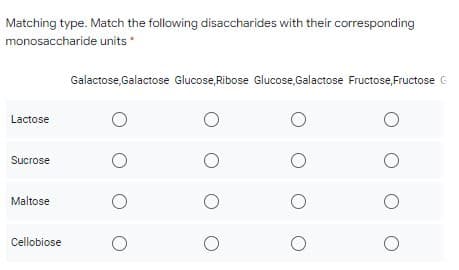
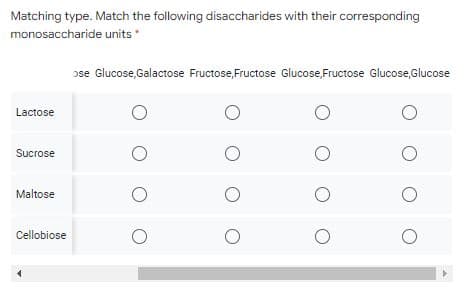
Matching type. Match the following disaccharides with their corresponding monosaccharide units Galactose,Galactose Glucose,Ribose Glucose,Galactose Fructose,Fructose G Lactose Sucrose Maltose Cellobiose
Q: What class of drug does atropine belong? What symptoms do you expect after intoxication with atropin...
A: Atropine is a prescription drug that is used to treat symptoms of bradycardia (low heart rate), redu...
Q: 1. Carols first question was, “What is an enzyme” a. Use the enzyme lactase as an example, and expla...
A:
Q: Give the chemical principles involved in a positive Biuret test, Millon’s Test and Sakaguchi Test. W...
A: The color reactions of the proteins are useful in identifying the amino acids present in them. The c...
Q: The common amino acid residues in B-turis are proline and lysine
A: Proteins are the polymers of amino acids. There are four levels of structural organizations for the ...
Q: What would be the consequence for actin filament assembly/disassembly if a mutation prevented actin’...
A: The kinetics of filament formation are modulated by ATP-binding on actin subunits, with ATP-binding ...
Q: Describe the first step of B-Oxidation in peroxisomes ( name substrates , products, and eznymes) Wha...
A: Beta Oxidation: this is the process during which Fatty acids are broken down in mitochondria to rele...
Q: Use of Microbes for Improving Food Safety and Quality Yeast Probiotics Please have a whole paragrap...
A: An eaten probiotic is a live microorganism that has a favourable effect on the prevention and treatm...
Q: Describe the properties of laboratory-produced protocells.
A: Protocells are structures that arise when abiotic (non-living) components clump together. Despite th...
Q: Explain how gel filtration chromatography works. What type of gel will you used when the protein siz...
A: Chromatography is a process for separating components of a mixture based on their differential parti...
Q: Hello sir, I khow it lakes preciouS Lab. Sheet-Level time, Please, I have a report on this Semester ...
A: The measure of Cytotoxicity or cytotoxicity assays are required to measure or find out the ability o...
Q: Peptide Concentration (M) 2.5 x 104 5.0 x 104 10.0 x 104 15.0 x 104 Velocity (M/min) 2.2 x 106 5.8 x...
A: Km is the substrate concentration at half vmax.vmax is the maximum rate of reaction catalysed by Enz...
Q: Based on the relative increase in purity, place the purification procedures used for this enzyme in ...
A: Hi! Thank you for the question. We are authorized to answer one question at a time, since you have n...
Q: It is becoming possible for molecular biologists to synthesizealmost any protein desired. Suppose yo...
A: Comparative physiology is a branch of physiology that investigates and exploits the wide range of fu...
Q: What is measured at the A260 absorbance values?
A: DNA and RNA absorbs UV light due to heterocyclic rings of the nucleotides that is present in the nuc...
Q: How does adjusting Mg2+ concentration affect the PCR yield and specificity? How does adjusting prim...
A: Introduction: Polymerase chain reaction involves the ampliifcation of a particular piece of DNA to p...
Q: A small generic section of the primary structure of an a helix is given by -amino acid, -amino acid,...
A: Alpha-helix is an example of a secondary structure. Alpha-helix is formed by the hydrogen bonding be...
Q: While performing the Biuret test, a student added HCl instead of NaOH. How will a resulting solution...
A: Biuret test is the specific test for the identification of proteins in a sample solution. Biuret rea...
Q: 2. V The following plots show an oxygen binding curve for hemoglobin under a certain set of conditio...
A: The shown in question is Oxygen-Hemoglobin (O2-Hb) dissociation curve where Y axis represent O2-Hb s...
Q: What is an amino acid that has aromatic heterocyclic side chain?
A: Amino acids are monomers of protein they are linked with each other by forming peptide bond...
Q: How many protons are pumped out of the mitochondrial matrix for each pair of electrons extracted by ...
A: The NAD+ dependent Beta-hydroxacyl coA dehydrogenase cataluzes the third step of beta oxidation to f...
Q: From natural to synthetic ingredients in perfumery Explain and give example
A: When we smell perfume, we recognise certain fragrances. Scents of flowers, fruits, some foods, leath...
Q: Describe the glucose oxidase's general enzymatic class (oxidoreductase, transferase, isomerase, hydr...
A: Since you have asked multiple questions, we will solve only first question for you. If you want any ...
Q: Which of the following pairs of sugar/s and description is/are INCORRECT? * A. Xylose: Glycoprotei...
A: Monosaccharides are the simplest form of carbohydrates. They can be either aldose (aldehyde as the f...
Q: 24. Add formal charges directly to the structures. 2+ FHF -P C-Si-C H-AI-H H CI
A: Introduction: The formal charge of an atom is the charge that is calculated for an atom in a Lewis s...
Q: Just as all life shares a last universal common ancestor,all eukaryotes share a last eukaryotic comm...
A: The most recent common ancestor of all existing life on Earth, also known as the last universal comm...
Q: Three polypeptides, the sequences of which are represented using the one-letter code for their amino...
A: Proteins are composed of amino acids, which are bound together by peptide linkage. Amino acids conta...
Q: The initial pH of a 0.2M arginine solution is 14. a. What is the predominant structure of arginine i...
A: Arginine is a basic amino acid and it contains a guanidino group in its side-chain. Guanidino group ...
Q: Using the provided data on the activity part, what is the acquired R value? Is it acceptable or not?...
A: Absorbance is the unit measure of amount of light that passes through a particular solution at a giv...
Q: 1 Active Site Reaction Mechanism Inhibitor HN- 2•Zn NH2 HO. HO, NH2 N N- NH `NH NH NH, N' 'N N 'N NH...
A: Hi! Thank you for the question, as per the honor code, we are allowed to answer the ...
Q: Assessing the efficacy of glucosamine and chondroitin sulfate for arthritis pain Treatment with cho...
A: Glucosamine and chondroitin are two chemicals that exist naturally in your cartilage. Available in s...
Q: What would be the best buffering agent to choose if you wanted to buffer an enzyme reaction or tissu...
A: pH is the measure of the strength of H+ ion or Hydronium ions in solution. pOH is the measure o...
Q: What biological rationale can explain why there are so few variants observed at position 65 of the h...
A: The iron atom in the heme group can form a total of 6 coordinate bonds . 4 of these are formed with ...
Q: What is the principle involved in the method used in determining blood glucose? Explain
A: Glucose is a simple sugar which serves as a main energy source to all of cells in our body. Human bo...
Q: Protein-4YU4 is given, choose a part of it (containing at least 30 amino acid residues), find the am...
A: Protein-4YU4 is a PDB (protein data bank) name of mongoose haemoglobin, it is consist of four chains...
Q: what are the similarities between cell surface receptors and the nuclear receptors.
A: Receptors are proteins that bind to a ligand and undertake a characteristic physiological response i...
Q: D-Galactose DGlucose CORRECT INCORRECT Glucose and galactose are both Aldohexoses They constitute th...
A: Glucose and galactose are biomolecules categorized under carbohydrates. Generally carbohydrates are ...
Q: Biological activity of proteins is dependent on which? A. peptide bonds B. thermodynamic properties...
A: Proteins are composed of a linear chain of amino acids attached together via peptide bonds. Proteins...
Q: Suppose that you are tasked to determine the protein concentration of an unknown protein solution vi...
A: The concentration of unknown protein solution can be determined by plotting a graph between concentr...
Q: Modified true or false. Write the correct answer if the underlined word is false. 1. All standard a...
A: Any of a collection of organic compounds made up of a basic amino group (NH2), an acidic carboxyl gr...
Q: Given that the reduction potential Eo'= -320, +10, +816, and +50mV for NAD+, fumarate, 02 and G3P DH...
A: NAD+ oxidises glyceraldehyde 3-phosphate, incorporating inorganic phosphate (Pi) to create 1,3-bisph...
Q: Why is there such a large range of ∆G for the second step of glycolysis?
A: Glycolysis is the sequence of reactions that are used to break down glucose into two three-carbon co...
Q: 4. Determine the 260/280 ratios for each of the four samples. Record these values in Table 2.
A: Absorbance is the most common method to measure the purity of DNA yeild. This method is simple, hand...
Q: List the systematic classification of enzymes according to the Enzyme Commission indicating the clas...
A: A protein or RNA produced by living cells that is highly selective and catalytic to its substrates i...
Q: Cells generate ATP in two ways, what are they and which is the first to evolve? Which generates the ...
A: ATP means adenine triphosphate. ATP is the energy currency of the cell. ATP molecules are used in bi...
Q: You are given a suspension of purified mitochondria that are known to have adequate reserves of NAD+...
A: ATP synthesis occurs in the mitochondrial matrix by the ATP synthase enzyme. ATP synthase uses the p...
Q: tabulate the blood type of the members of your family (Both my parents and I have blood type O).
A: Blood is a fluid connective tissue and the most component of the circulatory system. composition of ...
Q: Make a graphical representation of activation energy and change of free energy in a reaction and com...
A: Enzymes are the substances that catalyse reactions by increasing the rate of reaction. They are usua...
Q: Tests Principle behind functional group that will give evidence of a References the test a positive ...
A: Generally carbohydrates are defined as polyhydroxy aldehydes or ketones . carbohydrates also are ca...
Q: I. Research for structure (submit on short bond paper, handwritten only)
A: 1. Phenolphthalein, (C20H14O4), organic compound of the phthalein family commonly used as an acid-ba...
Q: 1. Predict the membrane orientation of a protein that is synthesized with the following features: (D...
A: The insertion of membrane proteins in their right orientation is decided by multiple sequences that ...
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Circle and label the hemiacetal functional group and the acetal functional group in each of thefollowing carbohydrates:Long explanations are not needed. Direct answers would suffice. a. Which of the following is not a complex conjugated carbohydrate? I. lipopolysaccharide II. proteoglycan III. glycoprotein IV. peptidoglycan V. oligosaccharide b. Gluconic acid is derived from _______ while glucuronic acid is derived from ______. I. glucose: gulose II. galactose: glucose III. gulose: glucose IV. glucose: glucose V. glucose: galactoseTru or false 1. Every time our sugar level drops, glycogen is broken down to provide glucose in a process called syneresis. 2. If plants store glucose in the form of starch, animals store glucose in the form of glycogen. 3. Maltose is the most abundant biopolymer and is the basic component of cell wall of plants. 4. A long chain of monosaccharides linked by glycosidic bonds is known as a monosaccharide. 5. People who suffer from lactose intolerance do not produce lactase necessary to break down glucose and galactose.
- Discuss the ease of hydrolysis (break down) for each of the 4 polysaccharides (glycogen, amylose, amylopectin, cellulose).Function of citric acid? Give me handwritten answer with explaination?Explain these results. Why did glucose result in the highest rate? Why did sucrose result in a lower rate than fructose, and starch even lower? Recall that sucrose is a disaccharide composed of glucose and fructose, and that starch is a polysaccharide composed of many glucose molecules.
- BIOMOLECULES - MULTIPLE CHOICE - Please answer properly QUESTION : From these enumerated functions of biomolecules, identify the one that is NOT a common function of lipids? A. provides flavor in food B. long term energy C. building of muscles D. used to make hormonesCompare and contrast lactose intolerance with galactosemia.(Hint: Make a table.)BIOMOLECULES - MULTIPLE CHOICE - Please answer properly QUESTION : Which of the following best describes an apoenzyme? A. it is a non-protein group B. it is a complete, biologically active conjugated enzyme C. it is a protein portion of an enzyme D. it is a prosthetic group
- Disadvantages of cholesterolTest for Monosaccharides: Clinistix Carbohydrate molecules range from the very simple to the very complex. The word “saccharide” means sugar, while the prefixes “mono”, “di” and “poly” categorize carbohydrates according to how many simple sugar molecules are joined together to form the carbohydrate. To test for the presence of monosaccharide, we will be using Clinistix (a product that is used to test for the presence of glucose in urine). See the label (in video) for details. These test strips are used by dipping the colored end into the solution, then comparing the color to the chart on the bottle. The positive color(s) is/are __________________________. The negative color is ________________. Test for Starch (a Polysaccharide): Povidone Iodide Test Plant cells manufacture glucose molecules during photosynthesis and store them in large starch molecules. Because starch is a long chain of many glucose units all joined together, it is a polysaccharide. Povidone Iodide (which…True or false? Cellulose microfibrils are bonded together with other polysaccharides hence providing resistance to compression.

