A CHM 122 student makes an aqueous solution that contains 19.7 gof Cr3(PO4)2 and 138.7 g H,O. Determine the freezing point of the solution (in units of °C). freezing point depression constant for water = 1.858 °C/m molar mass Cr3(PO4)2 = 345.9326 g/mol molar mass H2O= 18.01528 g/mol Recall:
A CHM 122 student makes an aqueous solution that contains 19.7 gof Cr3(PO4)2 and 138.7 g H,O. Determine the freezing point of the solution (in units of °C). freezing point depression constant for water = 1.858 °C/m molar mass Cr3(PO4)2 = 345.9326 g/mol molar mass H2O= 18.01528 g/mol Recall:
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter11: Properties Of Solutions
Section: Chapter Questions
Problem 141IP: An aqueous solution containing 0.250 mole of Q, a strong electrolyte, in 5.00 102 g water freezes...
Related questions
Question
Question is attached
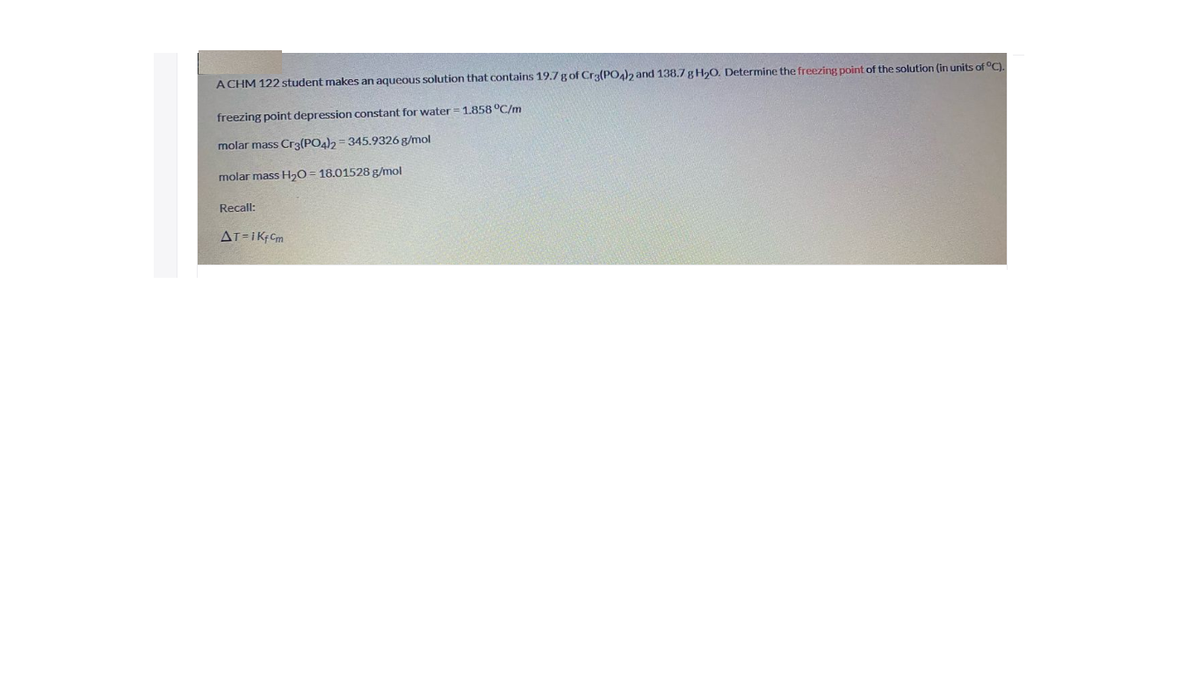
Transcribed Image Text:A CHM 122 student makes an aqueous solution that contains 19.7 gof Cr3(PO4)2 and 138.7 g H,O. Determine the freezing point of the solution (in units of °C).
freezing point depression constant for water = 1.858 °C/m
molar mass Cr3(PO4)2 = 345.9326 g/mol
molar mass H2O= 18.01528 g/mol
Recall:
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning