Chalcopyrite (CuFeS2), is an important source of copper. A typical chalcopyrite ore contains about 0.75% Cu. What volume of sulfur dioxide at 25°C and 1 atm pressure is produced when one boxcar load ( 4 x 10° ft³) of chalcopyrite ore (density = 2.6 ) is roasted? Assume all the sulfur in cm3 the ore is converted to SO2 and no other source of sulfur is present. Find the following: (do not express your answer in scientific notation, do not input the unit, space or comma, your answer should be in two decimal places) a. mass in grams of the chalcopyrite ore = g b. mass in grams of copper = c. no.of moles of SO2 = mol %3D d. volume of SO2 = %3D Note: Use the following in solving the problem 1 ft = 30.48 cm For atomic weight (see periodic table in your module) R= 0.082057 Leatm moleK
Chalcopyrite (CuFeS2), is an important source of copper. A typical chalcopyrite ore contains about 0.75% Cu. What volume of sulfur dioxide at 25°C and 1 atm pressure is produced when one boxcar load ( 4 x 10° ft³) of chalcopyrite ore (density = 2.6 ) is roasted? Assume all the sulfur in cm3 the ore is converted to SO2 and no other source of sulfur is present. Find the following: (do not express your answer in scientific notation, do not input the unit, space or comma, your answer should be in two decimal places) a. mass in grams of the chalcopyrite ore = g b. mass in grams of copper = c. no.of moles of SO2 = mol %3D d. volume of SO2 = %3D Note: Use the following in solving the problem 1 ft = 30.48 cm For atomic weight (see periodic table in your module) R= 0.082057 Leatm moleK
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter8: Molecules And Materials
Section: Chapter Questions
Problem 8.88PAE
Related questions
Question
Please solve this
ACTIVITY F
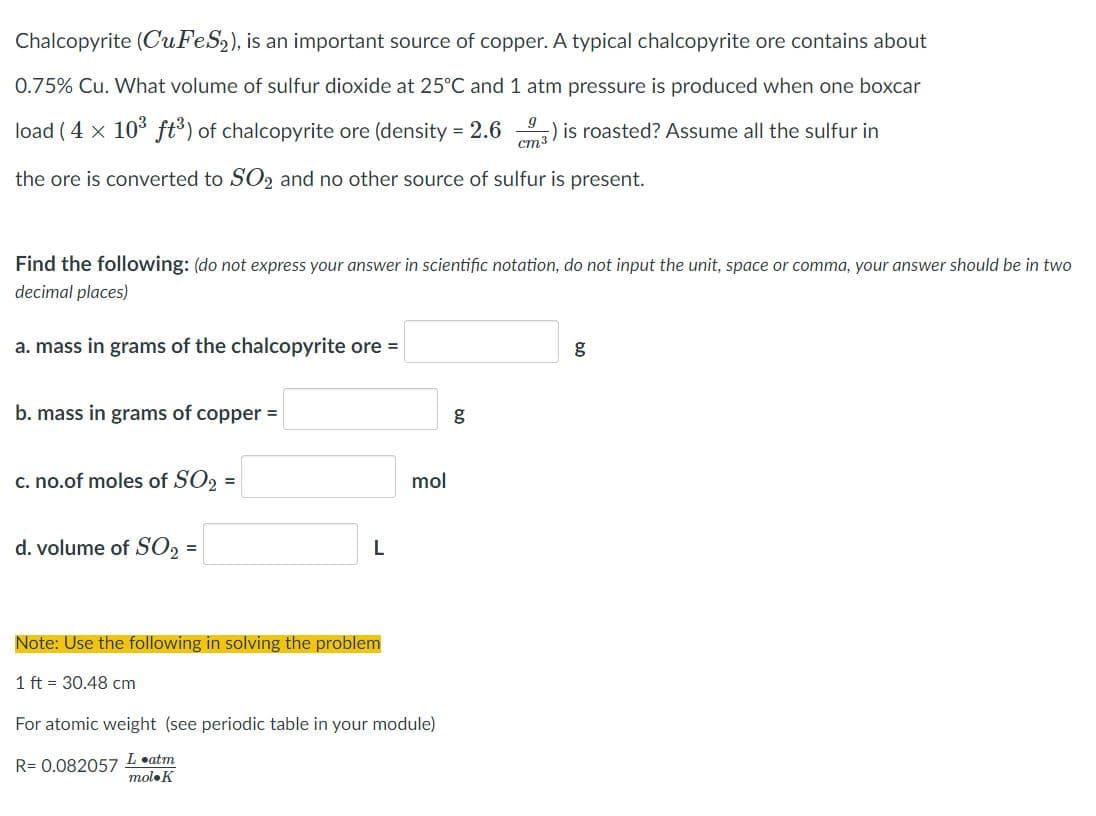
Transcribed Image Text:Chalcopyrite (CuFeS2), is an important source of copper. A typical chalcopyrite ore contains about
0.75% Cu. What volume of sulfur dioxide at 25°C and 1 atm pressure is produced when one boxcar
load ( 4 x 10 ft) of chalcopyrite ore (density = 2.6 ) is roasted? Assume all the sulfur in
cm3
the ore is converted to SO2 and no other source of sulfur is present.
Find the following: (do not express your answer in scientific notation, do not input the unit, space or comma, your answer should be in two
decimal places)
a. mass in grams of the chalcopyrite ore =
g
b. mass in grams of copper =
c. no.of moles of SO2 =
mol
d. volume of SO =
Note: Use the following in solving the problem
1 ft = 30.48 cm
For atomic weight (see periodic table in your module)
R= 0.082057 Leatm
moloK
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning