Compound D shows the following IR spectrum. 100 80 60 %T 40 3037 1650 20 1739 1678 4000 3000 2000 1500 1000 500 wavenumber, cm-1 (i) Match the spectrum with any of the suitable compounds listed below. ОН OH -CEN NEC- II II IV VI (ii) Explain your answer in b(i).
Compound D shows the following IR spectrum. 100 80 60 %T 40 3037 1650 20 1739 1678 4000 3000 2000 1500 1000 500 wavenumber, cm-1 (i) Match the spectrum with any of the suitable compounds listed below. ОН OH -CEN NEC- II II IV VI (ii) Explain your answer in b(i).
Chapter12: Structure Determination: Mass Spectrometry And Infrared Spectroscopy
Section12.SE: Something Extra
Problem 48AP: The infrared spectrum of the compound with the mass spectrum shown below lacks any significant...
Related questions
Question
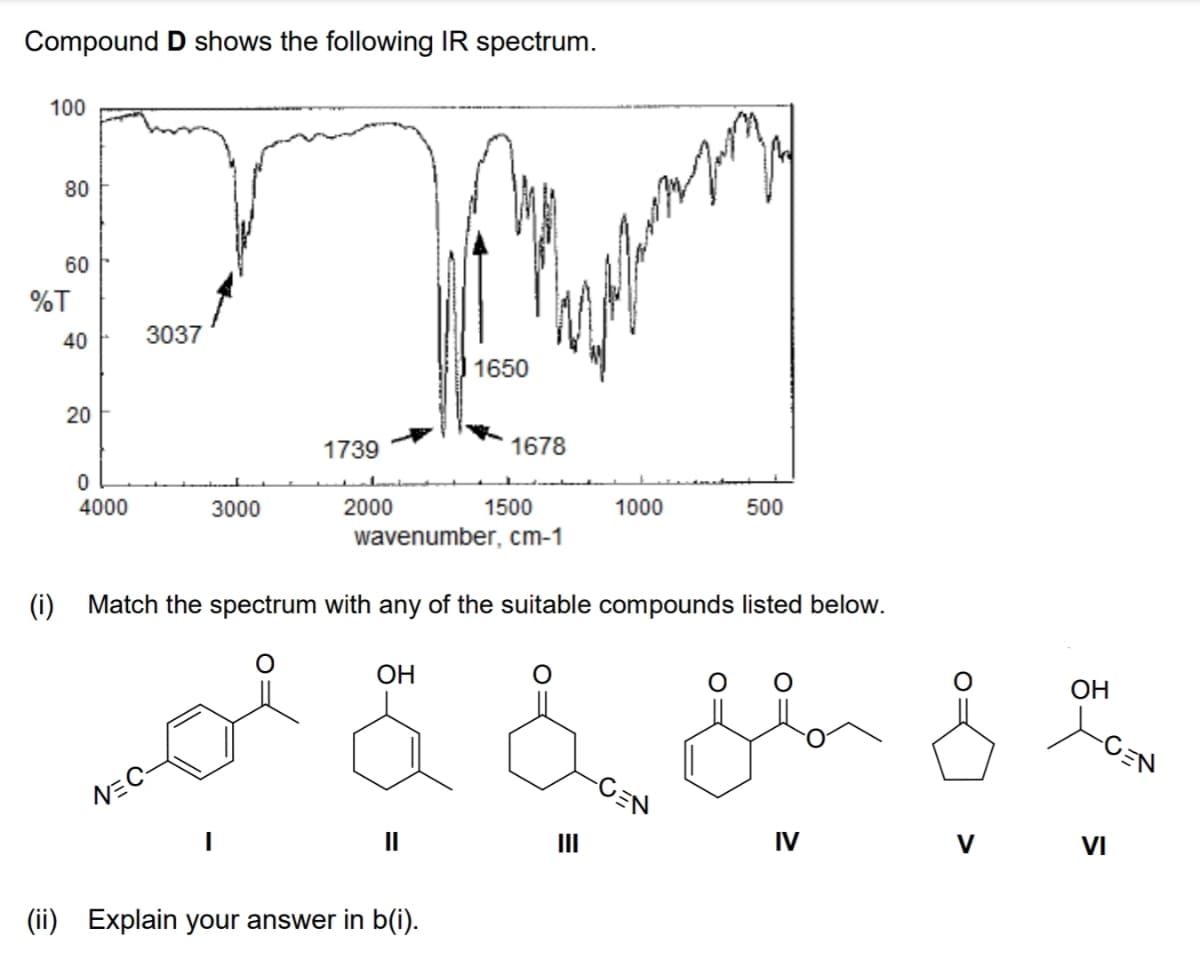
Transcribed Image Text:Compound D shows the following IR spectrum.
100
80
60
%T
40
3037
1650
1739
1678
4000
3000
2000
1500
1000
500
wavenumber, cm-1
(i) Match the spectrum with any of the suitable compounds listed below.
ОН
OH
-CEN
-CEN
NEC-
II
II
IV
V
VI
(ii)
Explain your answer in b(i).
20
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

