Separate a mixture of Vitamin D and Vitamin B2 (riboflavin) by paper chromatography. The stationary phase is methanol (CH;OH) and the mobile phase is pentane (CSH12). Place the vitamins in order of elution. Explain why using IMF. (Circle polar sites on each molecule and label all 4 molecules as polar or non-polar.) Vitamin B, CH, H,C CH OH Vitamin D Он CH,OH
Separate a mixture of Vitamin D and Vitamin B2 (riboflavin) by paper chromatography. The stationary phase is methanol (CH;OH) and the mobile phase is pentane (CSH12). Place the vitamins in order of elution. Explain why using IMF. (Circle polar sites on each molecule and label all 4 molecules as polar or non-polar.) Vitamin B, CH, H,C CH OH Vitamin D Он CH,OH
Macroscale and Microscale Organic Experiments
7th Edition
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Kenneth L. Williamson, Katherine M. Masters
Chapter7: Extraction
Section: Chapter Questions
Problem 1Q
Related questions
Question
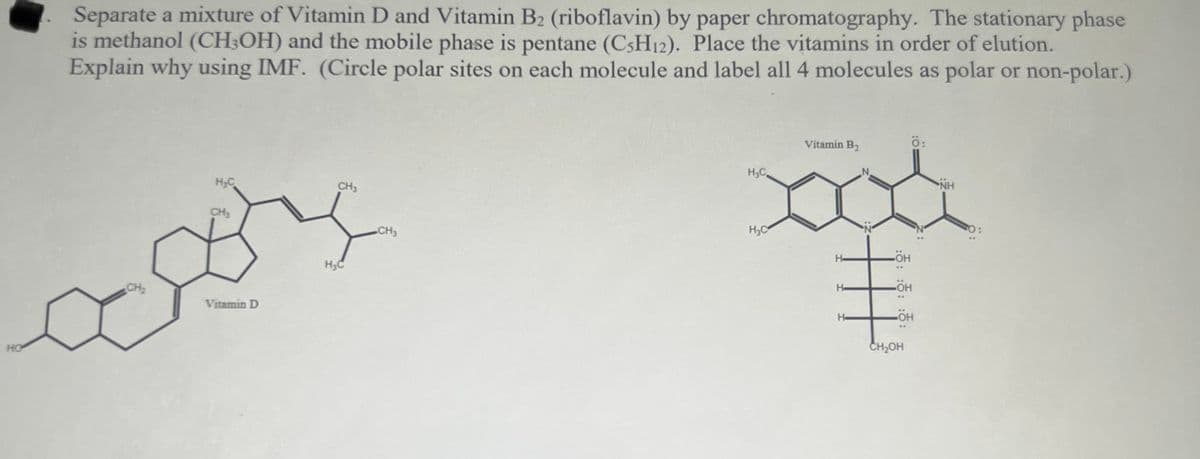
Transcribed Image Text:Separate a mixture of Vitamin D and Vitamin B2 (riboflavin) by paper chromatography. The stationary phase
is methanol (CH;OH) and the mobile phase is pentane (C3H12). Place the vitamins in order of elution.
Explain why using IMF. (Circle polar sites on each molecule and label all 4 molecules as polar or non-polar.)
Vitamin B2
H,C.
H;C
CH3
NH
CH,
CH3
H,C
H-
H3C
HO-
CH2
He
Vitamin D
He
HO
CH2OH
:애: :애: :애:
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT