NaBr catalyst about to be added to reaction miture но,(о) in acidic solution 2 Br (aq) + H,O,(aq) + 2 H* (aq) Brz(aq) + H,O,(aq) Br2(aq) +2 H2O(1) 2 Br (aq) + 2 H* (aq) + O2(8) brown colorless bubbles A Figure 14.21 Homogeneous catalysis. Effect of catalyst on the speed of hydrogen peroxide decomposition to water and coxygen gas.
NaBr catalyst about to be added to reaction miture но,(о) in acidic solution 2 Br (aq) + H,O,(aq) + 2 H* (aq) Brz(aq) + H,O,(aq) Br2(aq) +2 H2O(1) 2 Br (aq) + 2 H* (aq) + O2(8) brown colorless bubbles A Figure 14.21 Homogeneous catalysis. Effect of catalyst on the speed of hydrogen peroxide decomposition to water and coxygen gas.
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter13: Electrochemistry
Section: Chapter Questions
Problem 13.90PAE: A chemical engineering student is studying the effect of pH on the corrosion of iron. Ellie...
Related questions
Question
In Figure 14.21, we saw that Br - (aq) catalyzes the decomposition
of H2O2(aq) into H2O(l) and O2(g). Suppose that
some KBr(s) is added to an aqueous solution of hydrogen peroxide. Make a sketch of [Br - (aq)] versus time from
the addition of the solid to the end of the reaction.
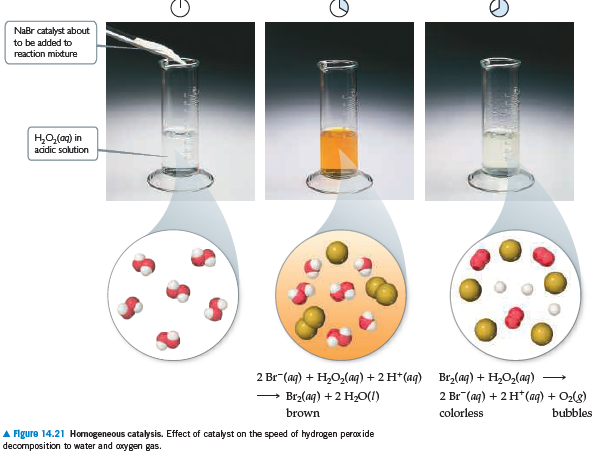
Transcribed Image Text:NaBr catalyst about
to be added to
reaction miture
но,(о) in
acidic solution
2 Br (aq) + H,O,(aq) + 2 H* (aq) Brz(aq) + H,O,(aq)
Br2(aq) +2 H2O(1)
2 Br (aq) + 2 H* (aq) + O2(8)
brown
colorless
bubbles
A Figure 14.21 Homogeneous catalysis. Effect of catalyst on the speed of hydrogen peroxide
decomposition to water and coxygen gas.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning