Concentrated sulphuric acid is used in this experiment to O help the crystal growth of acetylsalicylic acid speed up the reaction of aspirin synthesis increase the activation energy avoid impurities getting trapped into the aspirin crystals
Concentrated sulphuric acid is used in this experiment to O help the crystal growth of acetylsalicylic acid speed up the reaction of aspirin synthesis increase the activation energy avoid impurities getting trapped into the aspirin crystals
Chapter30: Kinetic Methods Of Analysis
Section: Chapter Questions
Problem 30.12QAP
Related questions
Question
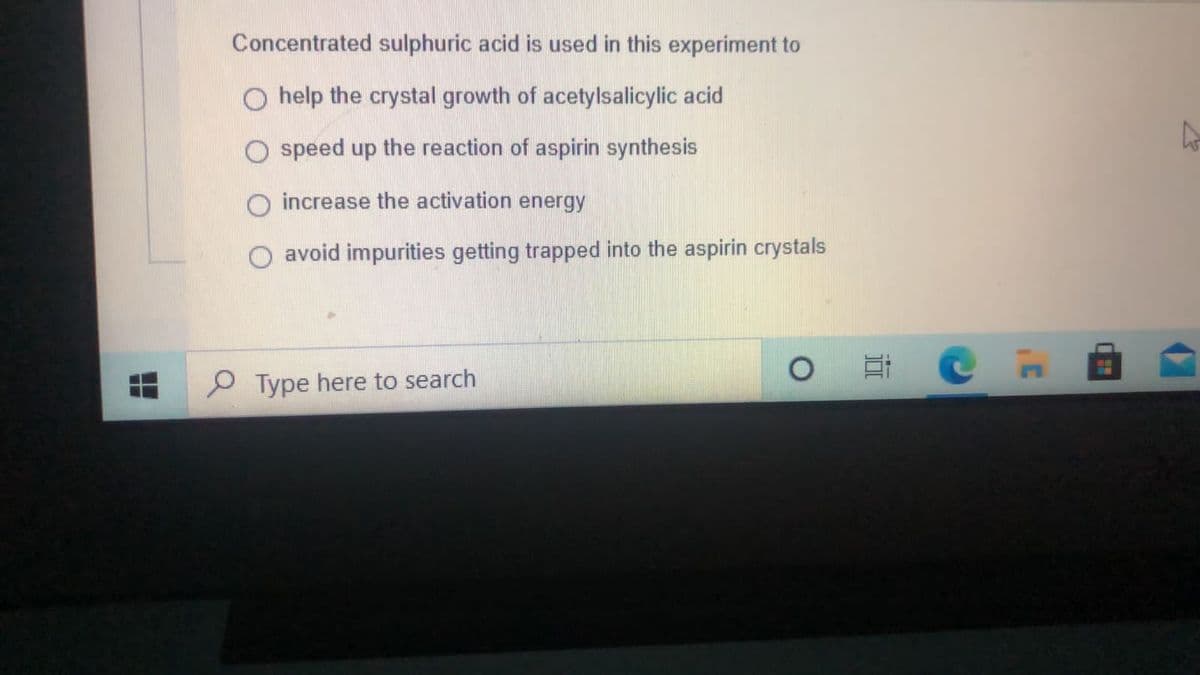
Transcribed Image Text:Concentrated sulphuric acid is used in this experiment to
O help the crystal growth of acetylsalicylic acid
speed up the reaction of aspirin synthesis
increase the activation energy
O avoid impurities getting trapped into the aspirin crystals
P Type here to search
![* Question Completion Status:
A Click Submit to complete this assessment.
Question 10
In a laboratory, the following should NOT be worn, from the safety point of view [more than one correct answer, choose only the right answer(s)]
O closed-toe shoes
unbuttoned lab coat
open sandals
V hanging jewelry
baseball cap, properly secured
loose clothing
Click Submit to complete this assessment.](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F662601a0-18b2-4601-928e-570472adf6eb%2Fa475c081-b417-4f31-9b75-eee54b78ec07%2F7uykscp_processed.jpeg&w=3840&q=75)
Transcribed Image Text:* Question Completion Status:
A Click Submit to complete this assessment.
Question 10
In a laboratory, the following should NOT be worn, from the safety point of view [more than one correct answer, choose only the right answer(s)]
O closed-toe shoes
unbuttoned lab coat
open sandals
V hanging jewelry
baseball cap, properly secured
loose clothing
Click Submit to complete this assessment.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

