Red 3 0.7 0.6 0.5 0.4 y = 14134x + 0.0109 0.3 R2 = 0.99982 %3D 0.2 0.1 0.00E+00 1.00E-05 2.00E-05 3.00E-05 4.00E-05 5.00E-05 MOLARITY ABSORBANCE
Q: I need help to produce a standard curve with the attached worksheet. The curve/graph should be…
A: The dissolution of the known mass of the solute in a specified volume of the solvent produces the…
Q: If, with a beam of monochromatic light, 65.0% of the incident light is absorbed in its passage…
A: Given : The percentage of monochromatic light absorbed by the solution is 65.0%. Assume the…
Q: A solution of a specific vitamin has a åmax of 271 nm and a concentration of 2.73 × 10-7M. The…
A: Given that : Concentration of the solution (C) = 2.73 x 10-7 M Absorbance of the solution (A) =…
Q: extinction coefficient = ε, path length = b, concentration C A sample containing an unknown…
A:
Q: Beers plot having concentration of cobalt nitrate solutions (in units M) gives best fit data as…
A: We have to predict the concentration in M and ppm.
Q: 4. To analyze the Cu(NO3)2 unknown solution in Part 3, your group constructed a plot of absorbance…
A: One of the techniques that is used in quantitative analysis is colorimetry. The technique uses color…
Q: Given the following values, use the absorbance formula to find the hemoglobin concentration of…
A: #1: Given the absorbance of standard solution, As = 0.600 concentration of the standard solution,…
Q: Carotene (MM=536.872 g/mol), which is found in carrot juice, was measured by visible…
A: Here standard addition of 0.020 mol of carotene standard was added to find the concentration of…
Q: Your lab partner gets a little confused and prepares a dilution of your protein sample by combining…
A: In case of dilution of solutions, we may use the expression: c1V1=c2V2where,c refers to the…
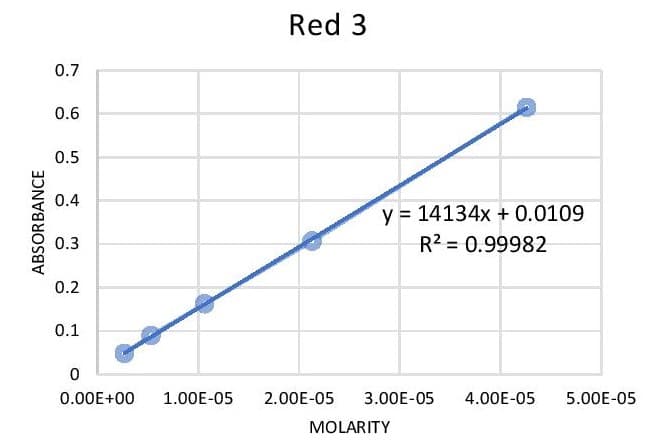
Q: You have a dilute solution of Red 3 that has an absorbance of 0.415. What is the molarity of the…
A: Calibration curve is used to determine the concentration of unknown sample. They can be constructed…
Q: What is the concentrations in mg/100 ml of the Carbimazole solution of drugs, a(1%, 1 cm)…
A: Given Carbimazole solution of drugs, a(1%, 1 cm) value = 600 at 291nm absorbance at 291nm = 1.2
Q: A standard curve for a compound was created with a series of standards, and is shown below. If an…
A: To calculate the concentration of unknown, we would use the straight line equation of the plot and…
Q: A solution of a specific vitamin has a Âmax of 245 nm and a concentration of 4.15 × 10-/M. The…
A:
Q: rue or False 1. Sample preparation of infrared spectroscopy uses KBr as the blank because Kbr has…
A: Ans.1. Sample preparation of infrared spectroscopy uses KBr as the blank because KBr has 100%…
Q: Еxercice 1: Complete the following equation 1) 1Pd → Ru + 106 2) "Rh → ... +He 3) 180Та -> +-je ....…
A: By calculating difference in atomic weight and atomic number of elements both side of arrow we can…
Q: f you were plotting a graph of light absorbance versus the concentration of Cu2+ in a solution,…
A: Answer is explained below. For plotting a graph of light absorbance versus the concentration of Cu2+…
Q: A scientist has an aqueous solution with an unknown concentration. He uses a 1-cm cuvette and…
A:
Q: Guanosine has a maximum absorbance of 275 nm. ϵ275=8400M−1cm−1, and the path length is 1 cm. Using a…
A: In this case we use the beer's formula A=€×l×c
Q: If A=absorbance, B=molar absorptivity, C=length of light path, D=concentration, and you plot a graph…
A: Introduction: Beer-Lambert law: Beer Lambert law shows the relationship between the absorbance and…
Q: Please refer to the graph in the lab document. If the absorbance of a solution containing FeSCN2+ is…
A: Given, Concentration of [FeSCN]2+ Absorbance Percent…
Q: What absorbance value corresponds to 40.0% T? A =
A: Note: According to our guidelines we are supposed to answer only one question. Kindly repost other…
Q: O The pH of the solution has decreased. O The pH of the solution has increased. O The green…
A: According to the question, the color of phenolphthalein changes from clear to pink in an acid-base…
Q: 1) of a solution appears green, what color of light is most likely to absorbed the strongest?
A: The segment of an electromagnetic spectrum which is visible by the human eye is known as visible…
Q: 1. Look at the spectrum shown and answer the following questions: 1.4 1.2 - 0.4 0.2 24n 280 320…
A: electronic transition from lower energy level to higher energy level leads to formation of energy…
Q: A solution of FeSCN2+ has an absorbance of 0.237. What is its concentration?
A:
Q: 2. What is the concentration (mM) of a solution of tryptophan if the absorbance at 280 nm is 0.850?…
A:
Q: Convert the 98% T to absorbance: a. 0.008 b. 0.08 c. 0.021 d. 0.21
A: A (absorbance) = - logT T = transmittance... Rule, A = 2 - log (T%) Given T% = 98
Q: Which of the following is FALSE about the absorbance of a compound? O A. Organic solvents have…
A: Ans. (A). In organic chemistry, solvents can have an effect on solubility, stability and reaction…
Q: Rank these colors from the best to the least absorber of radiation. (Best absorber - I; Least…
A: The color which absorbs least of radiation is white. The color which absorbs most of radiation is…
Q: A compound whose molecular mass is 158 g / mol has a molar absorptivity of 3.57 x 104 . How many…
A:
Q: a.) 1.00g KCI in 75.0mL of soutin = b.) 1.00g NazcrOy in 75.0mL of Selutiin= C.) 20.0g MaBrz in…
A: Molar Concentration of the following solutions can be calculated as -
Q: Absorbance Transsmission 0.730 17.8 0.641 22.8 0.400 31.8 0.340 45.2 0.211 62.0 0.076 85.4 0.020…
A: Given:
Q: What is the absorbance of a theoretical compound in a 5.26 x 10-5 M solution, a path length of 1.5…
A: The relationship between the absorbance and concentration is given by A = ECl where A = absorbance…
Q: solution with %T = 75.9% has an absorbance equal to: A = log (1 T) T= (% T 100)
A: Relationship between absorbance and transmittance, A = - log T And Relationship between absorbance…
Q: If the concentration of a food coloring solution is increased by a factor of 3, how will the…
A: According to Beer Lambert law, the absorbance of any solution is directly proportional to the…
Q: A sample was prepared by diluting 1 mL of a sample to 5 mL. The resulting solution has an…
A: Given: Absorbance is 0.463 of 5 mL solution. y = 0.463 y = 1.8711x 0.463 = 1.8711x x = 0.2474 M
Q: Does absorbance increase or decrease as the solution concentration of the absorbing substance…
A: From given Using Beer Lambert's law concentration of species and absorbance is interrelated and its…
Q: A scientist has an aqueous solution with an unknown concentration. He uses a 1-cm cuvette and…
A: Absorbance (A) of a solution at a specific wavelength is proportional to concentration (C) of…
Q: CONCENTRATION ABSORBANCE 0.879 1.00 M C. १52 0.4 0.50 M 0,221 0,089 0.25 M 0.10 M
A: A table of data showing relationship between concentration of CuSO4 and absorbance.
Q: Guanosine has a maximum absorbance of 275 nm. ϵ275= 8400M−1cm−1 and the path length is 1 cm. Using a…
A: The concentration of guanosine is calculated by using Beer’s law. The expression is as follows,
Q: 1. AL + HНе 30 15P + on -> 63 2Cu + H 2 on + 63 Zn. 30 65 3. 30Ca 65 29SC 2.
A: Since you have asked multiparts questions, we will solve the first three sub parts questions for…
Q: Can you help me on questions 2 AND 3
A: To solve this problem we can use beer’s law equation:
Q: What type of compound corresponds to the IR spectrum shown here? MICRONS 25 2.62.72.82.9 3 100 3.5…
A: IR spectrum is used to study and identify chemical substances or functional groups present in them…
Q: At 425 nm a 1.2 x 10-3 M solution of compound Q has Absorbance = 0.879. Another solution of Q is…
A: Given that, at 425 nm, A 1.2×10-3 M solution of compound Q has Absorbance is A1 = 0.879, ie,…
Q: 1- The compound with 3 electronic transitions in the UV-Visible spectrum a) [Cr(acaac)] b)…
A: b) [Cu(phthalocyanine)] b) εl.εD and λ a) they have the same absolute configuration
Q: (b) A compound having molecular weight 151.163 g/mol is determined in a sample. A 0.0302 g sample…
A:
Q: topes listed, pr 23 Na or 25Na b. 11 48 Ca or 48Sc 20 с. a. or 11 92TC 20Ne or 10Ne 10 40 Ca 45 ca…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: Procaine hydrochloride (MW = 273; L iso value = 3.37). Calculate its NaCl Equivalent 0.24 0.19…
A:
Q: Consider the diagram below to answer questions 5 and 6. R = 0.70 R = 0.55 R; = 0.47 Eluent: EtOAC…
A: Rf= 0.55 correspinding to compound 1. Since more is the polarity more will be the Rf value. Thus…
Write the equation for Abs = slope (M) + b using the actual values for slope and y-intercept.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Determine the percentage Fe in a sample of limonite from the following data:Sample : 0.5000g ; KMnO4 used = 50.00 ml ; 1.000ml of KMnO4 is equivalent 0.005317 g Fe,FeSO4 used = 6.00 ml; 1.000ml FeSO4 is equivalent 0.009200 g FeO ( ans 44.59 %)The formula for 1000 g of polyethylene glycol ointment calls for 600 g polyethylene glycol 400. At $ 19.15 per pint, what is the cost of the polyethylene glycol 400, specific gravity 1.140 , needed to prepare 4000 g of ointment?1) How many milliliters of 70% ethanol and 20% ethanol must be mixed to prepare 500ml of 30% ethanol? 2) A gaseous blend of nitric oxide and nitrogen (NOmax) contains 0.09% v/v nitric oxide (NO) and 99.92% v/v nitrogen. Express the strength of nitric oxide in PPM (round to whole number) 3) The ratio strength of a 10mL vial of neostigmine methylsulfate injection USP is 1:1500.Calculate the amount of neostigmine methylsulfate in milligrams contained in the 10mL vial. (Round to the whole number).
- Prepare 1500mL of 0.5N H2SO4 solution with specific gravity of 1.84g/mL and assay of 97%How would you prepare 75 mL of an H2SO4 solution at 20% in weight (density=1.1394 g/mL, richness=227.9 g/L) starting from the commercial H2SO4 solution (98%, density=1.8361 g/mL, richness=1799 g/L)? Please choose among the most appropriate procedure which better describes the preparation of the previous diluted H2SO4 solution: Select one or more: a. Take the required volume of commercial H2SO4 solution with an adequate pipette, pour it into a graduated flask and add water up to the final volume (75 mL in this case). Homogenize the flask by stirring it gently. b. Take a beaker and put about 40 mL of distilled water. Then take the required volume of commercial H2SO4 solution with an adequate pipette and pour them over the beaker. Let the solution be warm and pour the mixture into a graduated flask and add water up to the final volume (75 mL in this case). Homogenize the flask by stirring it gently. c. Take a beaker and put about 40 mL of distilled water. Then take the required volume…Referring to the Al-Si Eutectic diagram discussed on the Phase Diagrams-Lever Rule handout, calculate the mass fraction of the phase, silicon, given a sample with 51.7 wt% aluminum at 576.99 ˚C.
- Describe the solution's preparation: i). 500 mL 0.250 M ethylene glycol (MW = 62.07) from a concentrated 31 % (w/w)solution that has a density of 1.0350 g/mL ii) 2.00 L of 0.108 M BaCl2 from BaCl2.2H2O salt iii) 750 mL of 6.00 M H3PO4 from the commercial reagent that is 86% H3PO4 (w/w)and has specific gravity of 1.71.Polyethylene terephthalate is an example of synthetic material in Materials and Engineering. 1. Describe the relation of its building blocks in connection to its “bulk” (materials) properties. (No need for essay, just a brief scientific explaination!) If you may, please send link as well of the reference that you use for the answer).The %purity of a powdered crude sample of Na2CO3 containing only inert impurities is to be determined by reacting 225.0 mg of the crude sample to 10.0 mL of 3.00 M HCl solution, and bubbling the resulting CO2(g) product in water that is at exactly 29 °C. After the reaction has completed, the level of the liquid inside the eudiometer rests 4.30 cm above the water level in the beaker. The graduation on the eudiometer indicates that the trapped gas is 44.37 mL. The experiment was done under a barometric pressure of 755.2 torr. a. How many moles of CO2 were collected? b. What is the percent purity of the sample? Round off to the nearest whole number
- The %purity of a powdered crude sample of Na2CO3 containing only inert impurities is to be determined by reacting 225.0 mg of the crude sample to 10.0 mL of 3.00 M HCl solution, and bubbling the resulting CO2(g) product in water that is at exactly 29 °C. After the reaction has completed, the level of the liquid inside the eudiometer rests 4.30 cm above the water level in the beaker. The graduation on the eudiometer indicates that the trapped gas is 44.37 mL. The experiment was done under a barometric pressure of 755.2 torr. What CASE does this experiment satisfy?Volatile and semi-volatile compounds can be analyzed by GCGiven the following: Solutions Flask 1 Flask 2 Flask 3 0.04 M KI 25.0 mL 25.0 mL 12.5 mL 1% starch 5.00mL 5.00mL 5.00mL 0.03 M Na2S2O3 5.50 mL 5.50 mL 5.50 mL 0.05 M H2SO4 5.00 mL 5.00mL 5.00mL 0.08 M NaCl 0 0 10.0 mL and Solutions Beaker 1 Beaker 2 Beaker 3 0.04 M H2O2 25.0 mL 12.5 mL 25.0 mL deioinized water 0 12.5 mL 0 Following the procedure Run 1 is when Beaker 1 is added to Flask 1 and the timer is started giving the time recorded for the run in the table below. This is repeated with Beaker 2 and Flask 2 for Run 2 and finally Beaker 3 and Flask 3 for Run 3: The results for these runs are given below: Run Time (seconds) 1 35 2 66 3 72 Calculate the following for each run: the initial molarities of I- and H2O2 (from the solutions tables) the rates of reaction [I2]/time (calculate the [I2] from [S2O32-] and the reaction: I2(aq) + 2 S2O32-(aq) → 2I-(aq) + S4O62-(aq) Choose the correct rate law for this reaction from your calculations.…

