At roughly what pressure, P, and temperature, T, will diamond, graphite, and liquid carbon all exist in equilibrium? Carbon phase diagram 10- Solid I (diamond) 10- Liquid P = 10- atm 10- Solid II (graphite) 10- Gas T = K 10- 1- 0. 1000 2000 3000 4000 5000, 6000 Which solid form of carbon is more dense? T(K) diamond O graphite P (bar)
At roughly what pressure, P, and temperature, T, will diamond, graphite, and liquid carbon all exist in equilibrium? Carbon phase diagram 10- Solid I (diamond) 10- Liquid P = 10- atm 10- Solid II (graphite) 10- Gas T = K 10- 1- 0. 1000 2000 3000 4000 5000, 6000 Which solid form of carbon is more dense? T(K) diamond O graphite P (bar)
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter6: Equilibria In Single-component Systems
Section: Chapter Questions
Problem 6.50E
Related questions
Question
I need accurate and full details answers.
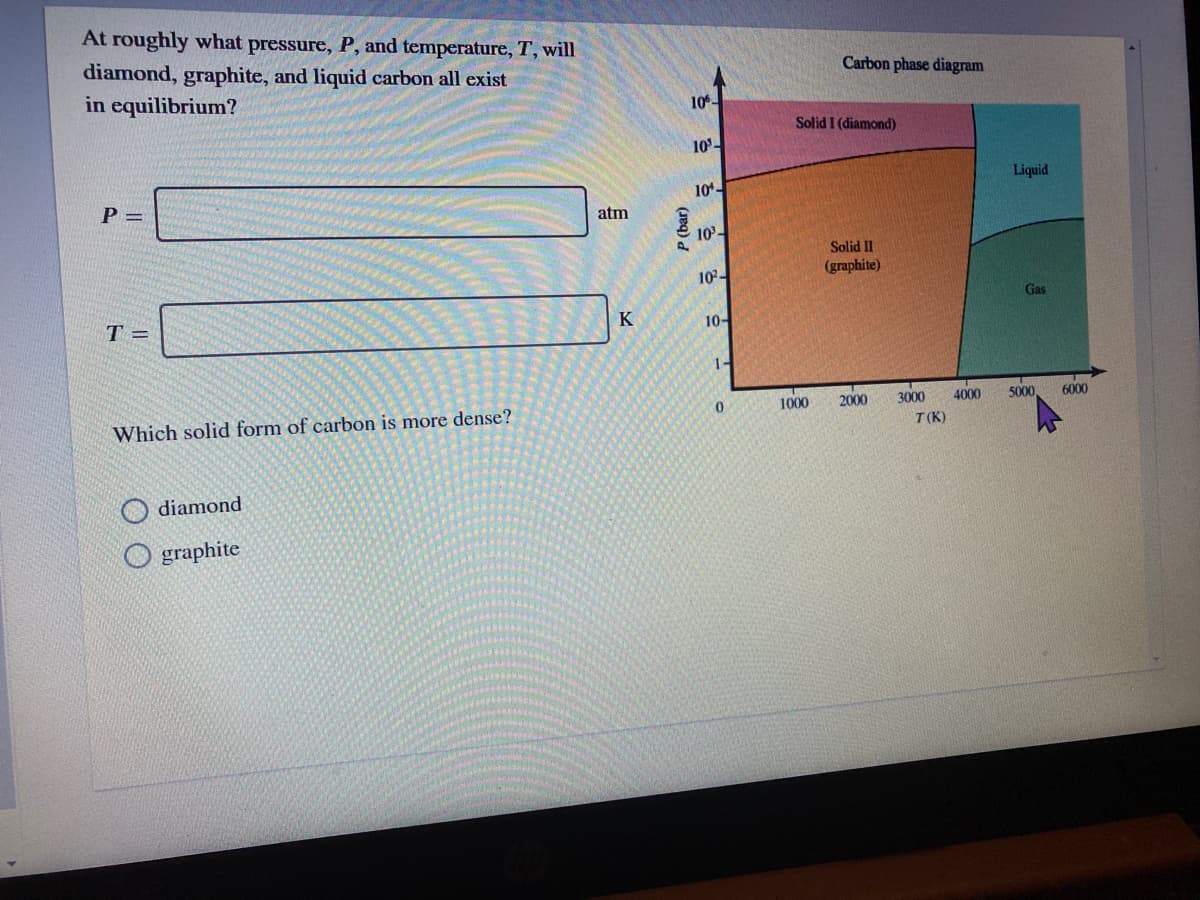
Transcribed Image Text:At roughly what pressure, P, and temperature, T, will
diamond, graphite, and liquid carbon all exist
in equilibrium?
Carbon phase diagram
10
Solid I (diamond)
10-
Liquid
P =
10-
atm
10-
Solid II
10-
(graphite)
Gas
T =
K
10-
1-
1000
2000
3000
4000
5000
6000
Which solid form of carbon is more dense?
T(K)
diamond
graphite
P (bar)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning