Which of the following is NOT a characteristic of an IDEAL PRECIPITATE of a precipitation…
A: The characteristic of an IDEAL PRECIPITATE of precipitation gravimetry has to be given:
Q: Thallium(I) iodate (TlIO3) is only slightly soluble in water. Its Ksp at 25°C is 3.07 × 10-6 .…
A: Solubility is defined as the ability of a solute to dissolve in the solvent. Different substances…
Q: P Chemical eg X P Chemical eq X P Chemical ec X P Chemical ec X P Chemical ec x P Chemi…
A: Given: The molar solubility of BaCrO4 is 1.08*10-5 M.
Q: One mixes aqueous NaCl with aqueous AgNO3. NaCl. AgNO3and NaNO3all have appreciable solubility in…
A: First we are write reaction and then use Ksp of AgCl to determine which Compound would precipitate.
Q: It is found that up to 0.0110 g of SrF2 dissolves in 100 mL of aqueous solution at a certain…
A: Mass of SrF2 = 0.011 gMolar mass of SrF2 = 125.62 g/moleNo of moles of SrF2 = Mass of SrF2Molar mass…
Q: Calculate the solublity at 25 °C of Ni(OH), In pure water and In a 0.0040M NaOH solution. You'll…
A:
Q: Using an experimentally determined value (2.5×10−10) of Ksp, determine the value for the reaction…
A: The solubility product (Ksp) belongs to a sparingly soluble salt in an aqueous medium is furnished…
Q: Calculate the solublity at 25 °C of BaCrO, In pure water and in a 0.0110M BaCl, solution. You'll…
A:
Q: Question attached
A: Gravimetric analysis: This kind of analysis in chemical science has the application in the…
Q: An attempt was made to dissolve 5.893 grams of PbCl2 in 1.000 liters of water. The Ksp of PbCl2 is…
A:
Q: Calculate the values for log Kow and the saturated aqueous solubility (Cwsat) for bisphenol A.
A:
Q: Calculate the solubility at 25 °C of BaCrO4 in pure water and in a 0.0130M BaCl₂ solution. You'll…
A: Given, Concentration of BaCl2 = 0.0130 M (a). Calculate the solubility of BaCrO4 in pure water in…
Q: Estimate the concentration [in mol/L] of KNO3 that results in an increased solubility of 2.93*10-5…
A: Given: AgCl solubility increased from 1.3 × 10-5 M to 2.93 × 10-5 M by adding KNO3.
Q: Calculate the solubility at 25 °C of PbCro, in pure water and in a 0.0190M Na,CrO4 solution. You'll…
A:
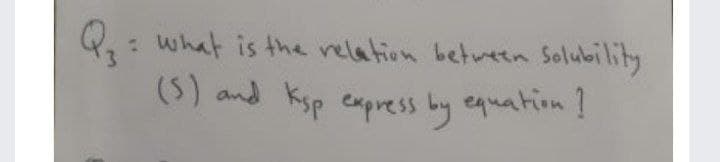
Step by step
Solved in 2 steps

- I would like to calculate the molar solubility and Ksp from the following data: I have calculated them (see the solutions in the table) but, I am concerned that I have done the calculation incorrectly. See images for my calculations. These calculations are for Sample 1. Thank you! Sample 1 Sample 2 Sample 3 Mass of Erlenmeyer Flask 23.59g 23.59g 23.60g Mass of Erlenmeyer Flask + Calcium Hydroxide Solution (lime water) 26.62g 26.65g 26.69g Mass of Calcium Hydroxide Solution 3.03g 3.06g 3.09g Volume of Ca(OH)2 Density = 1.000 g/mL 3.03mL 3.06mL 3.09mL Concentration of HCl (M) 0.1M 0.1M 0.1M Initial HCl Volume in Syringe 1.00mL(1) 1.00mL(2) 1.00mL(3) 1.00mL(1) 1.00mL(2) 1.00mL(3) 1.00mL(1) 1.00mL(2) 1.00mL(3) Final HCl Volume in Syringe 0ml (1) 0ml (2) 0.65mL (3) 0ml (1) 0ml (2) 0.63mL (3) 0ml (1) 0ml (2) 0.63mL (3) Volume of HCl Delivered 2.35mL 2.37mL 2.37mL Moles of HCl Delivered…Calculate the solubility at 25°C of CuBr in pure water and in a 0.0100M CoBr2 solution. You'll find Ksp data in the ALEKS Data tab. Round both of your answers to 2 significant digits. solubility in pure water: gL solubility in 0.0100 M CoBr2solution: gLThe following evidence was obtained from an experiment to determine the solubility of calcium chloride at room temperature. A sample of saturated calcium chloride solution was evaporated to dryness, and the mass of solid residue was measured.EvidenceVolume of solution (mL) = 15.0Mass of empty beaker (g) = 90.54Mass of beaker and residue (g) = 101.36The solubility of calcium chloride is g/100 mL
- Calculate the solubility at 25°C of CoOH2 in pure water and in a 0.0100M CoCl2 solution. You'll find Ksp data in the ALEKS Data tab. Round both of your answers to 2 significant digits. solubility in pure water: gL solubility in 0.0100 M CoCl2 solution: gLDetermine the molar solubility (moles/L) of Ni(OH)2. Ksp = 2.0 x 10-15 Express the answer to 3 sig. figs.1. The Ksp of Ca3(PO4)2 is 1.3 × 10−26. Estimate the solubility of this salt in units of g. L−12. If a sample of solid Ca3(PO4)2 is stirred into exactly one litre of a 0.550M solution of Na3PO4, how will the solubility of the salt compare with the answer that you have obtained in question 2.1? Explain you answer in a short sentence.
- 1.1The Ksp of Ca3 (PO4 ) 2 is 1.3 × 10−26 . Estimate the solubility of this salt in units of g. L −1 . You must show any reaction equation(s) that you may think are necessary. 1.2 If a sample of solid Ca3(PO4)2 is stirred into exactly one litre of a 0.550M solution of Na3PO4, how will the solubility of the salt compare with the answer that you have obtained in question 1.1? Explain you answer in a short sentence.Develop the solubility diagrams for CuCO3 in a closed system and an open system, respectively. Please provide rationale or workings for your answer.A solution is prepared by dissolving 40.00 g of NaCl (f.w. = 58.44 g mol–1), a non-volatile solute, in enough water (m.w. = 18.02 g mol–1) to result in exactly 1 L of solution at 25 °C. Assume the density of the solution is that of pure water (dsolution = 1.000 g mL–1). The ebullioscopic constant (Kb) for water is 0.513 °C m–1. The cryoscopic constant (Kf) for water is 1.86 °C m–1. The vapor pressure of pure water is 0.0313 atm. Find the freezing point of the solution(in C to 2 decimal places)
- A solution is prepared by dissolving 40.00 g of NaCl (f.w. = 58.44 g mol–1), a non-volatile solute, in enough water (m.w. = 18.02 g mol–1) to result in exactly 1 L of solution at 25 °C. Assume the density of the solution is that of pure water (dsolution = 1.000 g mL–1). The ebullioscopic constant (Kb) for water is 0.513 °C m–1. The cryoscopic constant (Kf) for water is 1.86 °C m–1. The vapor pressure of pure water is 0.0313 atm. Find the osmotic pressure in atm to three decimal placesA solution is prepared by dissolving 40.00 g of NaCl (f.w. = 58.44 g mol–1), a non-volatile solute, in enough water (m.w. = 18.02 g mol–1) to result in exactly 1 L of solution at 25 °C. Assume the density of the solution is that of pure water (dsolution = 1.000 g mL–1). The ebullioscopic constant (Kb) for water is 0.513 °C m–1. The cryoscopic constant (Kf) for water is 1.86 °C m–1. The vapor pressure of pure water is 0.0313 atm. Determine the boiling point of the solution(in C to 2 decimal places)A solution is prepared by dissolving 40.00 g of NaCl (f.w. = 58.44 g mol–1), a non-volatile solute, in enough water (m.w. = 18.02 g mol–1) to result in exactly 1 L of solution at 25 °C. Assume the density of the solution is that of pure water (dsolution = 1.000 g mL–1). The ebullioscopic constant (Kb) for water is 0.513 °C m–1. The cryoscopic constant (Kf) for water is 1.86 °C m–1. The vapor pressure of pure water is 0.0313 atm. Determine the following: Boiling point of solution (in °C to two decimal places) Freezing point of solution (in °C to two decimal places) Vapor pressure of the solution (in atm to three decimal places) Osmotic pressure (in atm to three decimal places)



