
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
thumb_up100%
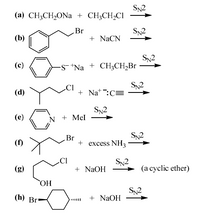
Transcribed Image Text:Sn2
(а) СН,СH-ONa + CH;CH-CI
Br
+ NaCN
SN2
(b)
SN2
-S- *Na + CH3CH,Br
(c)
SN2
+ Na*:C=
(d)
SN2
(e)
N + Mel
S2
(f)
Br
+ excess NH3
CI
SN2
+ NaOH
(g)
(a cyclic ether)
ОН
SN2
(h) Br
+ NaOH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- (d) the ion with 74 electrons, 116 neutrons, and a +3 chargearrow_forwardRank these confirmations from least to most stable (le H. エ H,C. エ H. Br Br. CH, H,C CH3 HH,C H,C H. H. %3D H. Br |< ||< |I| III< ||arrow_forward5) ✓ PbS04 → PbSO3 + _ 02 6) _ Al + Fe2O3 → Fe + . Al203 7) SF6 → S + F2 8) _ C2H4 + 02 → CO2 + _ 9) _ KCIO3 → KCl + 02 H₂O 10) Pb(NO3)2 + _ NaCl → _ NaNO3 + _ PbCl2arrow_forwardYou have 1 million atoms each of MN As S Cd and W. Which million of attoms is the heaviest? lightest?arrow_forwardn-BUCOH(Me)2 A 1) 2) n-BUCHO n-BUCOCI H2NOH/H * (Et), CuLi ******* ******** ***** A E Dry HCI HO(CH2);CHO DHP HCrO4 7A 1) PhLi/Et,0 2) H* (aq) Garrow_forwardCH;COCI pyridine IZarrow_forward*(iii) The reaction of ethene with aqueous potassium manganate(VII), KMNO., produces ethane-1,2-diol, CH,OHCH,OH. Data: molar mass of ethane-1,2-diol = 62 g mol In an experiment, KMNO, containing only "O reacts with ethene. Suggest how the mass spectrum of ethane-1,2-diol data could be used to decide whether the oxygen atoms in ethane-1,2-diol came from the manganate(VII) ion, water, or a combination of the two. (2)arrow_forwardA dose of 5 milligrams of penicillin is administered to a patient. The absorption constant for penicillin is 11% per hour and its minimum effective concentration is 2 milligrams. Use the formula C= Coe^kt. a). What amount of penicillin is present in this patient’s body at 30 minutes? Round your answer to one decimal place. b).When should another dose of penicillin be administered to this patient in order to maintain the minimum effective concentration? Round your answer to one decimal place.arrow_forward(c) 3.23 М КЗАSO4 lons: 04- KAS+ O AsO4 AsO43 O K* K3+ masscation i massanion IIarrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY