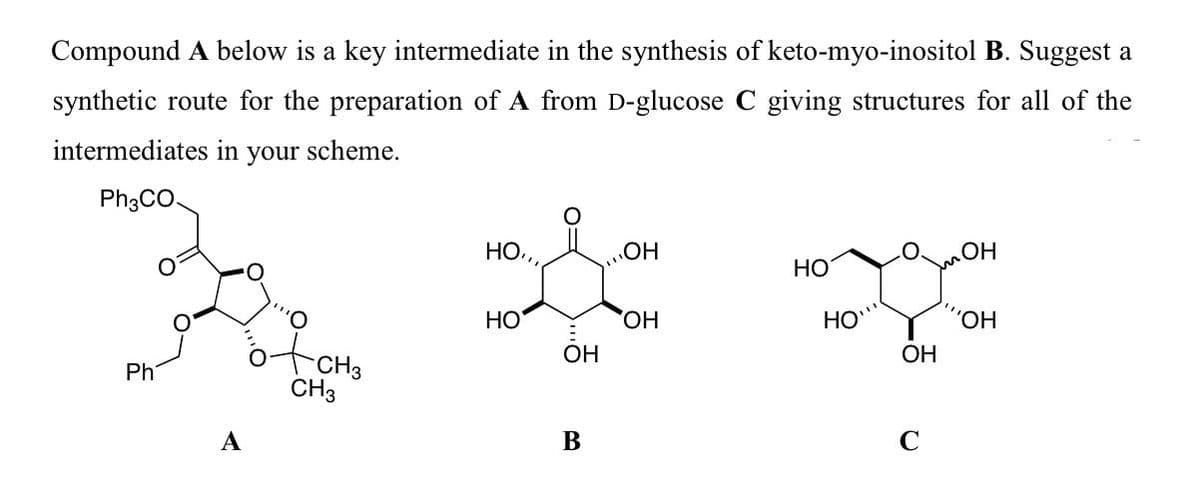
Compound A below is a key intermediate in the synthesis of keto-myo-inositol B. Suggest a synthetic route for the preparation of A from D-glucose C giving structures for all of the intermediates in your scheme. Ph3CO. Но, „OH HO НО HO, HO 'HO, OH ОН Ph CH3 A В
Q: What is the IUPAC name of the 2nd fatty acid ? * H-c-o- .C 'C H. H. H. H H. H-c-o- H. H. H. H. H. !し...
A: A fatty acid is a carboxylic acid with an aliphatic chain.
Q: 9) Propose a synthetic sequence for the dipeptide A-D-G (single-letter codes) starting from the free...
A: Hi! Since you have posted a multipart question , we will solve the first 3 subparts for you accordin...
Q: Well 1 contains 20 uL of serum. Wells 2-6 contain 90 uL of water. To do a serial dilution of 10% c...
A: 10% serial dilution is mixing 1 part of stock solution with 9 parts of diluent so that concentration...
Q: What are the concepts of specificity, competition, and saturation as they relate to enzymes. Include...
A: Enzymes are biocatalyst that increases the speed of reaction by lowering the activation energy....
Q: The conversion of succinate to fumarate in the TCA cycle is shown below. Need carbon a and b ans...
A: The series of chemical reactions that occur inside the living body for the production of energy are ...
Q: Prokaryotic cells are very small and appear to be restricted in terms of their size, but eukaryotic ...
A: A prokaryotic cells ranges in size from 0.1 to 5.0 micrometers in diameter. An eukaryotic cells are ...
Q: In First order, the rate of reaction would be . ., if the concertation of an enzyme is increased by ...
A: The reaction may be classified according to the order of reaction, which is the number of reacting s...
Q: how can accelerate the chemical reaction
A: Chemical reaction: A chemical reaction is a process where the reactant gets converted into a produ...
Q: What biotechnology product that has improved life, and what would our life be without this product?
A: Biotechnology is a technology that uses living beings and biological processes to bring new solution...
Q: Explain why all mono- and disaccharides are soluble in water? What are some examples of artificial s...
A: All mono- and disaccharides are soluble in water. Monosaccharides are glucose, fructose, and galacto...
Q: What will be the color of saliva extraction with iodine in 3, 6 9, 12, 15, and 18 mins if: you put 5...
A: Carbohydrates are divided into 3 classes monosaccharide, disaccharide, and polysacchari...
Q: In a sample dsDNA from an organism, the amount of thymine is analyzed to be 12 µmoles when the A+T/G...
A: DNA is also known as deoxyribonucleic acid. DNA acts as genetic material in most organisms. DNA is c...
Q: A. Characteristic Tests of Carbohydrates Sample Barfoed's Molisch Test Benedict's Test lodine's Test...
A: Benedict’s Test :- used to test for simple carbohydrates. The Benedict’s test identifies reducing s...
Q: Draw the Haworth projection of β-D-Altopyranose given the structure of D-Altrose.
A: Altrose is an aldohexose. Altrose is a C-3 epimer of mannose. The Fischer projection shows the open ...
Q: The fact the DNA bears the genetic information of an organism implies that A Base composition should...
A: All living organisms have cells, which are the most basic and essential unit of life. Cells are resp...
Q: Fatty Acids are good fuels and release energy in the form of heat. This is primarily because: a) Hy...
A: Fatty acid oxidation occurs in the mitochondria and is an aerobic process of breaking down a fatty a...
Q: Explain the importance of buffers and what are the main buffers in the body?
A: Almost all the biological processes are pH-dependent. A small change in the pH creates a drastic cha...
Q: Each of the following statements concerning mitochondria is true, EXCEPT a.The mitochondria require...
A:
Q: A) illustrate in molecular detail how hemoglobin's reduced oxygen affinity is caused by protonation ...
A: In oxygen-breathing animals, hemoglobin (Hb) has developed as an efficient transport protein that tr...
Q: The coenzyme is
A: Coenzymes are cofactors (non-protein component) that help in the completion of a reaction by boostin...
Q: True or False? The 24-h LC50 is typically lower than the 96-h LC50 bioassays.
A: In toxicology, a dose descriptor is used to find the relationship between a specific effect of chemi...
Q: Discuss the role of mould in food process industry and analyze the process of one food product that ...
A: Role of mould in food process industry- 1. Moulds are basically a fungus that are used in the prepa...
Q: A protein P has two identical binding sites for a molecule A. The shape of the protein is dependent ...
A: Proteins are organic polymers of amino acids which are folded in three dimensional structure by both...
Q: How do I convert from DPM to picomoles per milligram of protein (pmol/mg)? I have the specific activ...
A: DPM refer to Disintegration per minute for a Radioactive decay. When a compound is labeled with a ra...
Q: Consider the following free energy diagram for an uncatalyzed and enzyme-catalyzed reaction. Select ...
A: Those proteins or biological catalysts which help to speed up the chemical reaction are termed enzym...
Q: POST-LAB QUESTIONS E. Seliwanoff's Test 1. Describe the evidence for a positive test. 2. What does a...
A: Most of the carbohydrates can be distinguished from each other based on the condensation reaction us...
Q: How to use the sterile filter? Show the different sizes and specifications of sterile filters.
A: Sterilization is the method that is used to eliminate all forms of life and biological agents presen...
Q: I. Name the following carbohydrates using the proper way of naming/IUPAC system. a. Erythrose b. Lyx...
A: Since there are 3 questions asked, we will solve the first question. If you need the solutions of th...
Q: Create 5 water molecules with appropriate bond formation. You must Include images of the water molec...
A: The main constituent of Earth's hydrosphere is water and all Known living organisms' fluid ...
Q: 1. What is the chemical basis for a positive test in Barfoed's Test? 2. What is the chemical basis f...
A: The qualitative tests are used to detect the presence or absence of a substance. Different qualitati...
Q: SDS-PAGE gels are useful in determining the molecular weights of proteins; however, the molecular we...
A: SDS-PAGE is an acronym for sodium dodecyl sulfate–polyacrylamide gel electrophoresis. It’s a mass-ba...
Q: In a sample dsDNA from an organism, the amount of thymine is analyzed to be 12 μmoles when the A+T/G...
A: In double stranded DNA the composition of bases follows the rule: A+T= G+C.
Q: Water production from fatty acid oxidation is a survival mechanism in animals where water is scarce....
A: Water is one of the essential elements for most living things. It is found in all cells, tissues, an...
Q: what molecule is responsible for protein synthesis *
A: Proteins are polymers of amino acids, linked by amide/peptide bond with release of a water molecule....
Q: Complete the following paragraph describing the fate of the second 14CO-acetyl-CoA molecule incorpor...
A: The glyoxylate produced in the isocitrate reaction combines with 2nd molecule of acetyl coa in the m...
Q: It's important to note that MnO is an alkaline metal, whereas Mn207 is an acidic metal. Why? (ii) Al...
A: Introduction Chemical compounds are a critical component of life. Organic substances are those deriv...
Q: Order the following from 1-12 events chronologically based on the next generation sequencing methodo...
A: Genome sequencing is a biotechnological technique which is prominently used in sequencing the genome...
Q: What determines the characteristics of an amino acid?
A: Amino acids are organic compounds with two functional group namely amino and carboxyl. There are 20...
Q: Materials: Pipette/pipet Volumetric Flask Reagent Bottle Reagent Classifications: (USP, CP, AR, ...
A: Pipette, volumetric flask, reagent bottle and reagents are used in a laboratory for performing exper...
Q: In moelcular genetics, initiation is often accomplished using proteins that prevent elongation. Nam...
A: Initiation of a process in genetics is an important event. At this stage, cell prepare itself for th...
Q: Chemically, Olestra is made of a six-carbon backbone and six fatty acid attachments. No nutrients ar...
A: Olestra- Indigestible fat and composed of 6 fatty acid attachments.
Q: D. Barfoed's Test 1. Describe the evidence for a positive test. 2. What does a positive test indicat...
A: Barfoed's test was introduced by Thomas Barfoed. This test is used for the detection of the presence...
Q: D- galactose and L- galactose is . O Structural isomer O Enantiomers O Epimers O Anomers O Stereoiso...
A: Answer D- Galactose and L- Galactose is Enantiomers.
Q: 1. Place 5 mL of starch solution in the test tubes. 2. Heat the test tubes to boiling and add to 1 ...
A: The food we consume is broken down to simpler molecules that are used to yield energy for the body. ...
Q: Using diagrams of your own design, demonstrate how pH affects the charge distribution of an amino ac...
A: Amino acid is a compound that contains both amine group and a carboxyl group in a same molecules. ...
Q: nucleic acid. b) . What is/are the major chemical difference(s) between RNA and DNA?
A: It is the question about nucleic acid i. DNA and RNA. Both DNA and RNA are nucleic acid but though ...
Q: Qualitative Analysis of Proteins 1. Fill out the table below by providing the necessary information...
A: Proteins are composed of amino acids, which are bound together by peptide linkage. Amino acids conta...
Q: The allosteric inhibitor of an 1 point enzyme Causes the enzyme to work faster O Binds to the active...
A: Answer Tha allosteric inhibitor of an enzyme participates in feedback regulation
Q: 6. Kwashiorkor is the discase causcd by a deficiency of proteins in the diet that is adequate in cal...
A: Protein deficiency (in Kwashoirkor) and hence the lack of protein reduces the production of lipoprot...
Q: 14 If we start with glucose-1-P, with carbon #1 being "C labeled, what happens to this carbon at the...
A: Embeden Myerhoff Parnas (EMP) and Entner-Doudoroff (ED) Pathways are two pathways for Glucose breakd...
Step by step
Solved in 2 steps with 1 images

- Consider the synthesis and degradation of tyrosine as shown in Figures 25.37, 25.38, and 25.48 to determine where the carbon atoms in PEP and erythrose-4-P would end up in acetoacetate and fumarate.Figure 27.3 illustrates the response of R (ATP-regenerating) and U (ATP-utilizing) enzymes to energy charge. a. Would hexokinase be an R enzyme or a U enzyme? Would glutamine: PRPP amidotransferase, the second enzyme in purine biosynthesis, be an R enzyme or a U enzyme? b. If energy charge = 0.5: Is the activity of hexokinase high or low? Is ribose-5-P pyrophosphokinase activity high or low? c. If energy charge = 0.95: Is the activity of hexokinase high or low? Is ribose-5-P pyrophosphokinase activity high or low?DCCD (diocyclohexylcarbodiimide) inhibits oxidative phosphorylation when the substrate is mitochondrial NADH. DCCD is a drug that binds to ATP synthase and blocks proton transport through the ion channel. a) Explain what the consequences of DCCD on cellular energy production are. b) Suggest at least one other cellular effect of DCCD and explain this effect.
- . Students carry out a laboratory experiment with avidin (a protein in eg white) having a very high aflinity for biotin enzymes. Which enzyme of glucose metabolism would be inhibited by the addition of avidin to a cell homogenate? For answer: a) draw a reaction that would be blocked by the addition of avidin to a cell homogenate; b) write a diagram of the carbohydrate metabolism in which this reaction takes place.Select the chemical events that occur between isopentenyl pyrophosphate and lanosterol during cholesterol synthesis: Choose ALL that apply. (A) phosphorylation and decarboxylation of mevalonate(B) condensation of acetyl CoA(C) pyrophosphate (PPi) hydrolysis(D) condensation of C5 units to form C10 and C15 intermediates (E) formation of a tetracyclic intermediate (F) ATP hydrolysisIn another key reaction in glycolysis, dihydroxyacetone phosphate (DHAP)is isomerized into glyceraldehyde-3-phosphate (GAP): Because ΔG°′ is positive, the equilibrium lies to the left.(a) Calculate the equilibrium constant and the equilibrium fraction of GAPfrom the above, at 37 °C.(b) In the cell, depletion of GAP makes the reaction proceed. What will ΔGbe if the concentration of GAP is always kept at 1/100 of the concentrationof DHAP?
- a. Use the values in Problem 23.31 to calculate the energy change in the following reaction. fructose 1,6-bisphosphate + ADP--------> fructose 6-phosphate + ATP b. Is this reaction energetically favorable or unfavorable? c. Write this reaction using curved arrow symbolism. d. Can this reaction be used to synthesize ATP from ADP? Explain.Which of the statements below regarding glyphosate are true? (Choose all true statements) A.Glyphosate interacts with EPSP synthase because it resembles the substrate S-3P B.The interaction of S-3P is necessary for glyphosate binding. C.Glyphosate likely binds to both conformations of EPSP synthase shown in question 7. D.Glyphosate decreases Km for S-3P and thus likely the increases the affinity of the enzyme for substrate E.Glyphosate likely increases feedback inhibition by the product of the reaction EPSPIntramitochondrial ATP concentrations are about 5 mM, and phosphate concentration is about 10 mM. Consider that ADP is five times more abundant than AMP. a. Calculate the molar concentrations of ADP and AMP at an energy charge of 0.85. b. Calculate ∆G' for ATP hydrolysis under these conditions (∆G0' for ATP hydrolysis is -32.2 kJ/mol) The energy charge is defined as ( [ATP] + 1/2 [ADP] ) / ( [ATP] + [ADP] + [AMP] )
- Which of the following statements about the malate-aspartate shuttle is false? a) 2.5 ATPs are formed per cytoplasmic NADH shuttled. b) Transamination is involved. c) Cytoplasmic oxaloacetate is converted to malate. d) Mitochondrial malate is converted to oxaloacetate. e) Antiport transport is not involved.Select the chemical events that occur between mevalonate and geranyl pyrophosphate during cholesterol synthesis: Choose ALL that apply. (A) decarboxylation to create a C5 molecule from a C6 precursor (B) condensation of (2) C5 molecules to form a C10 intermediate (C) reduction of HMG CoA(D) ATP hydrolysis (E) isomerization of isopentenyl pyrophosphate(F) condensation of a C5 molecule and a C10 molecule to form a C15 intermediate (G) formation of a tetracyclic intermediate(H) pyrophosphate (PPi) hydrolysisWhich of these is associated with production of ATP during glycolysis? (Select all that apply) a) Conversion of 1,3-BPG to 3PG b) Conversion of 1,3-BPG to 2,3-BPG c) Conversion of PEP to pyruvate d) Conversion of 2,3-BPG to 2PG e) Phosphorylation of glucose

