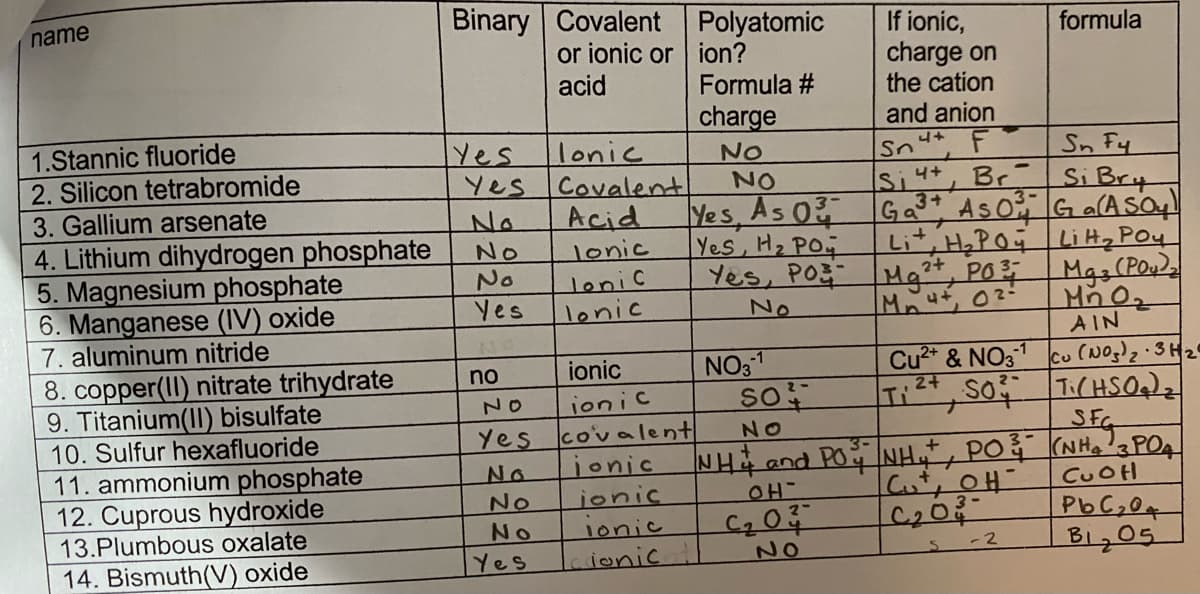
Binary Covalent Polyatomic or ionic or ion? name If ionic, charge on the cation formula acid Formula # charge and anion 1.Stannic fluoride 2. Silicon tetrabromide 3. Gallium arsenate 4. Lithium dihydrogen phosphate 5. Magnesium phosphate 6. Manganese (IV) oxide 7. aluminum nitride 8. copper(II) nitrate trihydrate 9. Titanium(II) bisulfate 10. Sulfur hexafluoride 11. ammonium phosphate 12. Cuprous hydroxide 13.Plumbous oxalate 14. Bismuth(V) oxide Yes lonic Yes Covalent Acid Sn Fy Si Bry ASOG a(ASo NO Sn NO Si4+ Br Yes, As 0 Yes, H2 PO4 Ga3+ Lit, H,PO Li Hz Poy Yes, Po Mg²*, PO Mgs(Po Mut, 02- No No lonic No Yes lonic lonic No AIN Cu2* & NO31 Cu (NO)2 3H2 NO31 so no ionic Ti, So TCHSO)z SFC 2- ionic covalent jonic ionic NO Yes NO No NH4 and PO NH, PO (NH,a POA No No Yes CUOH PbC,0+ B1,05 lonic -2 Laionic NO
Types of Chemical Bonds
The attractive force which has the ability of holding various constituent elements like atoms, ions, molecules, etc. together in different chemical species is termed as a chemical bond. Chemical compounds are dependent on the strength of chemical bonds between its constituents. Stronger the chemical bond, more will be the stability in the chemical compounds. Hence, it can be said that bonding defines the stability of chemical compounds.
Polarizability In Organic Chemistry
Polarizability refers to the ability of an atom/molecule to distort the electron cloud of neighboring species towards itself and the process of distortion of electron cloud is known as polarization.
Coordinate Covalent Bonds
A coordinate covalent bond is also known as a dative bond, which is a type of covalent bond. It is formed between two atoms, where the two electrons required to form the bond come from the same atom resulting in a semi-polar bond. The study of coordinate covalent bond or dative bond is important to know about the special type of bonding that leads to different properties. Since covalent compounds are non-polar whereas coordinate bonds results always in polar compounds due to charge separation.
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images









