Classify each of these reactions. Pb(NO,),(aq) + 2 KCI(aq) PbCl, (s) +2 KNO,(aq) - CH, +30,(g) –2C0,(g) + 2H,0(1) | 2 AGNO, (aq)+Zn(s) Zn(NO,),(aq) +2 Ag(s) - 2 KOH(aq) + H, SO, (aq) K,SO, (aq) + 2H,O(1) - Answer Bank precipitation redox acid-base neutralization
Classify each of these reactions. Pb(NO,),(aq) + 2 KCI(aq) PbCl, (s) +2 KNO,(aq) - CH, +30,(g) –2C0,(g) + 2H,0(1) | 2 AGNO, (aq)+Zn(s) Zn(NO,),(aq) +2 Ag(s) - 2 KOH(aq) + H, SO, (aq) K,SO, (aq) + 2H,O(1) - Answer Bank precipitation redox acid-base neutralization
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter7: Reactions In Aqueous Solutions
Section: Chapter Questions
Problem 9CR: n general terms, what are the spectator ions in a precipitation reaction? Why are the spectator ions...
Related questions
Question
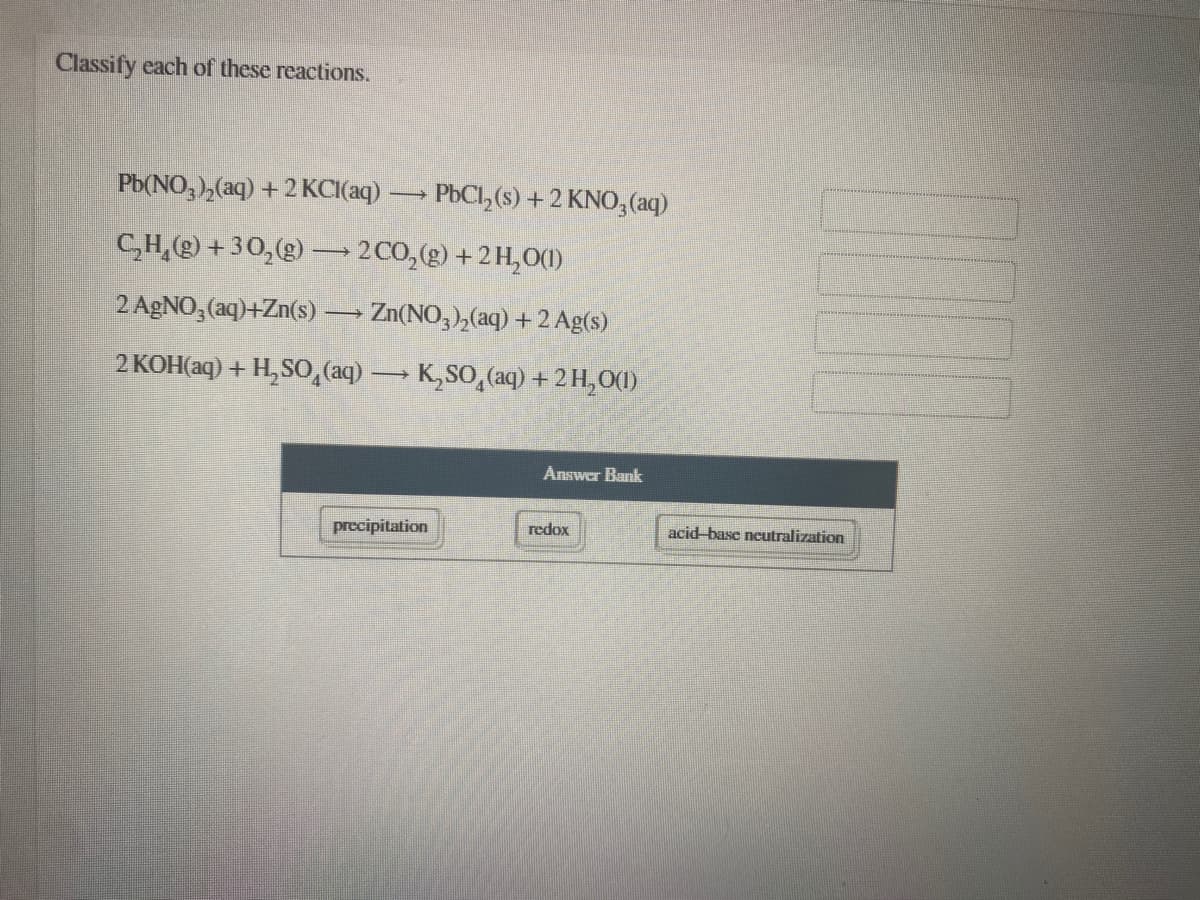
Transcribed Image Text:Classify each of these reactions.
Pb(NO, ),(aq) + 2 KCI(aq)
PbCl, (s) + 2 KNO,(aq)
C,H,) +30,(g) – 2C0,(g) +2 H,0()
2 AGNO, (aq)+Zn(s)
Zn(NO, ),(aq) + 2 Ag(s)
2 KOH(aq) + H,SO, (aq) K,SO,(aq) +2H,O()
Answer Bank
precipitation
redox
acid-base neutralization
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning
