college.com/course.html?courseld=154322748HeplD=2b3e48e6520860bfd5591538a4a5a27b#10001 Search... A AOL Video-Serving the best vi... TripAdvisor 34 of 40 Calculate the specific heat (J/g °C) for a 18.5-g sample of tin that absorbs 183 J when temperature increases from 35.0 °C to 78.6 °C. V AZ ΑΣφ SH = J/g °C Request Answer Submit Part B Calculate the specific heat (J/g °C) for a 23.0 g sample of a metal that absorbs 669 J when temperature increases from 42.1 °C to92.3 °C. ΑΣ. P Pearson Contact Us Privacy Policy Terms of Use Permissions Copyright © 2019 Pearson Education Inc. All rights reserved. 8:51 PM 12/4/2019 Cip Ins prt sc dele fg fg f6 144 fs & %23 back 8. 4. 7. K H. pau N. C. Σ %24 %23
college.com/course.html?courseld=154322748HeplD=2b3e48e6520860bfd5591538a4a5a27b#10001 Search... A AOL Video-Serving the best vi... TripAdvisor 34 of 40 Calculate the specific heat (J/g °C) for a 18.5-g sample of tin that absorbs 183 J when temperature increases from 35.0 °C to 78.6 °C. V AZ ΑΣφ SH = J/g °C Request Answer Submit Part B Calculate the specific heat (J/g °C) for a 23.0 g sample of a metal that absorbs 669 J when temperature increases from 42.1 °C to92.3 °C. ΑΣ. P Pearson Contact Us Privacy Policy Terms of Use Permissions Copyright © 2019 Pearson Education Inc. All rights reserved. 8:51 PM 12/4/2019 Cip Ins prt sc dele fg fg f6 144 fs & %23 back 8. 4. 7. K H. pau N. C. Σ %24 %23
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter6: Thermochemistry
Section: Chapter Questions
Problem 143IP: A cubic piece of uranium metal (specific heat capacity = 0.117 J/C g) at 200.0C is dropped into...
Related questions
Question
Transcribed Image Text:college.com/course.html?courseld=154322748HeplD=2b3e48e6520860bfd5591538a4a5a27b#10001
Search...
A
AOL Video-Serving the best vi...
TripAdvisor
<MC 13 on Temperature, Changes in Energy, and Gases
Item 34
<>
34 of 40
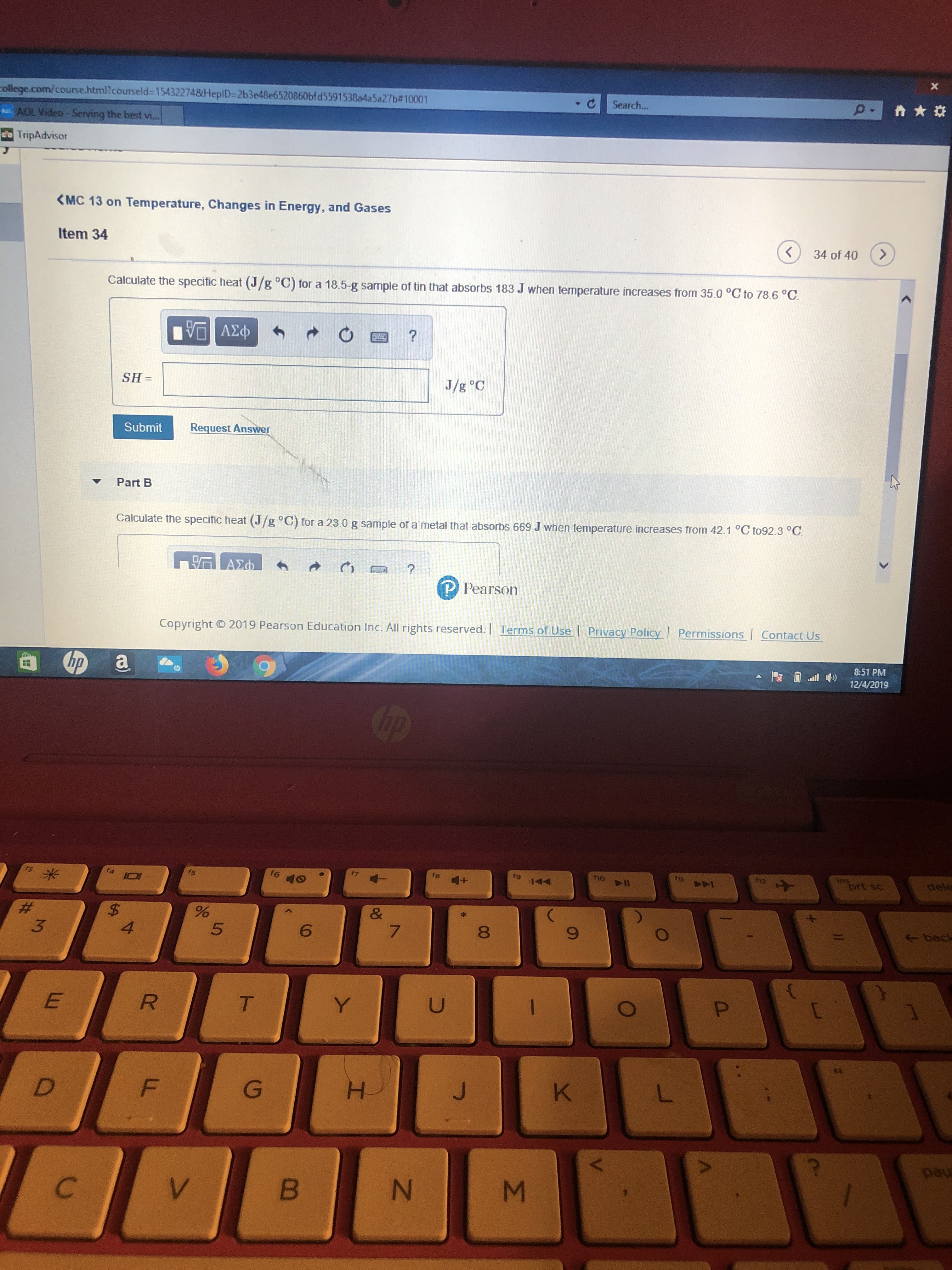
Calculate the specific heat (J/g °C) for a 18.5-g sample of tin that absorbs 183 J when temperature increases from 35.0 °C to 78.6 °C.
V AZ
ΑΣφ
SH =
J/g °C
Request Answer
Submit
Part B
Calculate the specific heat (J/g °C) for a 23.0 g sample of a metal that absorbs 669 J when temperature increases from 42.1 °C to92.3 °C.
ΑΣ.
P Pearson
Contact Us
Privacy Policy
Terms of Use
Permissions
Copyright © 2019 Pearson Education Inc. All rights reserved.
8:51 PM
12/4/2019
Cip
Ins
prt sc
dele
fg
fg
f6
144
fs
&
%23
back
8.
4.
7.
K
H.
pau
N.
C.
Σ
%24
%23
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning