Active Raw Actual Yield Formulation Dosage Form Packaging Ingredient Materials Calcium 1.75kgs calcium 2 kgs calcium 16omg per Oral 100mL per carbonate oxide and 10mL Suspension bottle 1.75 kg H,O carbonate suspension Compute for the following using the Balanced Chemical Reaction: Cao + H,O → Ca(OH); Please provide a step-by-step solution for these. Thank you very much! % composition by mass of each compound Mass to Mass Stoichiometry Calculation Limiting Reagent
Active Raw Actual Yield Formulation Dosage Form Packaging Ingredient Materials Calcium 1.75kgs calcium 2 kgs calcium 16omg per Oral 100mL per carbonate oxide and 10mL Suspension bottle 1.75 kg H,O carbonate suspension Compute for the following using the Balanced Chemical Reaction: Cao + H,O → Ca(OH); Please provide a step-by-step solution for these. Thank you very much! % composition by mass of each compound Mass to Mass Stoichiometry Calculation Limiting Reagent
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter12: Chemical Equilibrium
Section: Chapter Questions
Problem 121QRT: When a mixture of hydrogen and bromine is maintained at normal atmospheric pressure and heated above...
Related questions
Question
Can someone provide a step-by-step answer for the following blanks in the second table? Thank you in advance! :)
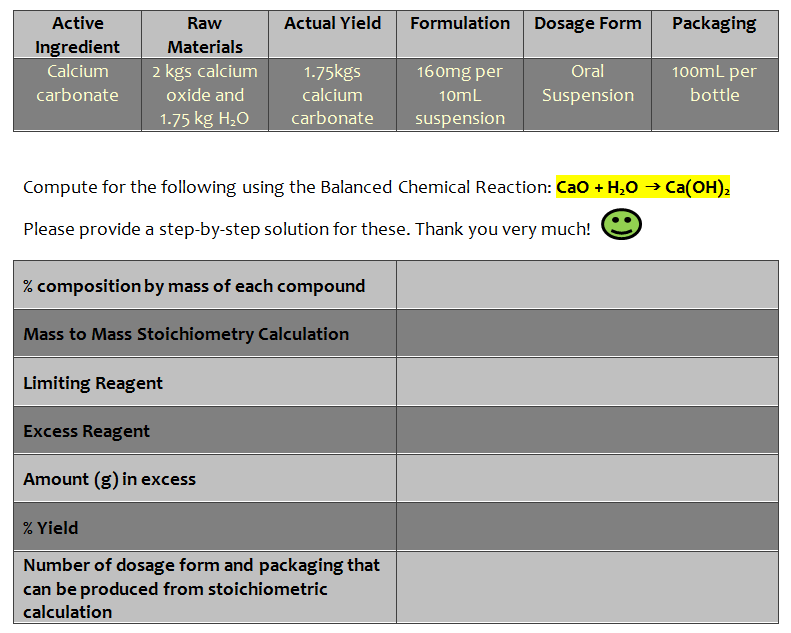
Transcribed Image Text:Active
Raw
Actual Yield
Formulation Dosage Form
Packaging
Ingredient
Materials
Calcium
2 kgs calcium
1.75kgs
160mg per
Oral
100mL per
carbonate
oxide and
calcium
10ml
Suspension
bottle
1.75 kg H,0
carbonate
suspension
Compute for the following using the Balanced Chemical Reaction: Cao + H,O → Ca(OH),
Please provide a step-by-step solution for these. Thank you very much!
% composition by mass of each compound
Mass to Mass Stoichiometry Calculation
Limiting Reagent
Excess Reagent
Amount (g) in excess
% Yield
Number of dosage form and packaging that
can be produced from stoichiometric
calculation
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 8 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning