Reaction 1 BaCh and NazSO4 Initial appearance of of BaCl2 solution Transparent clear liquid Initial appearance of Na2SO4 solution Transparent clear liquid Observation for the Cloudy solution, White precipitate formed reaction Balance the chemical equation BaCl, (aq) + _NaySO, (aq) BaSO, (s) + NaCl (aq) Identify type of reaction Reaction 2 CaClz and NazCOs Initial appearance of CaCl2 solution Transparent clear liquid Initial appearance of NazCO3 solution Transparent clear liquid Observation for the Cloudy solution, White precipitate formed reaction Balance the chemical equation _CaCl, (aq) + NayCO, (aq) CaCO, (s) + NaCl (aq) Identify type of reaction Reaction 3 NaCl and AgNO Initial appearance of NaCl solution Transparent clear liquid
Reaction 1 BaCh and NazSO4 Initial appearance of of BaCl2 solution Transparent clear liquid Initial appearance of Na2SO4 solution Transparent clear liquid Observation for the Cloudy solution, White precipitate formed reaction Balance the chemical equation BaCl, (aq) + _NaySO, (aq) BaSO, (s) + NaCl (aq) Identify type of reaction Reaction 2 CaClz and NazCOs Initial appearance of CaCl2 solution Transparent clear liquid Initial appearance of NazCO3 solution Transparent clear liquid Observation for the Cloudy solution, White precipitate formed reaction Balance the chemical equation _CaCl, (aq) + NayCO, (aq) CaCO, (s) + NaCl (aq) Identify type of reaction Reaction 3 NaCl and AgNO Initial appearance of NaCl solution Transparent clear liquid
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter11: Solutions
Section: Chapter Questions
Problem 39P
Related questions
Question
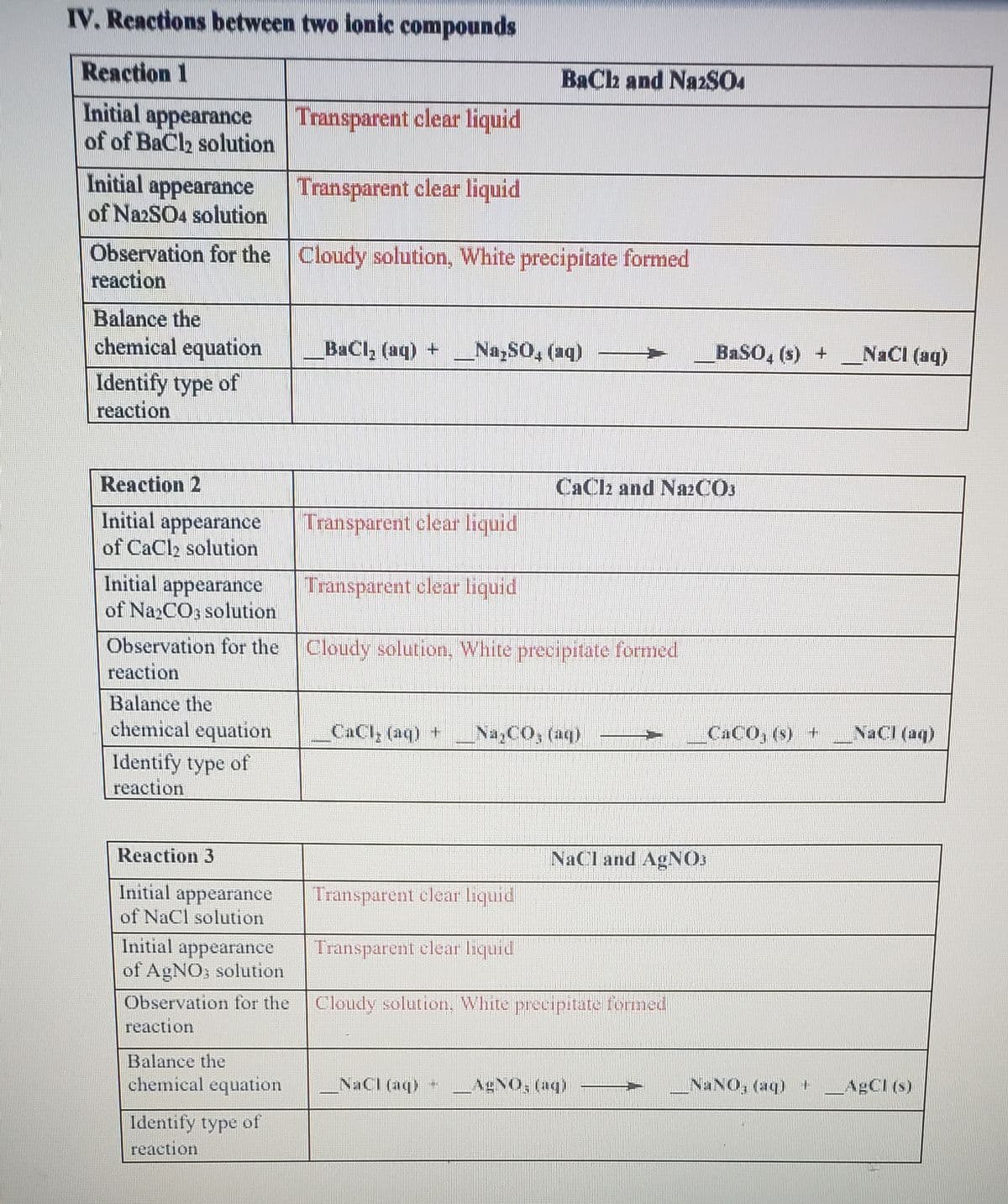
Transcribed Image Text:IV. Reactions between two lonic compounds
Reaction 1
BaCh and NazSO
Initial
appearance
Transparent clear liquid
of of BaCl2 solution
Initial appearance
of NazSO4 solution
Transparent clear liquid
Observation for the Cloudy solution, White precipitate formed
reaction
Balance the
chemical equation
BaCl, (aq) +
Na,SO, (aq)
BASO, (s) +
NaCl (aq)
Identify type of
reaction
Reaction 2
CaClz and N22CO3
Initial appearance
of CaCl solution
Transparent clear liquid
Initial appearance
of Na2CO3 solution
Transparent clear liquid
Observation for the Cloudy solution, White precipitate formed
reaction
Balance the
chemical equation
_CaCl, (aq) +
Na,CO, (aq)
CACO, (s) +
NaCI (aq)
Identify type of
reaction
Reaction 3
NaCl and AgNOs
Initial appearance
of NaCl solution
Transparent clear liquid
Initial appearance
of AgNO; solution
Transparent clear liquid
Observation for the
reaction
Cloudy solution, White precipitate formed
Balance the
chemical equation
NaCl (aq)
AgNO, (aq)
NANO, (aq) LABCI (s)
Identify type of
reaction
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax



Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning