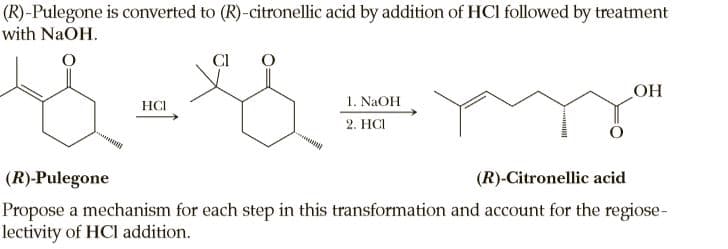
(R)-Pulegone is converted to (R)-citronellic acid by addition of HCI followed by treatment with NaOH. CI 1. NAOH 2. HCI HCI HO (R)-Pulegone (R)-Citronellic acid Propose a mechanism for each step in this transformation and account for the regiose- lectivity of HCI addition.
Q: 1. using curved arrows draw the mechanism to show the SN2 reaction of hydroxide ion with (R)…
A:
Q: Consider the following synthetic sequence: он A Cro/H* F CH;Br CH,MgBr E в Br2.FeBr3 G C3H,Bro…
A: In the given reaction sequence alkyl halide first converts into Grignard reagent which reacts with…
Q: Explain why hydroxide ion catalyzes the reaction of piperidine with 2,4-dinitroanisole but has no…
A: In the reaction of piperidine with 2,4-dinitroanisole, the rate determining step is the removal of…
Q: Sodium Borohydride Reduction 1. Give stereochemical formulas (such as Fischer projects) for all the…
A: Here we have to write structures of all stereo isomers that are fomed in the reduction of following…
Q: 1. NaNH/sivi NHy-78°C 1. NANH/sivi NH-78°C B(CHa) A( CHa) H-CEC-H 2. 1-Bromodekan 2.1-Bromo-5-metil…
A:
Q: Identify compounds A and B, two synthetic intermediates in the 1979 synthesis of the plant growth…
A: Gibberellic acid is a hormone found in plants and fungi. It has a chemical formula as C19H22O6. It…
Q: Illustrate Dehydration of 2° and 3° ROH—An E1 Mechanism ?
A: Alcohols have a hydroxyl functional group attached to an alkyl group. Because of electronegativity…
Q: Identify A, B, and C, intermediates in the synthesis of the ve-membered ring called an…
A: The base attacks the hydrogen attached to the carbon and formation of enolate ion takes place and…
Q: Ammonia and amines react with epoxides with the same stereospecificity as anionic nucleophiles. Draw…
A:
Q: The treatment of isoprene [CH2=C(CH3)CH=CH2] with one equivalent of mCPBA forms A as the major…
A:
Q: Give the reason of the following statements: 1. An achiral starting material in enolate alkylation…
A: Wagner-Meerwein rearrangement is basically the shifting of the alkyl ion of H+ ion to form the most…
Q: What spectral features including mass spectra, IR spectra, proton spectra and carbon spectra allow…
A: In case of benzonitrile, a weak IR-absorption occurs at 2260-2222 cm-1 which is due to C-N triple…
Q: An acid–base reaction of (R)-sec-butylamine with a racemic mixture of 2-phenylpropanoic acid forms…
A: The reaction between racemic mixture of 2 -phenylpropanoic acid with (R) -sec-butylamine is an acid…
Q: CH3 CH2 H3C CH3
A: A compound with carbon and hydrogens is known as hydrocarbons. These hydrocarbons can be synthesized…
Q: 1. Reaction of pyridine with Br2 yields what? 2. Reaction of pyrrolidine with Br2 in the presence…
A: Reaction of pyridine with Br2 :-
Q: What is the SN1 mechanism between t-Butyl bromide and ethanol?
A: In first step bromine will depart to form carbocation. In the second step ethanol will attack as…
Q: 0.1 a) Account for the differences observed in the pka vatues of the following saturated…
A:
Q: Propylene oxide is a chiral molecule. Hydrolysis of propylene oxide gives propylene glycol, another…
A: A. The mechanism of acid catalyzed hydrolysis of (R)-propylene oxide (treatment with H2SO4/H2O)…
Q: Propose a mechanism for the reaction of pent-1-yne with HBr in the presence of peroxides. Show why…
A: Addition reactions are reactions in which double or triple bonds are broken and substituents are…
Q: (a) Draw a stepwise mechanism for the reaction of ethyl hexa-2,4-dienoate with diethyl oxalate in…
A: a.
Q: Identify compounds A and B, two synthetic intermediates in the 1979 synthesis of the plant growth…
A: Gibberellic acid is a hormone found in plants and fungi. Its has a chemical formula as C19H22O6. It…
Q: 10: H :O: CHO CÁC CHỊ CHO C CH3 Baeyer- Villiger reaction 0 H CH3C-0-C-CH3 CH3 8. CH₂C-0-H
A: Given reaction is : Give the mechanism of the reaction = ?
Q: Busulfan is a cell cycle non-specific alkylating antineoplastic agent or simply a chemotherapy drug…
A: The final product is given below -
Q: Explain the difference in reactivity between CH3OH2 and CH3OH in a nucleophilic substitution…
A: The alcohol contains a two lone pair and therefore, will be reactive for the attack on reagent to…
Q: What is the best and less complex synthetic route to obtain (2‐methylprop‐1‐en‐1‐yl)benzene starting…
A: Synthesis of (2‐methylprop‐1‐en‐1‐yl)benzene starting from benzenesulfonic acid - Benzene sulfonic…
Q: 3. R-ZnX is not as reactive towards carbonyl compounds as Grignard reagents (R-MgX). 4. t- butyl >…
A: A chemical reaction mechanism represents the way to form and break the chemical bonds between…
Q: Arrange the following compounds in order of increasing reactivity to nucleophilic addition. I.…
A: Nucleophilic addition reactions are the addition reactions in which a nucleophile attacks the…
Q: What is the major product generated when p-t-butyl aniline undergoes Friedel-Crafts alkylation…
A:
Q: Propose a synthetic pathway for cis-[NiCl2(CO)2] using trans-effect. b. Propose the methods to…
A: The ability of a group to direct the incoming group in a direction trans to itself is known as trans…
Q: Q. Explain the following observation 1. Aldehyde group (-CHO) act as a meta director and…
A:
Q: _2. Which of the following is not true of Friedel-Crafts acylation reactions? a. They require an…
A: Friedal-Crafts acylation reaction is an electrophillic substitution of aromatic ring with an acyl…
Q: a) Wolff-Kishner reduction of compound W gave compound A. Treatment of A with m- chloroperbenzoic…
A: Oxidation and reduction are common class type of organic reactions. In oxidation reaction, oxygen is…
Q: Rank the aryl halides in each group in order of increasing reactivity in nucleophilic aromatic…
A:
Q: Explain why hydroxide ion catalyzes the reaction of piperidine with 2,4-dinitroanisole but has no…
A: The mechanism of the reaction taking place between piperidine and 2,4- dinitroanisole is as follows:
Q: Juvabione is produced by fir trees of the genus Abies and is known to inhibit insect reproduction…
A:
Q: A retrosynthetic analysis of 1 was undertaken suggesting a forward synthesis starting from compounds…
A: We have to synthesize organic compound 1 :
Q: Explain the difference in reactivity between CH3OH2 and CH3OH in a nucleophilic substitution…
A: The alcohol contains a two lone pair and therefore, will be reactive for the attack on reagent to…
Q: Illustrate the Dehydration of a 1° ROH—An E2 Mechanism ?
A:
Q: What is the detailed reaction mechanism between 1-indanone and veratraldhyde, with a NaOH base and…
A:
Q: I need to show a mechanism for trans cinnamic acid reacting with dibromide to get (2S,…
A: The reaction is passing through the formation of three member cyclic bromonium ion which is attacked…
Q: 2. Consider the following scheme. NH2 NH2 Reagent A Но ÓH Compound T HO Compound S KOH, NH,NH2…
A: a) The IUPAC name of the given compound T is, 1-amino-1-(4-hydroxy phenyl) propan-2-one b)…
Q: Zn D HNO, В A HCI H2SO4 C or Br2 FeBr3 Br2 FeBr3 F H30* E heat
A: Here, Reactions are follows as : Nitration, Reduction, Acetylation, Bromination and then Reductive…
Q: (m) DMP, CH2CI2 PCC, CH2CI2 Pyridinium chlorochromate in methylene chloride solvent Aco OAc -OAc OH…
A:
Q: The biosynthesis of lanosterol from squalene has intrigued chemists since its discovery. It is now…
A: a. The first step is the protonation of hydroxyl group. In the next step, elimination of water…
Q: (a) Give reasons :(i) CH3—CHO is more reactive than CH3COCH3 towards HCN.(ii) 4-nitrobenzoic acid is…
A: Answer (a) Introduction: An acid is a substance which can donate H+ ions. An acidic substance has…
Q: The treatment of isoprene [CH2 = C(CH3)CH = CH2] with one equivalent of mCPBA forms A as the major…
A: When alkenes treated with mCPBA it acts an epoxide agent an dconverts the alkene into epoxide ring.…
Q: As described in Section 9.17, the leukotrienes, important components in the asthmatic response, are…
A: The conversion of arachidonic acid to 5-HPETE takes place in three steps. The first step involves…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- A student wanted to use the Williamson ether synthesis to make (R)-2-ethoxybutane. He remembered that the Williamson synthesis involves an SN2 displacement, which takes place with inversion of configuration. He ordered a bottle of (S)-butan-2-ol for his chiral starting material. He also remembered that the SN2 goes best on primary halides and tosylates, so he made ethyl tosylate and sodium (S)-but-2-oxide. After warming these reagents together, he obtained an excellent yield of 2-ethoxybutane. What would have been the best synthesis of (R)-2-ethoxybutane?A student wanted to use the Williamson ether synthesis to make (R)-2-ethoxybutane. He remembered that the Williamson synthesis involves an SN2 displacement, which takes place with inversion of configuration. He ordered a bottle of (S)-butan-2-ol for his chiral starting material. He also remembered that the SN2 goes best on primary halides and tosylates, so he made ethyl tosylate and sodium (S)-but-2-oxide. After warming these reagents together, he obtained an excellent yield of 2-ethoxybutane. What enantiomer of 2-ethoxybutane did he obtain? Explain how this enantiomer results from the SN2 reaction of ethyl tosylate with sodium (S)-but-2-oxide.For butenafine, develop a synthesis that starts with napthalene and benzene. (aromatic methyl groups can be converted to alkyl bromides using Br2/light, and aldehydes by heating the alkyl bromide in DMSO, eg. ArCH3 to ArCH2Br then to ArCHO).
- (a) A compound known to be a substituted cyclohexanone derivative has lamda max of 235 nm. Could this compound be a conjugated dienone? explain (b) (i)For this compound, how many nm must be accounted for by substituents? (ii) What are the substituents and the points of substitution that may occur having accounted for the 20nm?A.) Draw two schemes, For the first scheme combine A and B. For the second, combine A and C. b.) For each scheme predict all products. c.) Label the major and minor products d.) Show all stereochemical configurations in the products, give reasons regarding sterochemical outcomes for each rxn. e.) Propose an appropriate solvent for each scheme, give a reason. f.) Discuss what by what mechanism your products are formed and why. (SN2, E2, E1, SN1)Explain how the reaction of (CH3)2CHCH(Cl)CH3 with H2O yields two substitutionproducts, (CH3)2CHCH(OH)CH3 and (CH3)2C(OH)CH2CH3
- Provide the mechansim using curved arrows of the reaction of p-t-butyl phenol treated with acetic anhydride in AlCl3. Include resonance stabilized intermediates and if more than one product is formed, label them as major, minor, etc. thank you for the helpmany useful synthetic produces involve sequences of adol, Claisen, Michael, and Mannich reactions. Write the mechanism for each transformation.Propose a mechanism and use stability factors to account for the formation of a major product in the reaction of pyrrole with Br2, FeBr3.
- While dehydrating 2-Methlycyclohexanol to 1-Methlycyclohexanol, and 3-Methlycyclohexanol 1)what is the common problem associate with E1 reaction? Discuss the intermediate. 2) Briefly compare and contrast E1 vs E2 Elimination reactions. INclude details such as speed of the reactions in relation to each other, rate-determining steps (E1), and stereochemical requirements (E2). Mention whether the steps of the reactions are reversible and discuss the conditions that can drive the equilibrium to favor product formation.Disparlure, 7,8-cis-epoxy-2-methyloctadecane, is an attractant for the Gypsy Moth. Disparlurecan be synthesized in high yield and stereoselectivity. The synthetic material is as active as the natural substance.a) Show how you could make (+/-)-disparlure from 6-methyl-1-bromoheptane and undecanal.(a) Illustrate the following name reactions giving suitable example in each case :(i) Clemmensen reduction (ii) Hell-Volhard-Zelinsky reaction(b) How are the following conversions carried out?(i) Ethylcyanide to ethanoic acid (ii) Butan-l-ol to butanoic acid(iii) Benzoic acid to m-bromobenzoic acid

