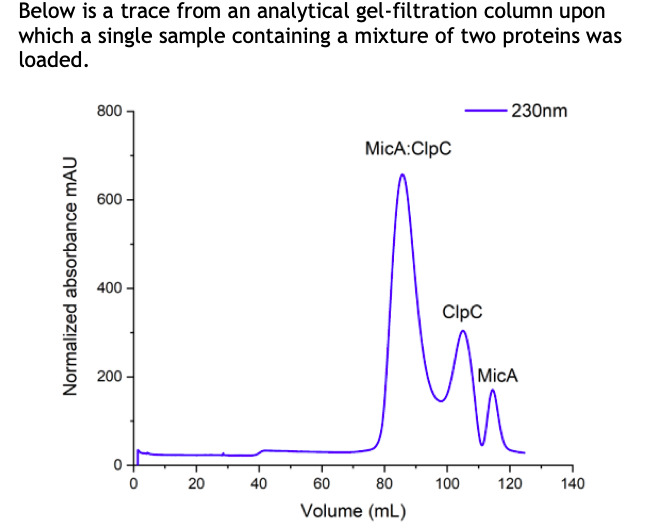
Below is a trace from an analytical gel-filtration column upon which a single sample containing a mixture of two proteins was loaded. - 230nm 800 MicA:ClpC 600 - 400 - ClpC 200 - MicA 20 40 60 80 100 120 140 Volume (mL) Normalized absorbance mAU
Q: Based on the information given, what separation technique(s) is/are most likely feasible? Please…
A: The selective boiling and subsequent condensation of a part in a liquid mixture is referred to as…
Q: You want to set up a 1:10 dilution series, so that you can plate a 10-1, 10-2, and a 10-3dilution;…
A: It is basically done for counting the microorganisms like viruses, bacteria etc from an unknown…
Q: Which of the following indicates buffering in the titration given in the image below? 12 11 10 Point…
A: A buffer is a aqueous solution which is used to resist the pH change upon addition of acid or base.…
Q: The table below summarizes the results for the solubility test. Which solvent the lipid samples are…
A: The solubility of a substance in a solvent depends on the nature of the solute and the solvent.
Q: Why are we using this materials and centrifuging at 15000 rpm? Lipid peroxidation experiment:…
A: The oxidative degeneration of lipids refers to lipid peroxidation, where the electrons from the…
Q: .A stock solution has a concentration of 10M. 50 mL of this stock solution is used to make 1L of a…
A: NOTE:- Since you have posted multiple questions so we will be solving the first one for you. As per…
Q: What are the mechanisms of samples separation work in Thin layer Chromatography?
A: Chromatography is a physicochemical technique for the division of compound mixtures, in view of the…
Q: (a) In an experiment to determine the glycine concentration of test sample A, the Ninhydrin method…
A: Absorbance of a solution is directly proportional to its concentration. This is as per Lambert Beer…
Q: In Gel filtration chromatography, when will you stop collecting eluents if sample is not colored?
A: Gel-filtration chromatography is a type of partition chromatography that is used to separate…
Q: A mixture of proteins contains Pepsinogen (35 kDa), Fumarase (49 kDa), Transferrin (80 kDa) and…
A: Chromatography refers to an analytical technique that is used to separate the chemical mixture into…
Q: Advantages and disadvantages of the following dosage forms: ○ Suspension ○ Parenteral Solutions ○…
A: A drug is described as a substance that is used for illness diagnosis, prevention, and treatment.…
Q: Table 1: Absorbance data @ 500nm for Glucose standards (0-20mM) and tests solutions Tube Number…
A: Standard curve for glucose concentration is constructed by taking concentration of glucose on x axis…
Q: Which of the following can most likely increase the yield of recrystallized benzoic acid? CHOICES:…
A: Recrystallization is a method that is used for the purification of solid compounds by the removal of…
Q: Construct a table showing the composition, advantage and disadvantage of these Platelet Diluting…
A: The fluid used as diluents must be isotonic, too have a certain high gravitational pull barrier cell…
Q: Which reagent removes contaminant proteins from DNA solution? Sodium dodecyl sulfate (SDS) O Sodium…
A: A major goal of the nucleic acid isolation is the removal of proteins . The seperation of nucleic…
Q: Samples Absorbance undiluted 0.715 1:4 dilution ratio 0.447 1:9 dilution ratio 0.321 a. Calculate…
A: From the given BSA standard absorbance data, I have plotted above linear XY graph and derived Y=mx+C…
Q: Which is not a Limit test in the Sodium Chloride drug substance/raw material monograph? (Note: NaCl…
A: The limit test analyzes and compares the effect of sodium chloride in the different samples like the…
Q: Label the void volume (V0), elution volume (Ve) and total elution volume (Vt) appropriately on the…
A: By the gel filtration chromatography the protein or other biological molecules of solutions…
Q: Results of testing for effect of concentration Tube Amount of Amount of Amount of Bubble enzyme in…
A: Catalase is a common antioxidant enzyme. It belongs to the oxidoreductase class. Catalase catalyzes…
Q: calculate the CFU/ml of a urine sample if 84 E. coli colonies were counted on NA when 50 microliters…
A: Serial dilution is the process of diluting a sample serially by taking the aliquot of one dilution…
Q: Briefly explain the principle behind the following techniques: (a) ion-exchange chromatography (b)…
A: Chromatography is a biophysical method for separating compounds from a mixture. It works on the…
Q: explain what the results of this experiment are and the purpose Table 2 Absorbance vs CoCl2…
A: The Beer-Lambert Rule: The concentration of the absorbing species and the route length of light…
Q: To achieve a hundred fold dilution you would add _____ microliters of sample to the appropriate…
A: The dilution method is used to achieve the desired concentration of the sample in a tube or a…
Q: Total Protein Determination Spectroscopy Values CREATE CALIBRATION CURVE? DRAWN CONCENTRATION AND…
A: The protein estimation in a sample can be done by analyzing and measuring the absorbance of the…
Q: What will be the results after you reconstitued a cefixime 60 ml a powder for suspension and stand…
A: A coarse dispersion suspension is a system in which an internal phase(also called the solid…
Q: By applying Gravimetry, Titration (Chemical indicator) and Potentiometry lessons which technique…
A: The technique of titration can be used for analyzing the total acid content per tablet for a newly…
Q: Using native chromatographic conditions which preserve enzymatic activity, what would be the…
A: Gel filtration chromatography is also known as size exclusion chromatography. Gel filtration…
Q: You are testing a sample of water from a swimming pool. You count a plate made from a 1 mL transfer…
A: Dilution is a process by which solution (water or diluents) are added to the solvent (main solution)…
Q: Using absorbance readings and a standard curve relating absorbance to cell number, you suspect that…
A: The countable range of colonies for an ideal plate count method is considered 30-300 colonies per…
Q: Using the sodium chloride equivalent method, make the following solution isotonic with respect to…
A: Any solute that dissolves will create an osmotic pressure when the solution is present across a…
Q: Below is a BSA standard curve and the duplicate values of absorbance for 3 samples. Calculate the…
A: Standard curve is made with known concentrations of Bovine serum albumin (BSA) to find the unknown…
Q: Drug Clearance For example, if the ClT of penicillin is 15mL/min in a patient and penicillin has a…
A: Clearance : rate of drug elimination divided by the drug plasma concentration
Q: A sample to be used for medical imaging is labeled with 18F,which has a half-life of 110 min. What…
A:
Q: Procedure Crude extract GFC IEX Volume (mL) 120 20 10 Protein Total content protein (mg/mL) (mg) 20…
A: Since you have asked a question with multiple subparts, we will answer first three subparts for you.…
Q: A sample is diluted by a factor of 10 five times. The 10^-3 dilution has 272 colonies on it.…
A: A serial dilution is a process of sequential dilution in order to decrease the density of cell…
Q: CFU/mL
A: in calculation via conventional formula , CFUs/ml in 2nd dilution are greater than 1st dilution…
Q: Make the following prescription isotonic with lacrimal fluid: 0.06 g Ephedrine sulfate (E = 0.23)…
A: Asked : Volume of sterilized distilled water (in milliliters) that should be added to the…
Q: Solubility Test Ninhydrin test iodoform test Observations + or - no colored spots on filter paper…
A: Asked : Fill the table with + or -
Q: A bacterial suspension is standardized using a 0.5 McFarland Standard. 1 mL of that bacterial…
A: Introduction: The stepwise dilution of a solution is referred to as serial dilution. Usually, the…
Q: Calculate the CFU/ml of a urine sample if 49 coli colonies were counted on NA when 50 microliters…
A: In both basic and applied research, microbiology is a highly significant area. Proteomics, genomics,…
Q: You are asked to add 2 mL of bacterial culture to 98 mL of water. What is your dilution factor? Show…
A: Introduction :- A test to determine whether you have a bacterial infection is a bacterium culture.…
Q: A 73-year-old male has been hospitalized for 2 days with fatigue and bone pain. His family stated…
A: The patients show normal values of calcium and phosphorus and the person is still experiencing…
Q: 1 mL of supernatant is required for a procedure. The final colored solution proves to be too high to…
A: Introduction: The concentration of a solution is an estimation of the amount of solute that is…
Q: Which molecules, or function groups, or complexes that may be responsible for the absorbance at 595…
A: Spectrophotometry applications are useful to measure the absorbance, reflectance, and transmission…
Q: Lead nitrate Stock Solution— Dissolve 159.8 mg of lead nitrate in 100 mL of water to which has been…
A:
Q: Granulation of powder is an essential step in pr of all types of tablets [] True
A: Tablets are solid dosage unit. It consists of active substances in the powder form and can be…
Q: Explain each type of anti-fouling system (application and procedure) Electrolytic system Chemical…
A: Anti fouling is a coating which can be done on the hull of any ship It prevents the overgrowth of…
Q: BIOLOGICAL BUFFERS functions for biological pH control rbonate Buffer Phosphate Buffer Protein…
A: The pH of a solution can be understood as a measure of its acidity or alkalinity (base) and the pH…
What is happening in this trace? What do the peaks represent? Why is absorbance measured at 230 nm?
Step by step
Solved in 2 steps

- Construct a standard curve (absorbance versus protein concentration in the standards) Conc. (mg/ml) 0 0.25 0.5 0.75 1.0 Absorbance 0.000 0.126 0.249 0.361 0.464Table 2. Volume of BSA, protein content, and absorbance readings of reference solutions Solution Volume of BSA standard solution (μL) Protein content(μg/mL) Absorbance value At 595 nm 1 0 0 0 2 10 1 0.022 3 30 3 0.065 4 50 5 0.106 5 70 7 0.178 6 100 10 0.299 7 120 12 0.380 Make a graph by plotting the absorbance values versus the BSA protein content (in μg) for theseven reference solutions. When constructing the graph, be…The physician has ordered 500,00 units of IV Pfizerpen every 4 hours. How much diluent will you add for a concentration of 500,000 units/mL? How many milliliters will you administer?
- You record the absorbance of your unknown BSA sample at 0.276. What is the concentration in ug/mL of the unknown assuming you have a standard curve with the line of best fit with y=0.0001x+0.0038 as its equationcalculate the CFU/ml of a urine sample if 84 E. coli colonies were counted on NA when 50 microliters were plated on the NA plate from a 10-6 dilution tubeWhich is not a Limit test in the Sodium Chloride drug substance/raw material monograph? (Note: NaCl is used as a drug to counter hypotension (low blood pressure), and as a raw material in saline solution.)
- Solution Absorbance mg/ml aspirin Standard solution - 1.6 mg/mL A 0.638 0.08 mg/mL B 0.504 0.064 mg/mL C 0.376 0.048 mg/mL D 0.259 0.032 mg/mL E 0.126 0.016 mg/mL A = -log T where T = %T ÷ 100 Construct a callibration curve using the above data. Absorbance should be on the vertical axis and "mg/mL of acetylsalicylic acid" on the horizontal axis. The line should go through the origin. Using the data provided, the graph you have generated, and the procedure that was used to generate the solutions which were examined by spectroscopy, calculate the amount of acetylsalicylic acid per tablet. Commercial tablet 1 labelled as 100 mg enteric coated Absorbance = 0.16 Commercial tablet 2 labelled as 300 mg Absorbance = 0.45 Student prepared tablet from practical 5 Absorbance = 0.19 Using the data provided, the graph you have generated, and the procedure that was used…can you show how to calculate the amount of two components for the required volume of a buffer with the receipes? Prepare enough 10X stock solution § Prepare 1LProportions§ 0.25M tris§ 1.92M glycine§ pH 8.3§ 1% (w/v) SDSInstructions to prepare 100mL of 10X running buffer1. Weigh 3.02g of Tris base and 14.42g of glycine into ~90mL of ddH2O in a clean beaker and stir todissolve.2. Check the pH with a calibrated pH metre once dissolved.§ The pH of the buffer should be 8.3. pH adjustment is not required if between 8.1-8.5.3. Add 5mL 20% (w/v) SDS stock solution (a liquid) to make a 1% (w/v) solution.4. Top the volume to 100mL by adding 5mL ddH2O from a graduated cylinder.The pH probe/meter uses following equations: Ecell = L + 0.0592 log a1 = L - 0.0592 pH Where L = L1 + EAg/AgCI + Easy= constants L1 = - 0.0592 log a2 a1 = activity of analyte solution a2 = activity of internal solution How will measured pH value be affected vs “real” pH if the temperature of the sample is 30C when pH was measured ? a.measured pH is lower than real pH b.impact can not be determined c.measured pH is higher than real pH d.measured pH is same as real pH





