Consider the following equilibrium: N, e) + 3H, (e) - 2NH, (e) aG° - = -34. kJ ow suppose a reaction vessel is filled with 5.56 atm of nitrogen (N,) and 0.868 atm of ammonia (NH) at 972. °C. Answer the following questions about thi ystem: nder these conditions, will the pressure of NH, tend to rise or fall? rise O fall Is R possible to reverse this tendency by adding H, In other words, if you said the pressure of NH, will tend to rise, can that O yes O no be changed to a tendency to fall by adding H? Similaty, if you said the pressure of NH, will tend to fall, can that be changed to a tendency to rise by adding H,? If you said the tendency can be reversed in the second question, calculate the minimum pressure of H, needed to reverse it. Eound your answer to 2 significant digits.
Consider the following equilibrium: N, e) + 3H, (e) - 2NH, (e) aG° - = -34. kJ ow suppose a reaction vessel is filled with 5.56 atm of nitrogen (N,) and 0.868 atm of ammonia (NH) at 972. °C. Answer the following questions about thi ystem: nder these conditions, will the pressure of NH, tend to rise or fall? rise O fall Is R possible to reverse this tendency by adding H, In other words, if you said the pressure of NH, will tend to rise, can that O yes O no be changed to a tendency to fall by adding H? Similaty, if you said the pressure of NH, will tend to fall, can that be changed to a tendency to rise by adding H,? If you said the tendency can be reversed in the second question, calculate the minimum pressure of H, needed to reverse it. Eound your answer to 2 significant digits.
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter17: Equilibrium
Section: Chapter Questions
Problem 6ALQ
Related questions
Question
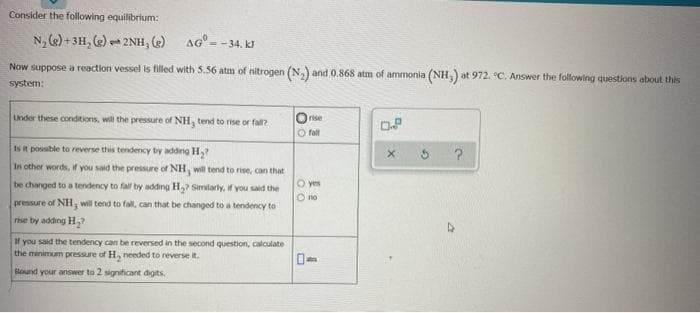
Transcribed Image Text:Consider the following equilibrium:
N, (e) + 3H, (e) - 2NH, e AG=
1.
= -34. kJ
Now suppose a reaction vessel is filled with 5.56 atm of nitrogen (N,) and 0.868 atm of ammonia (NH,) at 972. °C. Answer the following questions about this
system:
Lnder these conditions, will the pressure of NH, tend to rise or fall?
rise
DP
O fall
Is R possible to reverse this tendency by adding H,?
In other words, if you said the pressure of NH, will tend to rise, can that
be changed to a tendency to fall by adding H,? Simtarly, if you said the
O yes
O no
pressure of NH, will tend to fall, can that be changed to a tendency to
rise by adding H,
If you said the tendency can be reversed in the second question, calculate
the minimum pressure of H, needed to reverse it.
lound your answer to 2 significant digits.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning