Q: What cause the spikes in the spectrophotometer reading? Explain
A: In electronic spectroscopy, generally the electron excites from an initial low energy state to a…
Q: If the TLC plates had been developed in a more polar solvent, would retention factors on the whole…
A:
Q: Calculate the Re value for a spot in a TLC experiment if the solvent moved 14.5 cm and the spot…
A: Given Distance moved by solvent = 14.5 cm Spot of Compound moved from the origin = 6.1 cm Rf =…
Q: Calculate the Rf value for a spot in a TLC experiment if the solvent moved 14.9 cm and the spot…
A: Given values, Distance travelled by solvent = 14.9 cm Distance travelled by solute= 9.5 cm
Q: Consider the retention time of a pesticide is.13 8.5 min and the peak width at the base = 1.7 min,…
A: Given that, The retention time of a pesticide tr = 8.5 min Peak width at the base σt = 1.7 min…
Q: A student performs this and records the following data from the complted chromatogram. the solvent…
A: The Rf value is the ratio of distance travelled by the sample (b) to the distance travelled by the…
Q: An analgesic drug sample was analyzed using TLC to determine its probable active ingredient. The…
A: TLC is thin layer chromatography technique which is used to separate the mixture of non-volatile…
Q: Which of the following methods cannot be used to visualize spots on a TLC plate? Group of answer…
A: Which of the following methods cannot be used to visualize spots on a TLC plate? Group of answer…
Q: What is the Rf value for the uppermost yellow spot in TLC plate 3 (plate imaged below)? It is…
A: The TLC plate 3 with all the spots is given below :
Q: A spot of black ink was placed on a strip of filter paper. The chromatogram was developed using an…
A: The fastest moving spot has the highest Rf value. here we see C covered a long distance so it has…
Q: What adsorbents are typically used in TLC? On a TLC, a compound traveled 3.5 cm when the solvent…
A: Thin layer chromatography is the technique that is used to separate or purify the compound from a…
Q: How can you vary the retention factor and selectivity factor?
A: Retention factor is a variable which indicates the time duration a component spends in the…
Q: analgesic drug sample was analyzed using TLC to determine its probable active ingredient. The…
A:
Q: Which technique measures secondary structure? a. Size exclusion chromatography b. Circular Dichroism…
A: The secondary structure is formed by bending of the 1o structure. In this, an intermolecular force…
Q: Calculate the Rf value for a spot in a TLC experiment if the solvent moved 13.8 cm and the spot…
A: In the given TLC experiment the movent of the solvent is = 13.8 cm The movement of the spot is = 6.5…
Q: A student measures the solvent height on a TLC plate as 11.5 cm. Spot A was measured at a height of…
A: Formula used :- RF = distance traveled by compound/distance traveled by solvent
Q: If a spot travels 2.3 cm while the mobile phase travels 6.6 cm, what is the Rf value of the…
A: Given: Distance travelled by spot = 2.3 cm Distance travelled by mobile phase = 6.6 cm
Q: Before developing a TLC plate you should _______. a. Make sure to mark the origin with a pen b.…
A: While developing a TLC plate, we should always use a pencil (not pen) because: Pencils are cheap and…
Q: Why is it that a warm cuvette does not lose any significant heat during the absorbance measurement…
A: Cuvettes are designed to hold samples specially for spectroscopic measurement. Here the light beam…
Q: spot of black ink was placed on a strip of filter paper. The chromatogram was developed using an…
A:
Q: Why should you not use a ballpoint pen when marking a TLC plate and why does the horizontal line…
A: If ball point pen is used, the ink will travel up with the solvent, just like the samples. The…
Q: What is the design of figure II? FT IR interferometer single beam spectrophotometer…
A: The design of figure II is
Q: 13. Consider the TLC plate below to answer the question. What is the Rf of the top component? Note:…
A: RF factor is given by the distance travelled by the solute divided by distance traveled by solvent .…
Q: While using organic solvent like chloroform (CHCl3) in the visible region measurements what cuvette…
A: Glass cuvettes are typically used in the wavelength range of visible light, where as quartz tends to…
Q: Can you TYPE the definitions of the all of words please Chromatography TLC Stationary phase Mobile…
A: Most of the time, the compounds we encounter are not in their purest form. They are essentially a…
Q: A retention time of 85 s is obtained from a gas-chromatographic peak. The base width from the…
A:
Q: Calculate the Rf value of a spot if it travelled 1.5 cm while the solvent travelled 2.5 cm. Express…
A: Given : 1. Spot distance = 1.5 cm 2. Solvent distance = 2.5 cm
Q: Calculate the RF value for a spot in a TLC experiment if the solvent move 13.5 cm and the spot move…
A:
Q: How is the retention factor calculated for a specific spot when doing the TLC analysis? O Distance…
A: We have to tell the process of calculating of retention factor for a particular spot. The definition…
Q: Comment on the differences and similarities between a traditional UV light spectrophotometer and the…
A: Answer - UV-Vis spectrometers - UV-Vis spectrometers use a deuterium lamp for the UV that produces…
Q: 5. Using the diagram answer the questions below: Increasing time Sample appled to top of column…
A: To answer question related to chromatography:
Q: What error is introduced into the determination of an Rf value if the top is left off the developing…
A: In chromatographic technique, the components of a mixture are separated by using a developing…
Q: A gel filtration column has a radius (r) of 0.80 cm and a length (l) of 20.0 cm. Calculate the total…
A:
Q: the efficiency of a chromatographic column improves the measure that a.increases the height of the…
A: To determine what improves the efficiency of a chromatographic column:
Q: calculate Rfs for each spot on your chromatogram
A: Retention factor is equal to the ratio of distance travelled by solute divided by the distance…
Q: The peak parameters for your internal standard, evaluated using a capillary column with a length of…
A: HETP is defined as unit of coloumn length sufficient to bring solute in the mobile phase. It is a…
Q: At the completion of the experiment, a student measures the solvent front to be 55 mm from the…
A:
Q: From the experiment in Question 1, the following data were obtained: 2. Distance traveled by spot O…
A:
Q: what will produce a smaller Rf value if the solvent and paper are both changed?
A: Increasing the polarity of solvent will result in increase in Rf value as the compound in even for…
Q: Calculate the Rf value for a spot in a TLC experiment if the solvent moved 13.5 cm and the spot…
A:
Q: In thin layer chromatography, changing the mobile phase will alter the Rf value of a compound. O…
A: This can be solved as follows
Q: Question 19 In AFM, what is the consequence of low scanning time? limited magnification thermal…
A: AFM refers to Atomic Force Microscopy or Scanning force Microscopy. It is a high resolution type of…
Q: What is the Rf of the compound if the compound distance is 2.59 cm and the solvent front distance is…
A: Given that, Distance travelled by solute= 2.59 cm Distance travelled by solvent= 5.81 cm Then ,…
Q: Question 1. Two components in an HPLC separation have retention times that differ by 22 s. The…
A: HPLC ( High performance liquid Chromatography) It is a useful method for separation of substances…
Q: Does the amount of radiation (cpm) increase or decrease as the distance between the sorce and the…
A: Amount of radiation between the source and sensor is determine using the inverse square formula:…
Q: Calculate the Rf value of a spot that travels 5.7 cm, with a solvent front that travels 13 cm.
A: Rf is a retardation factor that is defined as the distance traveled by a substance divided by the…
Q: Rf of spot A in the TLC plate below is: Solvent front A 10 cm OB 8 cm 6 cm 2 cm Origin Select one: O…
A: The Rf value of a component depends on the distance travelled by the component and the distance…
Q: how does TLC determine if a sample is pure?
A: checking for purity of a sample, the known sample which is pure(having only one spot) and has the…

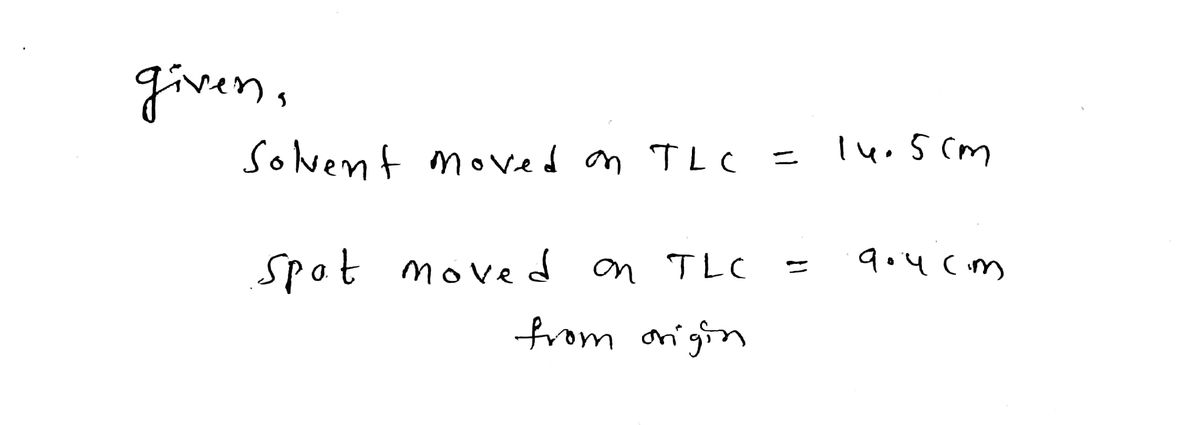
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Calculate the RF value for a spot in a TLC experiment if the solvent move 13.5 cm and the spot move 9.1 cm from the originCalculate the Rf value for a spot in a TLC experiment if the solvent moved 14.9 cm and the spot moved 9.5 cm from the origin.Calculate the Rf value of a spot that travels 5.7 cm, with a solvent front that travels 13 cm.
- What would be the effect of a thin alumina on the retention, R,? A.Higher Rf B.Lower Rf C.No effect D. Cannot be determinedIt is acceptable to draw the TLC spotting line with a pen as long as it is marked very lightly. True FalseAt the completion of the experiment, a student measures the solvent front to be 55 mm from the baseline. If a component in the sample being analyzed generates a spot whose diameter spans from 34 to 36 mm from the baseline, calculate the Rf value for that component.
- If a TLC was run in a solvent system that caused all the spots to show up at the baseline, how could the system be adjusted so that Rf values could be determined?If a spot travels 2.3 cm while the mobile phase travels 6.6 cm, what is the Rf value of the substance in the spot? What is the mobile phase used and stationary phase used in this lab?If the TLC plates had been developed in a more polar solvent, would retention factors on the whole be larger, smaller, or unchanged? Briefly (1 - 2 sentences) explain your reasoning
- Does the amount of radiation (cpm) increase or decrease as the distance between the sorce and the sensor increases? ExplainCalculate the Rf value if the solvent moved 13.3 cm and an ink component moved 8.7 cm.If you run a compound on 4 different TLC plates using the following four different solvent systems which solvent system would give the largest Rf value for the compound?



