GIVEN: (Probable atomic weights: N=14; O=16) 1. What is the most applicable state of matter for the given compounds in the table? a. solid b. liquid crystal c. gas d. liquid 2. Which has the lowest molecular weight among the three compounds? Your answer here will be the basis for answering the next items below. Nitrogen dioxide Nitrous oxide Nitric oxide
GIVEN: (Probable atomic weights: N=14; O=16) 1. What is the most applicable state of matter for the given compounds in the table? a. solid b. liquid crystal c. gas d. liquid 2. Which has the lowest molecular weight among the three compounds? Your answer here will be the basis for answering the next items below. Nitrogen dioxide Nitrous oxide Nitric oxide
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter10: Liquids And Solids
Section: Chapter Questions
Problem 38E: Carbon tetrachloride, CCl4, was once used as a dry cleaning solvent, but is no longer used because...
Related questions
Question
GIVEN: (Probable atomic weights: N=14; O=16)
1. What is the most applicable state of matter for the given compounds in the table?
a.
b. liquid crystal
c. gas
d. liquid
2. Which has the lowest molecular weight among the three compounds? Your answer here will be the basis for answering the next items below.
Nitrogen dioxide
Nitrous oxide
Nitric oxide
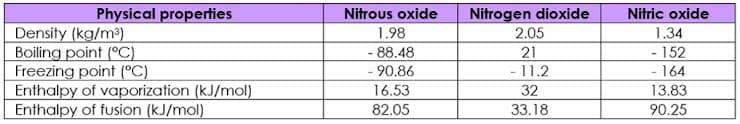
Transcribed Image Text:Nitrous oxide
Nitrogen dioxide
2.05
Physical properties
Nitric oxide
Density (kg/m³)
Boiling point (°C)
Freezing point (°C)
Enthalpy of vaporization (kJ/mol)
Enthalpy of fusion (kJ/mol)
1.98
1.34
- 88.48
21
- 11.2
- 152
- 164
- 90.86
16.53
32
13.83
82.05
33.18
90.25
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning