Chapter19: Aldehydes And Ketones: Nucleophilic Addition Reactions
Section19.SE: Something Extra
Problem 50MP
Related questions
Question
1. What kind of reaction is the reaction between N, N-diethylaniline, and diazonium salt? Supply a mechanism for this coupling reaction.
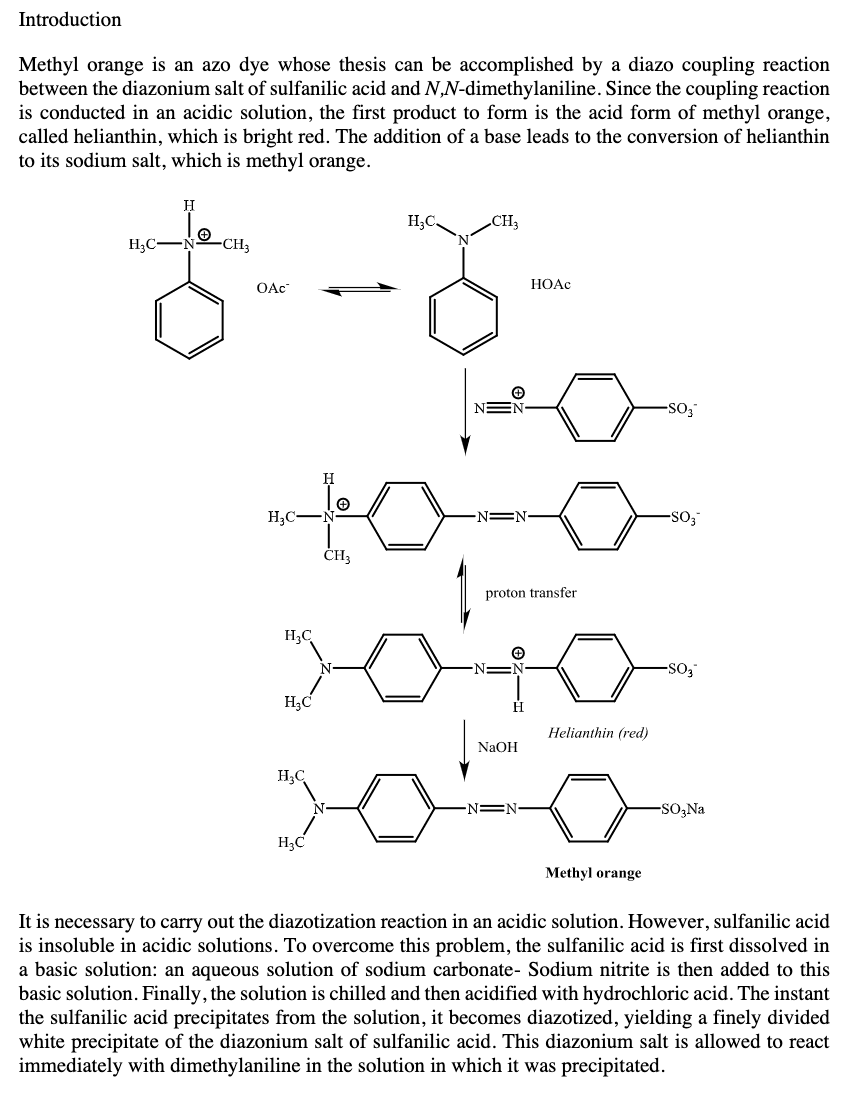
Transcribed Image Text:Introduction
Methyl orange is an azo dye whose thesis can be accomplished by a diazo coupling reaction
between the diazonium salt of sulfanilic acid and N,N-dimethylaniline. Since the coupling reaction
is conducted in an acidic solution, the first product to form is the acid form of methyl orange,
called helianthin, which is bright red. The addition of a base leads to the conversion of helianthin
to its sodium salt, which is methyl orange.
H
H;C.
CH3
OCH3
H3C
OAc
НОАс
H,C
ČH3
proton transfer
H3C
H3C
Helianthin (red)
NaOH
H3C
N=N
-SO,Na
H3C
Methyl oran
It is necessary to carry out the diazotization reaction in an acidic solution. However, sulfanilic acid
is insoluble in acidic solutions. To overcome this problem, the sulfanilic acid is first dissolved in
a basic solution: an aqueous solution of sodium carbonate- Sodium nitrite is then added to this
basic solution. Finally, the solution is chilled and then acidified with hydrochloric acid. The instant
the sulfanilic acid precipitates from the solution, it becomes diazotized, yielding a finely divided
white precipitate of the diazonium salt of sulfanilic acid. This diazonium salt is allowed to react
immediately with dimethylaniline in the solution in which it was precipitated.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

