Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter14: Elimination
Section: Chapter Questions
Problem 31CTQ
Related questions
Question
Drawing Reaction Mechanism Arrows

Transcribed Image Text:5. Drawing Reaction Mechanism Arrows.
For the reactions that have TWO starting materials, indicate and label which species is the nucleophile and which
is the electrophile. (HINT: Look at the reactants and look at the products to see where bonds form and where
bonds break! Using your pK, table and bond polarity predictions will help!).
Then, draw curved arrows to show the movement of electrons for Bond Forming and Bond Breaking in each of
the reaction steps.
Finally, identify and label what type of elementary step is represented for each mechanism.
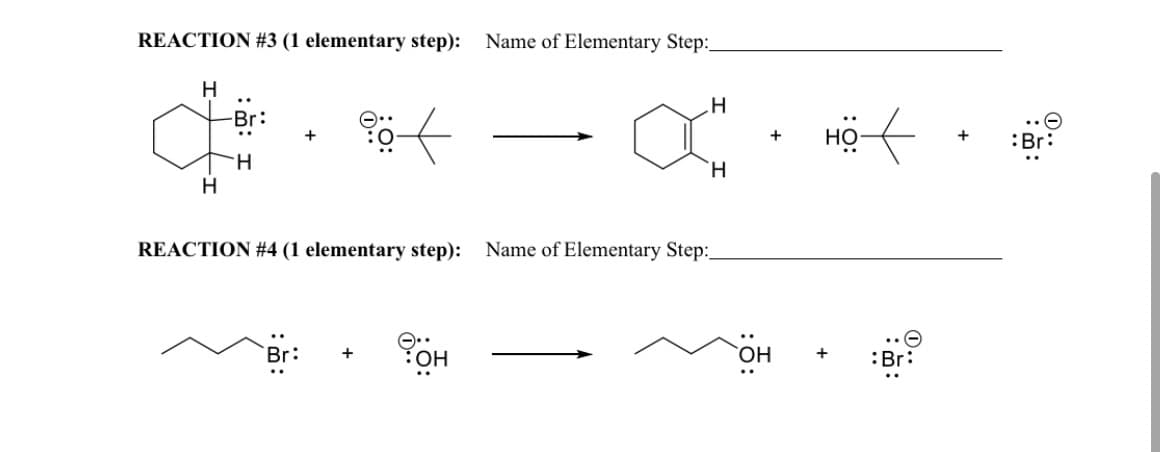
Transcribed Image Text:REACTION #3 (1 elementary step):
Name of Elementary Step:_
H
-Br:
но-
+
HO
..
:Br:
`H
H
REACTION #4 (1 elementary step):
Name of Elementary Step:
`OH
:Br:
0..
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning