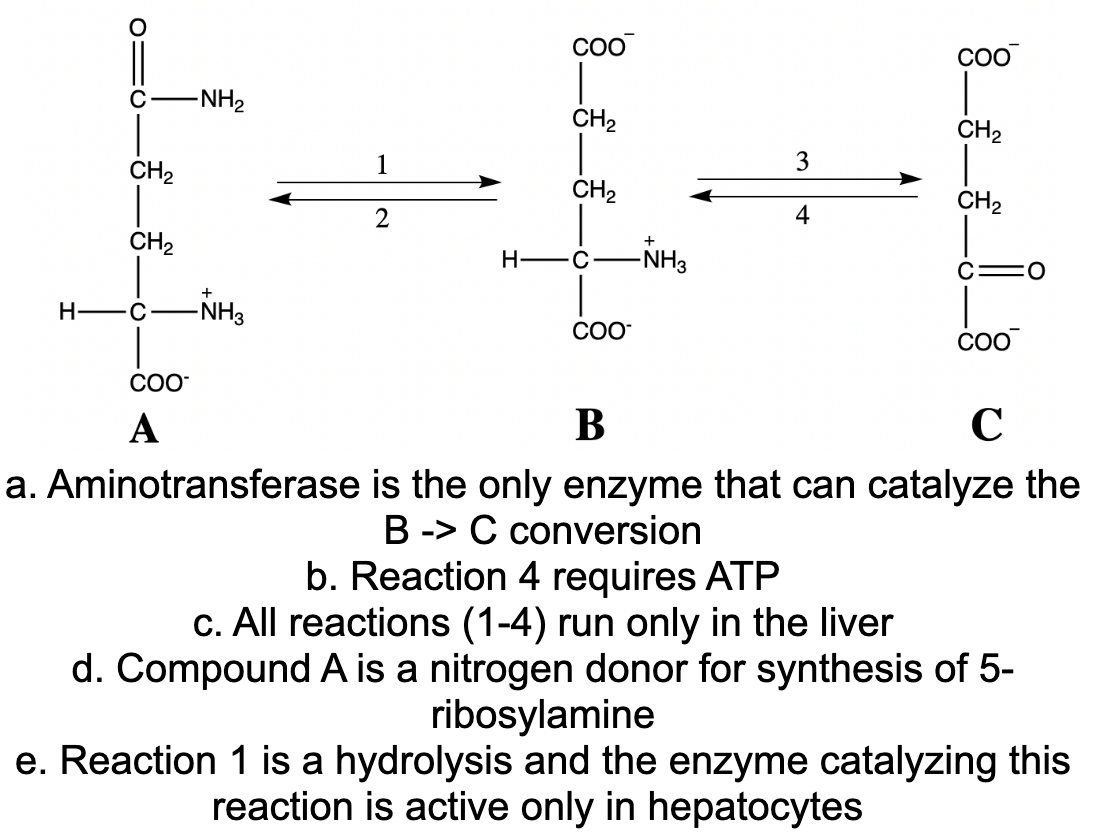
COO COO C -NH2 CH2 CH2 1 3 CH2 CH2 CH2 4 CH2 H- -NH3 -NH3 H- C COO CO- А C a. Aminotransferase is the only enzyme that can catalyze the B -> C conversion b. Reaction 4 requires ATP c. All reactions (1-4) run only in the liver d. Compound A is a nitrogen donor for synthesis of 5- ribosylamine e. Reaction 1 is a hydrolysis and the enzyme catalyzing this reaction is active only in hepatocytes
Q: what is a hydration number
A: Hydration refers to the interaction of a molecule or substance with water. Water interacts with seve...
Q: Shown below is the 5' end of an mRNA molecule. What are the first three (N-terminal) amino acids of ...
A: Through transcription the DNA molecule is converted into mRNA.
Q: In a spectrophotometer, what should be the appearance on the graph if the sample is pure? presence o...
A: Spectrophometry is a widely used technique in identification and confirmation of chemical substances...
Q: What advantage do alternative sigma factors have for bacterial gene expression?
A: Sigma factors are dissociable subunits of prokaryotic RNA polymerase that are required for its funct...
Q: a. Draw b furanose b. A polysaccharide which is a major constituent in the exoskeleton of arthropod...
A: NOTE: Subpart (a) must have β-furanose and not b furanose. Carbohydrates are biomolecules that are ...
Q: A membrane can separate gas mixture because different gases have different permeability through the ...
A: Gas separation by membrane is appealing in low-carbon technologies because it can be run in a contin...
Q: Antimicrobial Resistance (AMR) 2. Explain Antimicrobial stewardship (AMS
A: The question is all about the Microbes that antimicrobial resistant i.e AMR, so some bacteria, virus...
Q: escribe and give the results of an experiment that shows that polymerase III makes tRNA and 5S rRNA.
A: DNA and RNA are made up of long chains of nucleotides and ribonucleotides bases respectively. ...
Q: Define each of the following metabolic processes and write notes on importance and regulation of ea...
A: Urea cycle is known as Krebs-Henseleit urea cycle. As ornithine is the first member of the reaction...
Q: Connection between Electron Transport & Phosphorylation Task: 1. Define P/O ratio 2. Explain its...
A: Our body is always working, continuously doing various metabolic activities even when we are sleepin...
Q: what is the property of the solution that makes log(concentration) Vs electrochemical potential devi...
A: Answer: In an electrochemical cell, increasing the concentration of reactants will increase the volt...
Q: 1 TC: 200 250 300 350 Wavelength (nm) UV/Vis spectrum of four brands of polysorbate 80, a common det...
A: Coomassie dye (Blue G-250; also refer as CBB) binds to protein and gives a colorimetric change in th...
Q: What are examples of polyiodides? Include their structure
A: Polyiodides are a class of polyatomic halide anions that consists entirely of iodine atoms. It is pr...
Q: what receptor helps distinguish between organic and inorganic ? what does not remove neurotra...
A: Elements combine to form chemical substances called compounds. Further, all the compounds have been ...
Q: Why is ethanol a good solvent for the recrystallization of biphenyl, but acetone and water are not g...
A: Recrystallization is a process used to purify chemicals. In this, the compound to be purified is dis...
Q: What are the advantages and disadvantages of viable cell count and the turbidimetric methods?
A: In turbidimetric determination of cell growth ,after inoculation of the bacterial culture the optica...
Q: Complete the following paragraph describing the fate of the second 14CO-acetyl-CoA molecule incorpor...
A: The glyoxylate produced in the isocitrate reaction combines with 2nd molecule of acetyl coa in the m...
Q: What is the particular about the amino alcohol used in the synthesis of sphingomyelin?
A: Phospholipids: Phospholipids are compound lipids and it is of two types. These include glycerophosph...
Q: 22. A mutant algae has some of its mitochondria missing with inner mitochondrial membrane. Which of ...
A: The inner mitochondrial membrane is the mitochondrial membrane separating the mitochondrial matrix f...
Q: Multiple 3′ cleavage sites result in a. multiple genes of different lengths.b. multiple pre-mRNAs of...
A: Nucleic acids are a type of macromolecules comprised of carbon, hydrogen, oxygen, nitrogen, and phos...
Q: What is the concepts of the native conformation of proteins? Why and how do proteins refold and unfo...
A: Proteins are polypeptide structures consisting of one or more long-chain amino acid residues. They a...
Q: What are the possible effects and changes on the Michaelis-Menten equation that can result from comp...
A: COMPETITIVE INHIBITION:- structure of inhibitor closely resembles that of enzymes normal substrate...
Q: Explain the biological importance of of carbohydrates for a living things
A: Carbohydrates are the most abundant biomolecules in nature and form an important part of our diet to...
Q: 1. What is the name we give to structures which are not complete polypeptide chains but form superse...
A: "Since you have asked multiple questions, we will solve the first four questions for you. If you wan...
Q: for an in vitro translation reaction. Which one would you leave out? Cell sap containing ribosomes O...
A: In vitro translation is a technique that in which a given mRNA is translated (outide the cell using...
Q: AMP is derived from a reaction involving which of the following? ATP and ADP ADP and ADP NAD a...
A: Adenosine is a chemical found in human cells and they are of three different forms: adenosine monoph...
Q: Select the correct mechanism of each enzyme or drug enlisted below: * Inhibition via Enzyme Activity...
A: Hi! Thank you for the question. We are authorized to answer three subparts at a time, since you have...
Q: Below is the primary sequence of a viral protein. MSVVNTEIKFPTHLRSGDFAIIDGMVVEVTSVEYKPVEQAVYLKYRYHL...
A: The charge of a protein is determined by the charge of individual amino acids. The charge of individ...
Q: Using the data provided in the table, the estimated p50 for myoglobin is mmHg and the fraction satur...
A: Myoglobin (Mb) is an iron and oxygen binding protein that is present usually in the cardiac and ske...
Q: Define triacylglycerol
A: In living organisms, fats and oils are most commonly utilized as energy storage. They are fatty acid...
Q: Consider the following reaction at 25°C with the ΔG°’ = +1800 J/mol for the forward reaction. The mo...
A: Given Values: ∆G° = 1800 J/mol[A] = 16 mM[B] = 13 mMR = 8.315 J/mol-KT = 25+273 = 298 K
Q: What is Abetalipoproteinemia? explain in short 18:55
A: Abetalipoproteinemia is a uniquely rare disease. More than hundred cases of this disease has been re...
Q: DRAW 22:6A 4,7,10, 13,16,19 What Omega wa family Hhis fatty Acid Belong 22:6A4,7,10,13, 16, 19 what ...
A: The fatty acids are carboxylic acids with a hydrocarbon chain. They are the simplest form of lipids....
Q: 1. Explain the biochemical role of the phosphate that is removed in reaction B. 2. What would be a ...
A: "Since you have asked multiple questions, we will solve the first three questions for you. If you wa...
Q: It refer to the structures of the pathogen that are found associated with the infected plant (i,e., ...
A: The visible effects of diseases on plants are called symptoms and any detectable changes in color, s...
Q: do archaebacteria have autotroph or heterotroph nutrients
A: Nutrients are the components of food like carbohydrates, fats, vitamins, proteins, etc. These nutrie...
Q: Microbial products can be classified as three major categories. With the use of sketch, briefly expl...
A: Microorganism growth is a very complex and coordinated process that culminates in an increase in cel...
Q: Describe how the following properties affect the function of a protien: A.) R group orientation B.) ...
A: Amino acids are biomolecules that are comprised of two functional groups, these are an amino group (...
Q: 6. Celiac disease is a disorder of the small intestine characterized by autoimmune response to glute...
A: Gluten protein is commonly found in many cereals like Wheat, barley, rye etc and is difficult to dig...
Q: Define P/O ratio. State and explain the importance of P/O ratio.
A: P/O Ratio is also known as the phosphate/ Oxygen ratio.
Q: The following were obtained in a study of an enzyme known to follow Michaelis-Menten kinetics: R...
A: Enzymes are proteins which accelerate the rate of biochemical reactions. The Michaelis-Menten plot r...
Q: You've isolated a novel protein, but you believe what you actually have is a mixture of the unmodifi...
A: Amino acids are biomolecules that are comprised of two functional groups, these are an amino group (...
Q: Cytosol Mitochondrion A. PATHWAY 1. major pathway of ethanol metabolism NAD* NADH + H* NAD* NADH + H...
A: Ethanol - Small, uncharged molecule Oxidation- Loss of hydrogen; gain of oxygen Reduction- Gain of ...
Q: Write a short description on ALL of the following: a) The application of turbidostat against chemost...
A: In various assays and biological experiments, microoorganisms are allowed to grow in liquid cultures...
Q: In UV/Visible spectrophotometer analysis for a multicomponent system, there are only two dyes used i...
A: UV/Visible Spectrophotometry detects the absorbance of light in the UV/Visible range of the electrom...
Q: Which model for enzyme-substrate chemical complementarity is described by the following: Before sub...
A: Catalysts are substances that increase the rate of the chemical reaction by lowering its activation ...
Q: What are the three special proteins needed to form the initial replication bubble?
A: Replication is an essential process taking place in all living organisms that ensure the maintenance...
Q: describe how the pyruvate dehydrogenase complex is regulated?
A: Pyruvate dehydrogenase complex (PDC) is a three-enzyme complex that decarboxylates pyruvate into ace...
Q: True or False Galactose gives a positive result with Benedict’s test faster than lactose due to the ...
A: Carbohydrates, also known as sugars are the source of energy and one of the major macromolecules tha...
Q: Butanoic acid (C3H7COOH) is described as a weak acid. Define the term weak acid
A: Acids are chemical compounds that taste sour and turn the blue litmus red. They have a pH of less th...
Step by step
Solved in 2 steps with 1 images

- Energetic of Fructose-1 ,6-bis P Hydrolysis (Integrates with Chapter 3.) The standard free energy change (G) for hydrolysis of fructose-1. 6-bisphosphate (FBP) to fructose-S-phosphate (F-6-P) and P: is -16.7 KJ/mol: FBP + H2O fructose-6-P + Pi The standard free energy change (G) for ATP hydrolysis is -30.5 KJ/mol: ATP + H2O ADP + Pj What is the standard free energy change for the phosphofructokinase reaction: ATP + fructose-6-P ADP + FBP b. What is the equilibrium constant for this reaction? c. Assuming the intracellular concentrations of [ATP] and (ADP] are maintained constant at 4 mM and 1.6 mM, respectively, in a rat liver cell, what will be the ratio of [FBP]/[fructose-6-P] when the phosphofructokinase reaction reaches equilibrium?Chart is Given for you: Below is a chart of values for actual enzymes. Enzyme Km (M) kcat (1/s)Chymotrypsin 1.5 × 10^−2 0.14Pepsin 3.0 × 10^−4 0.5Tyrosyl-tRNA synthetase 9.0 × 10^−4 7.6Ribonuclease 7.9 × 10^−3 7.9 × 10^2Carbonic anhydrase 2.6 × 10^−2 4.0 × 10^5Fumarase 5.0 × 10^−6 8.0 × 10^2 Assume the enzyme concentration is equal across all samples (and is equal to 1). (Answer a and b only)a. Which enzyme will have the highest V0 at very high substrate concentrations? (1 M). Why? b. Which will have the highest V0 at very low substrate concentrations (5.0 × 10^−12). Why?Can u help me to explain to me, please? I stuggled. I don't understand The a-ketoglutarate that is produced inthe reaction is now free to accept a new quaternary ammonium group from another amino acid in a transamination reaction.
- Legend: Blue – wild-type β-galactosidase Red – mutant β-galactosidase _________ a. What is the optimum pH of wild type β-galactosidase? _________ b. What is the optimum temperature of mutant β-galactosidase? _________ c. Which enzyme has the greater activity at pH 7.2? _________ d. Which enzyme has the greater activity at a temperature of 42.5oC? _________ e. Which enzyme has greater activity if pH decreases from 7.5 to 6.4? _________ f. Which enzyme has greater activity if temperature increases from 40oC to 41 oC?Pepsin, a peptidase that hydrolyzes proteins, functions in the stomach at an optimum pH of 1.5–2.0. How is the rate of a pepsin-catalyzed reaction affected by each of thefollowing conditions? increasing the concentration ofproteins changing the pH to0 running the reaction at 0°C using lesspepsinif the reaction pH drops significantly (pH=2 for example), how would reaction catalyzed be serine proteases be affected? why? (assumin that the rest of the protein structure remain intact)
- The highest energy point of the serine protease reaction is the formation of the tetrahedral oxyanion intermediate on the original carbonyl carbon of the scissile peptide bond. True or False? DIPF is an irreversible inhibitor of serine proteases. True or False? The acyl-enzyme intermediate is the transition state of the reaction. True or False? Serine proteases are classified as isomerases. True or False?A(n) _________ reaction converts glycylalanine to glycine and alanine. This reaction requires __________. Blank 1 options- Oxidation-reduction Hydrolysis Isomerization Group transfer Internal rearrangement Blank 2 options- H2O and apeptidase or a protease H2O and a phosphatase ADP and a phosphatase NAD+ and a peptidase or a proteasePut the steps of the chymotrypsin's enzymatic reaction in order of events from #1-10. -Tetrahedral intermediate collapses and product 2 released. -Aromatic R group of polypeptide substrate is positioned in hydrophobic pocket and the carbonyl of that residue is positioned in the oxyanion hole -His57 deprotonates Ser195 -Formation of acyl-enzyme intermediate -Triad in original form with Asp102 hydrogen bonding with His57 and His57 hydrogen bonding with Ser 195. -Tetrahedra intermediate collapses and product 1 released -H2O enters active site of chumotrunsir -Ser195 does nucleophilic attack on carbonyl of substrate positioned in oxyanion hole -His57 deprotonates H20 -H20 does nucleophilic attack on carbonyl of substrate in oxyanion hole
- complete the following mechanism showing the cleavge of the peptide bond for the following cysteine protease reaction. you only need a catalytic dyad for cystein proteases because the pKa of cysteine is low enough (8) and close enough to the pH of 7.4 that is can easily be deprotonated by histidine.Based on this information, what type of enzyme would ornithine decarboxylase be? I searched it up on Google and sources say it is a lyase because it catalyzes cleavage without H2O, but in equation 1, isn't water part of the reaction? Is decarboxylase still a lyase because water is a reactant, not a catalyst?for the following reactions: arrive at the Michaelis menten enzyme kinetics: E+2A <-----k1,k2----->A.E+A reversible A.E+A<------k3,k4------>A 2E reversible A2.E------k5--------> E+CD non-reverisble show all steps

