A tank contains 200L of a dye solution with a concentration of 1gram per liter. To prepare for the next experiment, the tank is to be rinsed with fresh water flowing in a rate 2 liters per minute; the well stirred solution flowing out with the same rate. Find the time that will elapse before the concentration of dye in the tank reaches 1% of its original value.
Q: A 600-gal tank initially contains 100gal of brine containing 25 lb of salt. Brine containing 1 lb…
A:
Q: Put the steps in order to make 100 mL of 2.00×10-4M from 2.00×10-2 M solution of NaSCN. You have…
A: In order to prepare a 100 mL of 2.00×10-4 M, the given solution, 2.00 ×10-2 M is diluted. First the…
Q: The method of standard additions was used to determine nitrite in a soil sample. A 1.00-mL portions…
A: The linear relationship between the concentration of absorbing species and the absorbance is known…
Q: A variation of the indicator-dilution method (see preceding problem) is used to measure total blood…
A: The relationship which relates the calibration curve of absorbance (A) and tracer concentration (C)…
Q: Drug Stock solution concentration = 10 millimolars Desired drug solution concentration = 10…
A: Dilution is defined when more solvent is added in the solution due to which concentration of the…
Q: In a fractional distillation of a cyclohexane/toluene mixture the effectiveness of the distillation…
A: The solution is given below -
Q: A pipet is used to transfer 6.00 mLmL of a 3.75 MM stock solution in flask “S” to a 25.00-mL…
A: The number of moles of compound in 25.00 mL flask “A” is; 1000 mL=1 Ln=3.75 M×6.00 mL1000…
Q: To transfer a solution, the stirring rod touches the delivery point of the reagent vessel and the…
A: Since you have posted multiple questions as per guidelines we can answer one . If you want remaining…
Q: A 108 is performed on a culture of bacteria in order to perform viable plate counts. From the…
A: When culture medium is dispersed on the viable plate, a very thin layer is formed in which bacteria…
Q: Data Fill in the time and temperature for each solution: Solution Temperature ("C) Time (s) 17.9°C 1…
A:
Q: Why is it that the method used to attack a problem is an important as the answer to the problem…
A: A problem can be solved by the following steps- • Understand the requirement of the problem •…
Q: A student prepares a 1.2 mM aqueous solution of butanoic acid (C,H,CO,H). Calculate the fraction of…
A: Given Concentration of butanoic acid = 1.2 mM
Q: A formula calls for 0.5 milliliter of hydrochloric acid. Using a 10-milliliter graduate calibrated…
A: • Aliquot is a type of sub-sample that is taken or extracted from an original sample. • If we…
Q: A tank initially contains 200 liters of fresh water. Brine containing 2.5 N/liter of dissolved salt…
A: If "s" represents the quantity of salt in the tank, then it must satisfy the differential equation..…
Q: Before Investigating the scene the technician must dilute the luminol solution to a concentration a…
A: Molarity of the solution = 2×10-2M Volume = 2L Moles =?
Q: When mixing sodium chlorate solution (volume 1.0L, density 1.1614 g/ml, mass fraction 22%) with…
A: A balanced reaction is one that contains equal number of atoms of each element in reactants and…
Q: A student performs a crystallization on an impure sample of biphenyl. The sample weighs 0.5 g and…
A: The boiling point of benzene is 80°C and melting point of biphenyl is 69.2°C. The solubility…
Q: 50 mg of granular salt is briefly stirred into a glass container of 1/5 liter volume filled with…
A: To calculate dissolved phase concentration , we would first calculate mg of salt dissolved in water…
Q: You have finished two trials of the sample analysis for the experiment and you noticed that the…
A: There are some tips and techniques which should be taken in mind while performing a titration.…
Q: The data in the problem has error. Make it 0.99 M KMnO4. Show the complete solution
A:
Q: Identify the type of error (systematic or random) for each of the following situations below and…
A: Systematic error : it is repeatable error and consistent . it can be done by the analysts . it comes…
Q: When answering this problem, report the answer with the appropriate number of significant figures.…
A: Given : Calibration curve equation : y = 21655 x + 0.0318 where y = Absorbance x = molar…
Q: A 0.2422 g sample of a certain fruit was analyzed for the content of a certain pesticide. After a…
A: “Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: 1) Mass of aspirin used (g) 0.088g 2) Absorbance for final unknown solution (measured) 3) Molarity…
A: Aspirin is a drug used for controlling pain, reducing fever etc. Aspirin tablet is not made up of…
Q: Approximately 10 mL of 6 M hydrochloric acid were initially added to the euiometer. The eudiometer…
A: The mass of the 10mL hydrochloric acid with 6M molarity can be calculated as follows:
Q: Arrange the following steps in performing a general procedure. I. work on the data II. Carry out the…
A: we are required to outline the steps to perform a general procedure
Q: 50.00 cm3 of a 1.5784 mol.dm-3 solution of potassium hydroxide is transferred to an empty 700.00 cm3…
A: Given: The initial concentration, (C1)=1.5784 mol.dm-3 The initial volume, (V1)=50 cm3 The final…
Q: Data Sheet II. Delivering Solution from Your Buret 17.28 (1) final buret reading, mL (2) initial…
A:
Q: You are a student in Mrs. Bateast's Chemistry class and you were given the assignment to compare…
A: The solubility of a compound is increased by increasing temperature. It would continue up to the…
Q: 1. Standardization of Sodium Hydroxide Primary Standard used: Formula Mass of Primary Standard: %…
A: In the given question we have to do the standardization of NaOH and to calculate the required NaOH…
Q: Standardization of Sodium Thiosulfate Solution Primary Standard used: Potassium Dichromate…
A: The formula of potassium dichromate is K2Cr2O7
Q: During lab, a student used a Mohr pipet to add the following solutions into a 25 mL volumetric…
A: Solutions concentration get altered on dilution, but the solutes moles initially present remains…
Q: A three-component mixture containing compounds A, B, and C is separated by simple distillation. A…
A:
Q: Some crushed ice was placed in a 400 mL beaker. Some of the ice was allowed to melt before the…
A: Given data :-
Q: Trituration is a process of reducing particle size which involves the addition of an appropriate…
A: There are various method of reducing the particle size such as grinding, mechanical mixing etc.
Q: A river water sample (250 mL) is filtered and 0.032 g of solid is collected on the filter paper.…
A: The total solid in the river sample in ppm, is to be determined from the given data. Given:Volume of…
Q: Sugar is the solute and water is the solvent. The drink mix will be considered negligible and is…
A: Calculate the concentration of sugar in this solution using the volume percent equation.,
Q: After assembling a gravity filtration apparatus, begin the separation of a mixture by Choose... it…
A: • In this problem, we have to fill suitable words in tha blank sapce. • This is a process of an…
Q: 2 mL of thiamine, 0.2 mL of folic acid, and 10 mL of multivitamin infusion are mixed with 500 mL of…
A: Total volume of the solution after infusion = 500 mL + 10 mL + 2 mL + 0.2 mL = 512.2 mL Target time…
Q: 4. Each of the two tanks shown below contain 150 L of water. Initially, Tanks 1 and 2 have 15 g and…
A: In order to solve this question, we will be using a basic formula which is that the accumulation is…
Q: A) Three teaspoons of water (approx. 15 ml) were added to one tablet said to contain 300 mg of…
A: Aspirin is a chemical compound also known as Acetylsalicylic acid. It is obtained from salicylic…
Q: STANDARD SAMPLE PREPARATIONS FOR ABSORBANCE & CONCENTRATION DATA Concentration of stock nickel…
A: In order to solve this table and find molarities/concentrations, we will use the law of dilution…
Q: 100 ml g 0:4 Nop HSq mixed with 100m) oro.2 N N<OH, Ihen Final Normaliliy og Resxelt toletian is A)…
A:
Q: Calculate the Standardized Molarity of NaOH via dimensional analysis. Show your process. Use this…
A:
Q: Method of separation/chemical reaction (with brief process) of raw materials of the assign drug…
A: Given: mass of cream of tartar = 1.5 kg = 1500 g mass of sodium carbonate monohydrate = 2.5 kg =…
Q: Explain briefly the percent volume and percent retrieval in separation of mixtures.
A:
Q: Analytical Chemistry Laboratory for Biology Majors Data Sheet After the addition of increments of…
A: The standardization of KHP was done with NaOH in which the number of moles of NaOH was equal to that…
Q: Cat Noir was tasked to determine the concentration of a CuSO4 unknown solution. She prepared 5…
A: Lambert Beer's Law depicts that when a monochromatic light flows through an adsorbing medium, the…
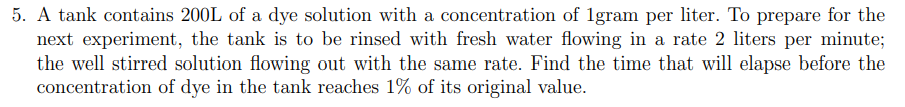
Step by step
Solved in 2 steps with 2 images

- Approximately 10 mL of 6 M hydrochloric acid were initially added to the euiometer. The eudiometer was then filled with deionized water and placed into a 400-mL beaker contaning ~300 mL of deionized water. Assuming the total volume of solution was 350 mL, and that the solution was uniformly mixed, what was the dilute concentration of the hydrochloric acid?A 1 mL sample of glycogen was calculated to contain 21 µmol (micromole) glucose. To 1 mL of this sample was added 2 mL of 2 M HCl. It was then hydrolysed by boiling the solution for 15 minutes. After boiling the hydrolysate was cooled and made up with H2O to a final volume of exactly 10 mL. The glucose was measured in this solution and found to have a concentration of 340 µg/mL (microgram/milliliter). i) Calculate the mass (mg) of glucose in the 10 mL of hydrolysate. As the 1 mL of glycogen sample was made up to a final volume of 10 mL, this mass of glucose was produced by the hydrolysis of the original 1 mL glycogen sample. ii) Calculate the amount (µmol) of glucose produced by the hydrolysis of the glycogen sample. iii) Calculate the purity of the glycogen used in the sample as % Purity = (moles of measured glucose/ moles of calculated glucose in glycogen) *100 iv) state your answer in a complete sentence. Show your working out such that the marker can easily understand it.…A 1 mL sample of glycogen was calculated to contain 35 µmol (micromole) glucose. To 1 mL of this sample was added 2 mL of 2 M HCL. It was then hydrolysed by boiling the solution for 15 minutes. After boiling the hydrolysate was cooled and made up with H2O to a final volume of exactly 10 mL. The glucose was measured in this solution and found to have a concentration of 570 µg/mL (microgram/milliliter). i) Calculate the mass (mg) of glucose in the 10 mL of hydrolysate. As the 1mL of glycogen sample was made up to a final volume of 10 mL, this mass of glucose was produced by the hydrolysis of the original 1 mL glycogen sample. ii) Calculate the amount (µmol) of glucose produced by the hydrolysis of the glycogen sample. iii) Calculate the purity of the glycogen used in the sample as % Purity = (moles of measured glucose/ moles of calculated glucose in glycogen) *100 iv) state your answer in a complete sentence.
- No plagiarism please! 11. Describe a method to separate salt from a homogeneous mixture of salt water so that it is in the form of a solid at the end of the separation process.1. Calculate the experimental density of a salt solution and the percent error (same as relative error percent) using some or all the data given below. solubility of NaCl salt in water: 0.357 g/mLmass of empty graduated cylinder: 25.19g mass of graduated cylinder + salt solution: 30.47g total volume of salt solution: 4.98 mLtrue density of salt solution: 1.07 g/mLA blood concentration of 0.065 M ethyl alchohol is sufficient to induce a coma. Blood alcohol content is a measure of alcohol in the blood as a percentage. It is calculated in grams per 100mL of blood, so a BAC os 0.08 means your blood is 0.08% alcohol by volume. a) What is the total mass of alcohol (in grams) that it represents for the same adult male whose total blood volume is 5.6L? b) What percentage does it represent from the coma induced level?
- Stock iron(II) solution (200Ug mL-1 Fe) ferrous ammonium sulfate hexahydrate mass= 0.1437g, transfer it to a 100 ml beaker. add 15 ml approx of water and 15m1 'approx of dilute sulphuric acid (2M H2SO.). then transfer FeII to 100 ml flask makeup to the mark with water. calculate the moles of ferrous ammonium sulfate hexahydrate solution in unit ug/mL.using exactly 5.00 mL of 0.0400 M stock CuSO4 solution. Add 100 mL of water. Data for Part IMass of empty dish: 32.470 g empty dishVolume of 0.0400 M solution: 5.00 mL CuSO4 solution.Mass of dish and 0.0400 M solution: 37.497 g dish and solutionMass of dish and CuSO4 solid: 32.503 g dish and CuSO4 solidCalculations for Part I1. Calculate the mass of solution? 5.027 g 2. Calculate the mass of solid CuSO4 dissolved in the solution? 0.033 g 3. Calculate the number of moles of solid CuSO4 dissolved in the solution? 2.07 x 10-4 mol4. Calculate the mass of water evaporated from the solution? 0.0414 M5. Calculate the density of solution, (g solution/mL solution)?6. Calculate the % by mass, CuSO4 in solution (100 x g CuSO4/g solution)?7. Calculate the molality of solution (moles CuSO4/kg solvent)?8. Calculate the molarity of solution (moles CuSO4/L solution)?9. Given that the true molarity is 0.0400 M, calculate the percent error…In this station, you will find a sample of table salt; the chemical formula of table salt is NaCl. Using the balance, weigh out 6.00 g of NaCl and dissolve in 50 mL of deionized (d.i.) water. Once com-pletely dissolved, add additional d.i. water to make the final volume of solution 100.0 mL. What is the mass percent NaCl in the solution? What is the molarity of the solution
- using exactly 5.00 mL of 0.0400 M stock CuSO4 solution. Add 100 mL of water. Data for Part IMass of empty dish: 32.470 g empty dishVolume of 0.0400 M solution: 5.00 mL CuSO4 solution.Mass of dish and 0.0400 M solution: 37.497 g dish and solutionMass of dish and CuSO4 solid: 32.503 g dish and CuSO4 solidCalculations for Part I1. Calculate the mass of solution2. Calculate the mass of solid CuSO4 dissolved in the solution.3. Calculate the number of moles of solid CuSO4 dissolved in the solution.4. Calculate the mass of water evaporated from the solution.5. Calculate the density of solution, (g solution/mL solution).6. Calculate the % by mass, CuSO4 in solution (100 x g CuSO4/g solution).7. Calculate the molality of solution (moles CuSO4/kg solvent).8. Calculate the molarity of solution (moles CuSO4/L solution).9. Given that the true molarity is 0.0400 M, calculate the percent error of your result.A) An aqueous solution of ethyl alcohol is made by transfering 4.72 mL of liquid ethyl alcohol to a 200 . mL volumetric flask, and then adding enough water to fill the flask to the mark. What is the volume/volume percentage of ethyl alcohol in the solution? Volume/volume percentage = ________% B) An aqueous solution of acetic acid is made by transfering 17.5 mL of liquid acetic acid to a 200 . mL volumetric flask, and then adding enough water to fill the flask to the mark. What is the volume/volume percentage of acetic acid in the solution? Volume/volume percentage = ________%A student dissolves a Jefferson nickel to make 100.00 mL of solution in a volumetric flask. The student takes a 5.00 mL5.00 mL aliquot of the first solution and dilutes it to make 100.00 mL100.00 mL of a second solution. The student places a sample of the second solution in a cuvette for analysis using spectrophotometry. The molarity of the copper solution in the cuvette was determined by spectrophotometric analysis to be 2.90×10−2 M Cu.2.90×10−2 M Cu. Calculate the mass of copper in the Jefferson nickel used to make the first solution.

