5 B-Glucose can be converted to D-ribose through the pentose phosphate pathway, shown below. 2- O,PO pentose phosphate pathway 2- O,PO HO OH но он (a) What byproduct (not shown) is also formed in this reaction? (b) The key step in this pathway is where the six carbon chain is shortened to five carbons. 2- но 2- O,PO- NADP O,PO- HOR HO- но он Which structure below is an intermediate in this reaction? 容 но HO O,PO 2- O,PO- OH O,PO- O,PO- HOT FOPO, но но B D
5 B-Glucose can be converted to D-ribose through the pentose phosphate pathway, shown below. 2- O,PO pentose phosphate pathway 2- O,PO HO OH но он (a) What byproduct (not shown) is also formed in this reaction? (b) The key step in this pathway is where the six carbon chain is shortened to five carbons. 2- но 2- O,PO- NADP O,PO- HOR HO- но он Which structure below is an intermediate in this reaction? 容 но HO O,PO 2- O,PO- OH O,PO- O,PO- HOT FOPO, но но B D
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter22: Gluconeogenesis, Glycogen Metabolism, And The Pentose Phosphate Pathway
Section: Chapter Questions
Problem 14P
Related questions
Question
Please answer it soon
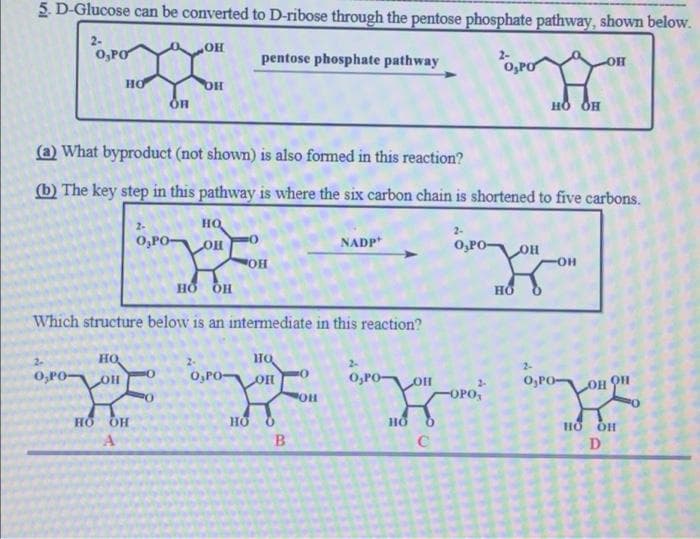
Transcribed Image Text:5 D-Glucose can be converted to D-ribose through the pentose phosphate pathway, shown below.
2-
0,PO
pentose phosphate pathway
O,PO
HO
HO QH
(a) What byproduct (not shown) is also formed in this reaction?
(b) The key step in this pathway is where the six carbon chain is shortened to five carbons.
2-
HO
2-
O,PO-
NADP
O,PO-
OH
Which structure below is an intermediate in this reaction?
но
HO
2-
2-
O,PO-
O,PO-
O,PO-
OH
O,PO-
OPO
но
HO
D
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning