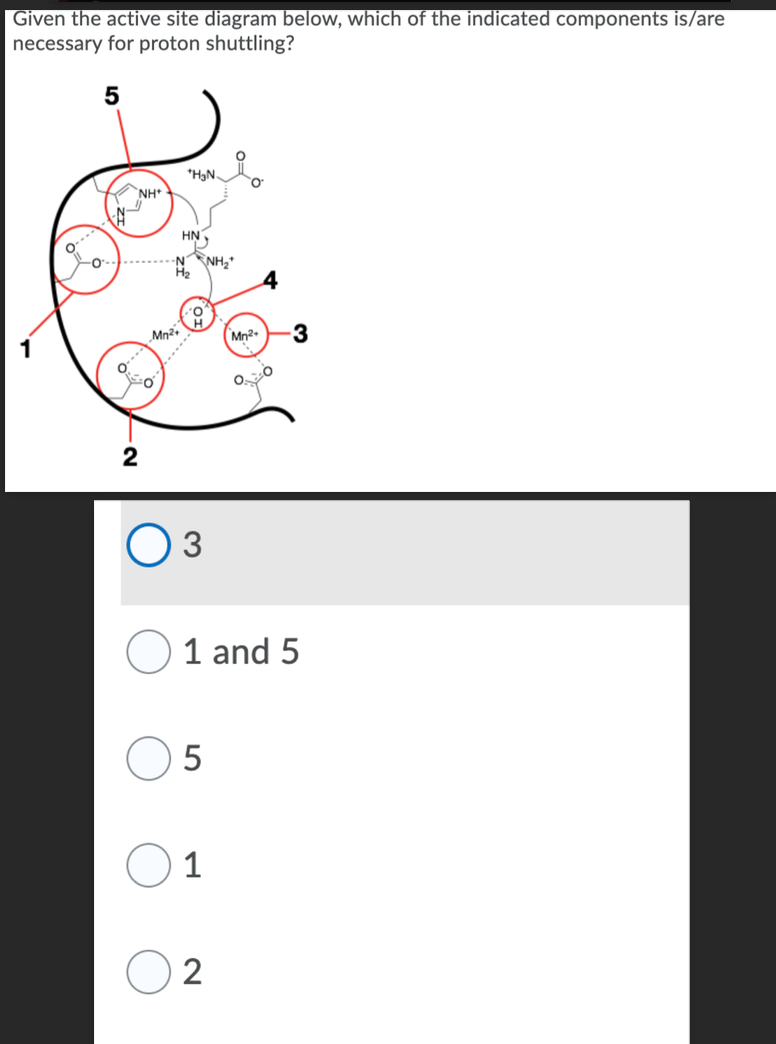
Given the active site diagram below, which of the indicated components is/are necessary for proton shuttling? O.... NH+ -~ 2 +H₂N HN NNH Mn²+ O 03 5 1 and 5 1 Mn²+ 2 3
Q: Which of the following mRNA sequences codes for the polypeptide sequence tyrosine-leucine-alanine?…
A: Transcription is the process that synthesizes mRNA from DNA. The mRNA strand synthesized through…
Q: Mach the terms left with as many terms GMP Nucleotide at right by entering @ Nucleoside 3 Z-DNA…
A: Thank you for your question, Here is the answers for the above match the following with…
Q: CHO CH₂OH CHO НО -H но- -H H-OH -OH H-OH НО -H H- HO-H H-OH CH.OH CH₂OH CH2OH PAS PES 16. an epimer…
A: The name for the following compounds are : PAS - D-arabinose PES - L-xylulose PIS - D-xylose POS -…
Q: Statement Analysis: Statement 1: Mitochondrion is composed of membranes, the inner membrane and the…
A: Mitochondria is an important cell organelle which is called as power house of the cell as it is main…
Q: 2,3-bisphosphoglycerate (BPG) is a negative allosteric regulator of hemoglobin that is produced red…
A: 2,3-Bisphosphoglycerate (BPG) is a compound that promotes hemoglobin transition from a…
Q: What defect in the cell causes the condition known as Zellweger's syndrome, ZS? Explain in detail…
A: Zellweger syndrome is the most severe form of the Zellweger spectrum of conditions. It is a rare…
Q: In the Watson-Crick structure of DNA, the: a. adenine content of one strand must equal the thymine…
A: Watson and Crick model of DNA has two strands that wound around each other and form double hellicle…
Q: Explain the chirality of amino acid molecules.
A: If any combination of rotations, translations, and conformational changes cannot superimpose a…
Q: In the pathogenesis of atherosclerosis, small oxidized LDLs…. a) are internalized in artery…
A: Atherosclerosis is a disease characterized by thickening or hardening of arteries due to…
Q: Q/What metabolic defect causes galacto semia? and what is lactose intolerance ?
A: When the enzyme galactose-1-phosphate uridyl-transferase (GALT) is deficient or not active, classic…
Q: The Hershey-Chase experiment, in which bacteria were infected with radioactively labeled bacterial…
A: The Hershey-Chase experiment, that also involved infecting bacteria with radioactively labelled…
Q: oncerning the process of the aerobic fate of glucose oxidation, which of the following statements is…
A: In the cytosol, 1 molecule of glucose is converted to 2 molecules of pyruvate via glycolysis .…
Q: Q2) Yogurt is produced from milk by the action of diary bacteria. These bacteria produce lactic acid…
A: Yogurt is produced by a process called lactic acid fermentation. Lactobacillus Sp. and streptococcus…
Q: What is the relation between GMO crops and the four of the principles of bioethics?
A: The word GMO stands for Genetically Modified Organism. GM crops or Genetically Modified crops have…
Q: write true if the statement if correct and change the bold word/phrase to make it correct lower…
A: Glycogen is the storage form of carbohydrates in animals. It is also called animal starch for this…
Q: During translation elongation cycle, which of the following step(s) is/are repeated for each amino…
A: Translation is a process by which proteins are synthesized from mRNA template. It involves all the…
Q: The most common cause of death from tobacco smoking is: O emphysema O strokes heart disease O lung…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: sing equilibrium argument, why does Km apparently increase, decrease or stay the same in…
A: Inhibition in biochemistry occurs in different enzymes. Inhibition of enzymes means blocking or…
Q: Item: Statement: a) Active site b) Induced fit c) Enzymes d) Enzyme-substrate complex 1. Decreases…
A: Metabolic activity is constant in living things. All live cells are constantly undergoing thousands…
Q: Which of the following statements is true? a) High insulin/glucagon ratio activates lipolysis in…
A: Lipolysis is the process in which the lipid, triacylglycerol is broken down into its components…
Q: Which vitamin is needed for FADH formation? Group of answer choices Niacin Riboflavin Vitamin C…
A: A number of significant metabolic reactions require the redox cofactor FAD. It exists in the redox…
Q: Which of the following statements regarding enzyme catalysis is false? All options are false. Once…
A: Normally an energy barrier exists between substrates (S) and products (P) i.e., S -> P…
Q: Q/What are the Irreversible inhibitor ?
A: Inhibitor is an agent that slows or interferes with the chemical reaction. Enzyme inhibitor is a…
Q: What will happen if the enzyme renin is mixed with 1N HCL before adding milk?
A: Rennin is a proteolytic enzyme or protein digesting enzyme also know as chymosin.
Q: A prosthetic group of a protein is a non-protein structure that is: a. a ligand of the protein. b.…
A: Proteins are composed of amino acids. They are linked together by peptide linkages. Proteins have…
Q: Explain how amino acids form proteins. In your explanation you will consider different types of…
A: Proteins are composed of amino acids. They are linked together by peptide linkages. Proteins have…
Q: What is the biological advantage to the sigmoidal binding curve of hemoglobin for oxygen? A. It…
A: The binding of oxygen to the haemoglobin increases with increase in oxygen partial pressure, Maximum…
Q: The task in this assessment is to describe how a specific gene is replicated and used to make its…
A: In most human cells there is a structure called nucleus. The nucleus contains the genome. in humans…
Q: Q1. Diffusion is: Directional Non-random Passive None of the above
A: The body's biochemical and physiological processes involve a variety of molecules and solutes.…
Q: Multiple Choice Each of the numbered items or incomplete statements is followed by answers or by…
A: Indicators are dye molecules that are used to indicate the pH of a solution. Normality, molarity,…
Q: Which of the following statements is true about brain metabolism in starvation? a) The brain can use…
A: A continuous supply of glucose and oxygen to neurons and astrocytes is required for brain…
Q: Based in your learning in this course, from what process was the siRNA cell protection mechanism…
A: As per the literature so far, there are many theories suggesting the evolution of siRNA cell…
Q: Many early attempts at enzyme engineering tried to design so-called catalytic antibodies. This…
A: Enzymes function by lowering the activation energy of the transition state of a chemical reaction.…
Q: When people are talking about ways to lose weight, cutting out carbohydrates is always on the…
A: General myths are: 1. Dieting (Less eating or skipping meals) to reduce weight 2. Skipping just the…
Q: give the significance/role/effect of the reagent/condition in the isolation or analysis of a…
A: DNA isolation is a process of isolation of DNA from biological sample like body fluid, tissue, etc.…
Q: ou have a polypeptide chain that is linked by disulfide bond. If beta-mercaptoethanol is applied,…
A: A polypeptide chain that is linked by disulfide bond. If beta-mercaptoethanol is applied, there is…
Q: Glucagon increases hepatic gluconeogenesis by decreasing the cytosolic concentration of which of the…
A: The hormone glucagon sends signals to the liver to produce more glucose and its release when the…
Q: Which of the following is true about the structure of DNA as proposed by Watson and Crick (B-form…
A: Introduction: Nucleic acids are large complex biomolecules that store hereditary information…
Q: What are proteins and what do they do?
A: One of the many intricate molecules your body uses for various functions is the protein. Bones,…
Q: Show, by hand drawing a diagram, how a disaccharide and polysaccharide is formed
A: Carbohydrates are the most abundant biomolecules on earth. These are the organic compounds having…
Q: Which cytochrome P450-catalyzed reaction is shown below? R-CH,NH2 [R-CH=NH] Oxidation of aromatic…
A: Cytochrome P450 is an enzyme that catalyzes various reactions such as oxidation, hydroxylation,…
Q: What is DRESS .Give full form of this syndrome.
A: It is a type of drug induced hypersensitivity reaction, caused by certain medication.
Q: Do you think obesity is a choice? What are the influences of lipids/fats mechanism in the body to…
A: Lipids are vital micronutrient that helps our body to absorbs nutrients , made hormones, even cell…
Q: The H-precursor of ABO blood group system consists of proteins on to which sugar moles not present…
A: The type of oligosaccharides present on the red blood cells surface determine the blood type of the…
Q: What covalent bonds are linking nucleic acid monomers? The bonds between oxygen and carbon…
A: Nucleic acids are made up of pentose sugar, nitrogenous base, and phosphate. Nucleic acids are…
Q: P9-11 The following data on baker's yeast in a particular medium at 23.4°C were obtained in the…
A: "Since you have posted a question with multiple sub-parts, we will solve the first two subparts for…
Q: We are utilizing BL21 DE3 bacterial cells for the expression of the ADA protein via autoinduction.…
A: BL21 DE3 Bacterial cells: It is an E. coli B strain that lacks the Lon protease. Recombinant…
Q: The nucleotide is adenine adenosine adenosine monophosphate cytidine prion
A: A nucleotide is the basic building block of nucleic acids like RNA and DNA. The nucleic acids act as…
Q: An intermediate of the citric acid cycle that undergoes reductive amination with glutamine as…
A: Citric acid cycle is the second stage of cellular respiration that occurs in mitochondrial matrix…
Q: Information for Part 2 The following table shows the concentration of ATP, ADP and phosphate into…
A: In the above question the change in free energy can be calculated by Δ?′ = Δ?0 + ?? ln ([???][??]/…
Step by step
Solved in 2 steps with 2 images

- Components of ETC involved in redox reactions are organised from high energy to low energy”. Explain this statement and how does this play a role in transfer of electrons in ETC?Define the following terms: a. electron transport chain b. oxidation-reduction reactions c. conjugate redox pair d. reduction potential e. standard reduction potentialProvide the reaction equations that illustrate the synthesis of ROS from electrons leaking from electron transport complexes
- When free water molecules are excluded from an enzyme’scatalytic pocket, is a catalytic —OH group a stronger orweaker nucleophile? Explain.If the hydrolysis of 1 M glucose 6-phosphate catalyzed by glucose 6-phosphatase has a ΔG′∘ of −11.386 kJ/mol at 25 °C, what percentage of substrate remains once the reaction reaches equilibrium assuming no product was initially present? (Round answer to the nearest whole number)Assume you have an enzyme that catalyzes a reaction that breaks down dopachrome. At t = 0 s, the absorbance at 475 nm is 0.2 when you add the enzyme. At t = 30 s, would you expect the absorbance to be less than or greater than 0.2?
- At what substrate concentration would an enzyme with a kcat of 25.0 s-1 and a KM of 3.5 mM operate at 25% of its maximal rate? How many reactions would the enzyme catalyze in 45 seconds when it is fully saturated with substate, assuming the enzyme has one active site?Shown below are reduction potentials for four half-reactions. Which of the coupled reactions is favorable? (Note that for Cytochrome c you must multiply the reduction potential by 2 for each coupled reaction because only one electron is involved) a) 2 Cytochrome c (Fe3+) + H2O ó 2 Cytochrome c (Fe2+) + O2 b) NADH + Succinate- ó NAD+ + Fumarate- c) Fumarate- + H2O ó Succinate- + O2 d) All of the abovea newly identified bacterium called Nomore biochem is unable to synthesize ubiquinone. A mobile electron carrier called CXC3 is used as a substitute. From the information provided in the table, calculate delta G' and Keq value at 298K for the redox reaction that occurs in the Nomore Biochem electron transport chain. (constants: R=8.3 J/degree x mol, F= 96.1 kJ/v x mol Half Reactions E' (V) ubiquinone + 2e- + 2H+--> Ubiquinol + H2 0.045 NAD+ + 2e- + 2H+ --> NADH + H+ -0.320 CXC3 + 2e- + 2H+ --> CXC3H2 -0.450 explain the impact that using CXC3 instead of ubiquinone will have on ATP production in the cell. How might the cell adapt to this situation?
- Explain why the coupled reaction ATP → ADP + Pi in the P-class ion pump mechanism does not involve direct hydrolysis of the phosphoanhydride bond.The oxyanion hole is comprised of hydrogen bonding atoms that project into a space on the active site that will have all H-bonding potential satisfied only during the catalytic cycle of the reaction. True or False? The catalytic triad residues are all next to each other in the primary sequence of trypsin. True or False?In each of the following, tell whether the substance gainselectrons or loses electrons in a redox reaction: (a) An oxidizing agent(b) A reducing agent(c) A substance undergoing oxidation(d) A substance undergoing reduction

