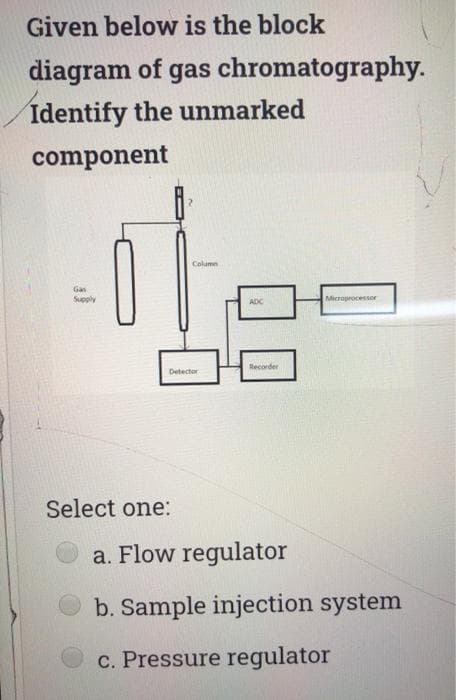
Given below is the block diagram of gas chromatography. Identify the unmarked component Column Gas Supply Microprocessor Recorder Detector Select one: a. Flow regulator b. Sample injection system c. Pressure regulator
Q: In a normal phase chromatography setup, which would be the predicted characteristics of the eluates…
A:
Q: In thin layer chromatography the initial spot is placed: A. on a pencil line B. four centimeters…
A: Thin layer chromatography is a type of chromatography.
Q: What part fo chromatography is being descibe by the following? a phase that is fixed in place either…
A:
Q: Fresh solvent - eluent Mobile phase Sample components A Celumn packing Stationary phase suspended in…
A: B is less polar and A is more polar Because the stationary phase is polar, polar molecules will…
Q: Which of the following matches is/are incorrect regarding the paper chromatography experiment? a.…
A:
Q: a. What does ECD mean. b. Discuss how column temperature influences separation in a chromatography…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: in what situation does one use thin and/or column chromatography? You can any of theme for anything…
A: Chromatography is an analytical technique to separate mixtures into it's components. It is also…
Q: David is in dilemma to choose a suitable chromatographic detector for gas chromatography analysis.…
A: The detector which detects the pesticide content in vegetables has to be given and the two…
Q: What safety precaution are needed when working with a.) centrifuge b.) Inductively Coupled Plasma…
A: a) safety precaution are needed when working with Centrifuge: Safety cups have to be used. The…
Q: a) In any gas chromatographic analysis, exceeding a column's maximum temperature limit will usually…
A: Gas chromatography
Q: spotting paper chromatography samples above the solvent level ______________________________________…
A:
Q: Why is splitless injection used with purge and trap sample preparation?
A: The collection of all of the analyte from the unknown and to inject whole of the analyte into the…
Q: be micro a
A: Electron probe microanalyzer is an analytical tool used to determine the chemical composition of…
Q: Select which of the following ways gas chromatography can be performed? A. Only in columns B. Only…
A:
Q: How does temperature, flow rate of carrier gas, boiling point and column suupport affect gas…
A: Explanation. Retention time- Retention time is the amount of time a compound spends on…
Q: Column Chromatography Chromatographic Separation Fresh solvent = eluent Mobile phase Sample…
A: A is more polar Because the stationary phase is polar, polar molecules will spend more time adsorbed…
Q: Which type of gas chromatographic column consists of a tube that is densely packed with uniform,…
A: An analytical technique which is used to separate components present in a mixture and to determine…
Q: calculate the following for each sample gas chromatograph. a) retention time for each peak (label…
A: Gas chromatography is a technique for separating and analyzing compounds that can be vaporized…
Q: Can you TYPE the definitions of the all of words please Chromatography TLC Stationary phase Mobile…
A: Most of the time, the compounds we encounter are not in their purest form. They are essentially a…
Q: What is a purification method that is more "green" than flash chromatography? (produces less waste).…
A:
Q: High performance liquid chromatography can separate eluates even if they are not present in the UV…
A: The actual separation of each component in a sample is always carried inside a column and to be able…
Q: The flame-ionization detector is used in gas chromatography because the eluates cannot be detected…
A: Detector is the instrument that can detect the eluate species. In gas chromatography analyte…
Q: Which of the following matches is/are incorrec regarding the paper chromatography experiment? A.…
A: In paper chromatography The mobile phase is generally the solvent also called as chromatography…
Q: . Column resolution is an indicator to identify the selectivities and efficiencies of the column…
A: Gas chromatography can be performed only in columns.
Q: In a normal phase chromatography setup, which would be the predicted characteristics of the eluates…
A: Ans : Eluates will be in decreasing polarity ( Benzoic acid is more polar than benzaldehyde.…
Q: Is the separation of the mixture shown here a physical orchemical process?
A: To explain whether the method of separation of mixture via column chromatography is physical or…
Q: 1. Provide two advantages and two disadvantage of flow cytometry analysis. Briefly explain each.
A:
Q: i) Put the steps of a column chromatography analysis in a correct order. a. Elute the column with…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: integrator A. measures absorbance data detector B. processes data into the display unit injection…
A:
Q: the efficiency of a chromatographic column improves the measure that a.increases the height of the…
A: We know that, In chromatography, column efficiency is measured in terms of number of theoretical…
Q: A chromatography column with a length of 10.3 cm and inner diameter of 4.61 mm is packed with a…
A:
Q: the efficiency of a chromatographic column improves the measure that a.increases the height of the…
A: To determine what improves the efficiency of a chromatographic column:
Q: Cuvette maintenance and cleaning is crucial for the success of spectroscopic analysis. Which is NOT…
A: Spectroscopic analysis is a technique that can be used to study the interaction between…
Q: Thin‑layer chromatography (TLC) can be used as a preliminary identification method for many…
A: Given that., Distance traveled by the spot from bottom = 3.52 cm Distance travel by mobile phase =…
Q: At the completion of the experiment, a student measures the solvent front to be 55 mm from the…
A:
Q: In thin layer chromatography, the stationary phase is made of and the mobile phase is made of solid,…
A: Thin layer chromatography: TLC is a chromatography technique used to seperate non-volatile mixture.…
Q: The fluid exiting a chromatographic column is called the eluent eluate analyte solvent Question 22…
A: Chromatography is the laboratory method to separate the components of a mixture. Elution is the…
Q: You are assigned to separate a mixture of colourless amino acids present in protein molecule, using…
A: Chromatography: It is the technique used to separate the mixture of colored compound. Separation is…
Q: By using The Graph below that showing detector response as a function of elution time, Complete the…
A: Gas chromatography is an analytical technique used for the separation of chemical components of a…
Q: Name two general methods for improving the resolution of two substances chromatographic column.
A: Good resolution columns provide a great separation of the components by a column chromatography.…
Q: Put all steps (below) of a TLC analysis in a correct order. Write the two main visualization methods…
A: TLC analysis in correct order is as follows.
Q: Choice of a paper and solvent are main back bones of paper chromatography .comment.
A: Chromatography is a analytical technique, which is used for separation of mixture of gases or…
Q: High performance liquid chromatography (HPLC) can be used to : a. identify the various pigments from…
A: High performance liquid chromatography (HPLC) is defined as a column chromatography where at high…
Q: DO R O a blackboard.uob.edu.bh View Assessment Given below is the diagram of Process of…
A: The unmarked component is detector. It is placed at the bottom of the column. It responds to solute…
Q: Paper Chromatography laboratory report explanation
A: Paper chromatography is an analytical method used to separate colored chemicals or substances. It is…
Q: Write exhaustively on paper chromatography
A: Chromatography is a special technique which is used to seprate the components of a mixture.
Q: Comments on choice of a paper and solvent are main back bones of paper chromatography.
A: Paper Chromatography is mailny based on the choice of paper and solvent used. In this technique,…
Q: In paper chromatography, what could happen if the lid was not immediately put on the beaker? a. It…
A: The paper chromatography performed with volatile solvent or may be mixture of volatile compounds.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- In order to execute paper chromatography, which of the following stages is required?I. Load the standard/s beside the sample spotsII. Allow the chromatogram to touch sides of beakerIII. Saturate the chamber with vapor of the mobile phaseIV. Place the spots at the same height as the solvent levelplease quickly ,within 1hour thanks ! If it takes longer than an hour, i don't need the answer (2) List the representative stationary phase for gas chromatography.What are the general characteristics of Gas Chromatography, Thin Layer Chromatography, Ion Exchange, HPLC, SEC and Affinity Chromatography? What elutes first? Additionally can you explain what exactly is stationary phase and mobile phase?
- separate a mixture containing acetone, acetamide and 1,2-dichloroethane using a high- performance liquid chromatography (HPLC) technique. The instrument was equipped with a 40-cm column packed with cyano propyl bonded to the siloxane backbone material. i) List THREE (3) examples of mobile phase solvent/solvent systems that can be used with the column above. ii) Determine the sequence in which the three analytes eluted from the column and explain the underlying principle applied. iii) Sketch the chromatogram that would be produced if the column was replaced with a column filled with C18 hydrocarbon bound to the siloxane backbone material and acetonitrile was employed as the mobile phase. Label the peaks observed.The following data give the recovery of bromide from spiked samples of vegetable matter, measuredusing a gas–liquid chromatographic method. The same amount of bromide was added to each specimen(Roughan, J.A., Roughan, P.A. and Wilkins, J.P.G., 1983, Analyst, 108: 742).Tomato: 777 790 759 790 770 758 764 ug/gCucumber: 782 773 778 765 789 797 782 ug/g(a) Test whether the recoveries from the two vegetables have variances which differ significantly.(b) Test whether the mean recovery rates differ significantly.Draw a schematic diagram of how a Flash Column Chromatography works? Please answer at your own words
- Brief discussion of column chromatographic separation (solvent polarity, elution ofcompounds etc.)I) Sketch the apparatus set up for the chromatographic technique used byIman. Use the correct chromatographic terminologies to label all of theapparatus, materials, and phases. II)Predict the sequence of elution when the mixture was loaded onto theseparation column. Justify your answer. III) After the first compound was eluted, Iman discovered that the secondcompound was taking too long to pass through the column. Please suggestany strategies that could be used to aid the elution process. Justify youranswer.Complex chromatographic patterns can be simplified bypassing the components emerging from the gas chromatographic column through a(n) ___________.
- As a newly appointed research officer, Marissa has been assigned a task to separate a mixture containing acetone, acetamide and 1,2-dichloroethane using a high-performance liquid chromatography (HPLC) technique. The instrument was equipped with a 40-cm column packed with cyano propyl bonded to the siloxane backbone material. i) List THREE (3) examples of mobile phase solvent/solvent systems that can be used with the column above. ii) Determine the sequence in which the three analytes eluted from the column and explain the underlying principle applied. iii) Sketch the chromatogram that would be produced if the column was replaced with a column filled with C18 hydrocarbon bound to the siloxane backbone material and acetonitrile was employed as the mobile phase. Label the peaks observed.the efficiency of a chromatographic column improves the measure thata.increases the height of the theoretical dishesb.decreases the number of theoretical platesc.platform height is reducedd.None of the above Please explain it Provide clear handwriting and clear imageArrange in INCREASING Rf if the mixture underwent paper chromatography with the same solvent system. Explain how you get the answer I. 2-chloropentaneII. pentanoic acidIII. cyclopentaneIV. pentanal a. II = IV < III < I b.II < IV < I < III c.I < II < III < IV d.III < I < IV < II



