A researcher prepared mixed solutions at different concentrations and measured the absorbances with the help of a spectrophotometer. In the spectrophotometer device, the distance between the cuvette and the light diffuser is 3 cm. The absorbance values of the mixture given in the table below. Mixture V (ml) %T Absorbans Concentrations (g/I) 5 37 ,55 0,444 0,21M 4 40,89 0,372 0,26M 3 44,97 0,311 0,35M 2 67,50 0,172 0,71M 1 82,65 0,084 0,97M a) Explain how to prepare 1N H2SO4 solution. b) Calculate the ɛ values and the total absorbance for each volume value opposite. c) Explain the relationship between permeability and absorbance.
A researcher prepared mixed solutions at different concentrations and measured the absorbances with the help of a spectrophotometer. In the spectrophotometer device, the distance between the cuvette and the light diffuser is 3 cm. The absorbance values of the mixture given in the table below. Mixture V (ml) %T Absorbans Concentrations (g/I) 5 37 ,55 0,444 0,21M 4 40,89 0,372 0,26M 3 44,97 0,311 0,35M 2 67,50 0,172 0,71M 1 82,65 0,084 0,97M a) Explain how to prepare 1N H2SO4 solution. b) Calculate the ɛ values and the total absorbance for each volume value opposite. c) Explain the relationship between permeability and absorbance.
Chapter25: Instruments For Optical Spectrometry
Section: Chapter Questions
Problem 25.6QAP
Related questions
Question
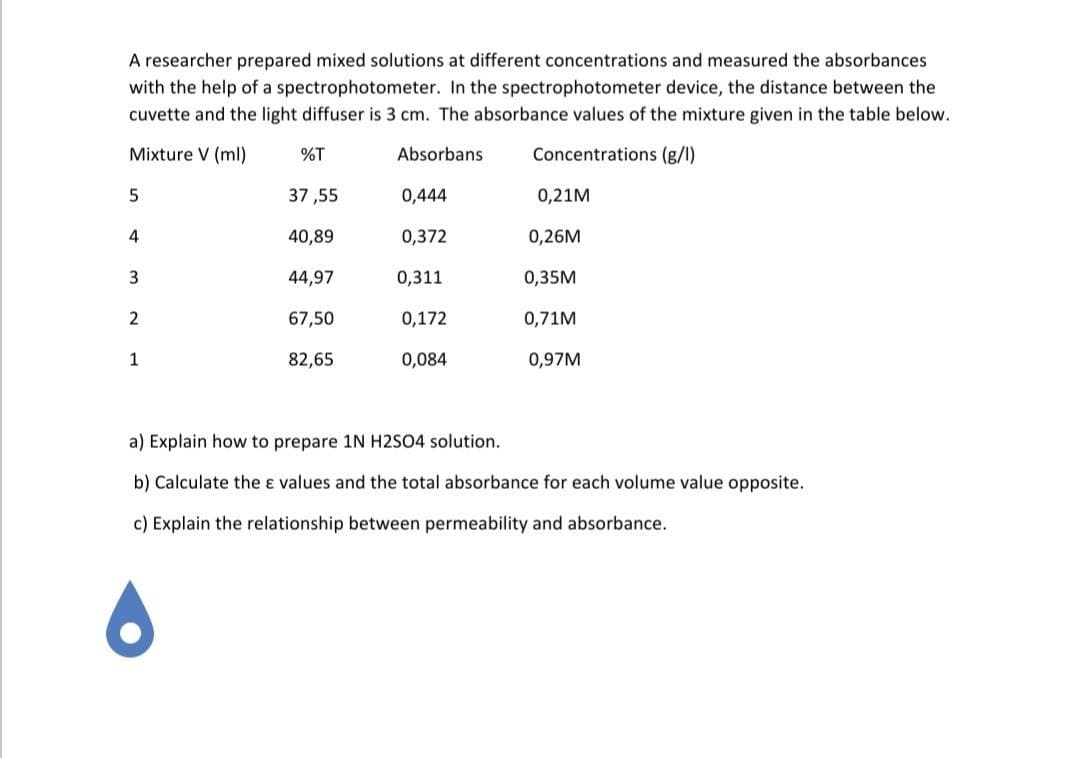
Transcribed Image Text:A researcher prepared mixed solutions at different concentrations and measured the absorbances
with the help of a spectrophotometer. In the spectrophotometer device, the distance between the
cuvette and the light diffuser is 3 cm. The absorbance values of the mixture given in the table below.
Mixture V (ml)
%T
Absorbans
Concentrations (g/l)
37 ,55
0,444
0,21M
4
40,89
0,372
0,26M
3
44,97
0,311
0,35M
2
67,50
0,172
0,71M
1
82,65
0,084
0,97M
a) Explain how to prepare 1N H2SO4 solution.
b) Calculate the ɛ values and the total absorbance for each volume value opposite.
c) Explain the relationship between permeability and absorbance.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 7 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning