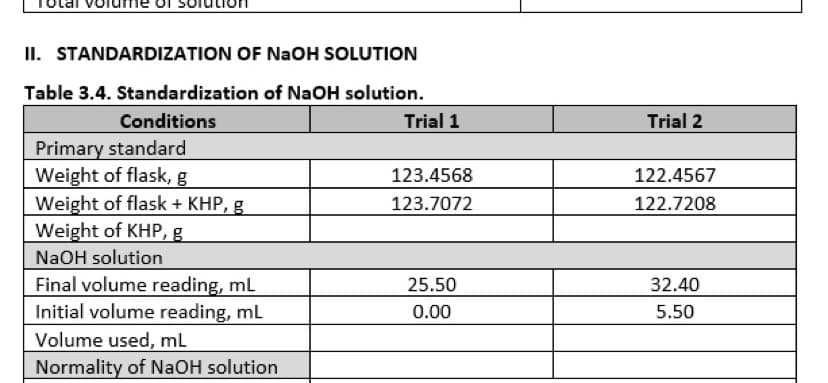
Table 3.4. Standardization of NaOH solution. Conditions Trial 1 Trial 2 Primary standard Weight of flask, g Weight of flask + KHP, g Weight of KHP, g NaOH solution Final volume reading, mL Initial volume reading, mL 123.4568 122.4567 123.7072 122.7208 25.50 32.40 0.00 5.50 Volume used, mL Normality of NAOH solution
Q: A nitrogen gas (MW = 28) occupies a volume of 5ft3 at 2550 lb/ft2 abs and 755oR. What are its…
A:
Q: Add Stoichmetric factor: ______CH3CH2OH + _______O2 yields ________CO2 + _______H20
A: Balancing reaction
Q: Brand of Sample: Trial 1 Volume of sample 10.00 mL 25.63 ml Final reading - standard NAOH solution…
A: Vinegar is a dilute solution of acetic acid. It is used as salad dressing and preservative. The data…
Q: Molarity of NaOH (phenophthalein) 0.1132 Volume of HC2H3O2 (acetic acid) 25.00 ml. vinegar %3D TRIAL…
A: Given data,Molarity of NaOH=0.1132MVolume of acetic acid=25.0mL=0.025L
Q: Procedure Part 1: Obtain mass of a liquid in a beaker using weighing by difference technique. 1.…
A: Given : For trial 1 : Empty Beaker = 19.2 g Full Beaker = 35.4 g . For trial…
Q: Table 1: 田 Average [NaOH) (M) Volume NaoH [NaOH) (M) Trial # KHP (g) (ml) 0.4510 22.16 0.4524 22.21…
A: The table gives mass of KHP (potassium hydrogen phthalate) and different volume of NaOH. To find:…
Q: Table 5.1. Determination of the purity of KHP sample. Parameter Trial 1 Trial 2 weight of container…
A:
Q: Dosage Form Active Raw Materials Actual Yield Formulation Packaging Ingredient 2.6 kgs paracetamol…
A: In this question, balanced chemical equation of each item is to be written.
Q: Solve for the Number of moles of the unknown with the given data. I do not no the given substance
A: Mass of the unknown, m = 0.5940 gPartial pressure of CO2 , P= 719.4 mm Hg = 719.4 / 760 = 0.9465…
Q: Concentration iron(III) nitrate [Fe(NO3)31 (M) 0.35000 Color of iron(III) nitrate yellow…
A: Given, Concentration of iron (III) thiocyanate ion = 0.000250 M
Q: Enter an equality for the following medication in stock: 1.0 g of Cefuroxime per 1 mL of Cefuroxime…
A:
Q: 4. Below left is a cylinder containing water. An object with a mass of 21g and a volume of 15 cm³ is…
A: “Since you have asked multiple question, we will solve the first question [Q4] for you. If you want…
Q: one Trial two KIO3 solution taken (mL) 1.70 16.5 Moles of KIO3 1.06 x 10^-3 1.06 x 10^-3…
A: We have find out solubility and solubility product of Ca(Io3)2 for first and second filtrate.
Q: calculate the volumetric analysis for Nitrogen in % given the ff Constituent Percentage Oxygen…
A:
Q: Procedure: Determine the correct weight of the empty graduated cylinder (measuring device). Fill…
A: Determine the correct weight of the empty graduated cylinder (measuring device). Fill up the…
Q: Sample Blank $2 $3 $4 $5 $6 $7 $8 $9 $10 Stock of Fe Stock of SC Volume Fe3+, mL 2.000 2.000 2.000…
A: From the given data we have Concentration of [FeNCS2+]=[SCN-] Basing on the given chart we have to…
Q: B) Analysis of unknown aspirin tablet 1.Mass of aspirin tablet= 2.Concentration of ASA unknown from…
A: First, we have to draw the absorbance versus concentration(M) plot. From the linear regression…
Q: 93.6g to ng and 93.6g to tons with the set up
A: Given : To convert 1) 93.6 g to ng i.e nanograms and 2) 93.6 g to tons.
Q: Fill the table and create a publication quality graph of density vs % mass for solutions 5. Fit the…
A: Use the excel to draw the trendline graph- write the value of x and y axis select them , choose…
Q: The gravimetric factor used to express CoCBr6·H20 in a sample that is finally weighed as PbClBr is…
A: Gravimetric factor
Q: gar sample 8.2000 g 8.1000 g
A: Density is defined as mass per volume It is the physical property of the matter different matter…
Q: Calculatian OF Fez Do. lucing02: . g-02 x. Conversion Factor 1 -X. Conversfon Factor 7 Corversion…
A: Mole relation gives the relationship among moles, molar mass, atomic mass, particles.…
Q: Calculate the suspended solids in mg/L given the following data: Weight of crucible: 21.0256 g,…
A: Given, Weight of crucible: 21.0256 g Weight of crucible and dry solids: 21.0301 g Weight of…
Q: 1-10. The table shows fuel efficiency for several automobiles CO2 emission (g CO2/km) Fuel…
A: (a)The number of miles in 1 km is 0.621371miles.
Q: aye = 5.0 x 10 2 Zna+ = 1.17 x 10-5 Znya-= 3.0 x 1016
A:
Q: Information Name: BAUTISTA Unknown Acid Volume: 12.150 mt 10.0M NƏOH IO00 ml te 25.0°C Temperature:…
A: Volume of unknown acid = 12.150 mL Molarity of NaOH =10.0 M Volume of NaOH used= 10 mL
Q: Conc. of NaOH: TRIAL 1 TRIAL 2 TRIAL 3 Mass of flask 81.061 g 69.692 g 96.039 g Mass of flask and…
A: We are given the table in the question: By checking the values from it we will find the required…
Q: An aqueous solution of sodium hydroxide contains 20.0% NaOH by mass. It is desired to produce an…
A:
Q: Some experts says that km can be calculated by saying Vmax from date divided by 2 and then they find…
A: We can calculate the approximate value of Km both from the graph and by solving the MM equation.
Q: Please give the full calculation + explanation for each step.
A: The formula for percent by mass or mass percent is given as :
Q: Sarah bought a neti pot from©. After using up the salt packets she decided to make one from scratch…
A: Molarity is the term used to express concentration. Molarity is defined as the number of moles of…
Q: Balanced Chemical Equation MnO4- +5Fe2+ +8H+ --> Mn2+ +5Fe3+ +4H2O Details mass (g) sample average…
A: The balanced redox titration reaction is: MnO4-(aq) + 5Fe2+(aq) + 8H+(aq) →Mn2+(aq) + 5Fe3+(aq) +…
Q: Calculate the Standardized Molarity of NaOH via dimensional analysis. Show your process. Use this…
A:
Q: Seconds (s) Tap H2O Temp (°C) Salt Solution (+ 2.5 mL salt) Temp (°C) Salt…
A: Freezing point temperature is the temperature when liquid phase and solid phase of the substance…
Q: Balancing Exit Ticket Fill in each blank with the appropriate coefficient. C + v H2S Au2S3 H2--> v…
A: A chemical reaction equation which contains same number of atoms on both reactant and product side…
Q: For the given drying process, a material (Called SRU) is dried and feed and outputs are as shown in…
A: The process flow diagram given is,
Q: Date lab performed: Name: Date due: Partner(s) ame Measurements: Density of a Sallne Solution Data:…
A: 10 mL graduated cylinder is most accurate and 100 mL beaker is least accurate. In the 10 mL…
Q: A tank with a capacity of 74 liters, initially contains 20 kilograms of salt dissolved in 50 liters…
A: Given, Total capacity - 74 L Initially - 20 kg salt in 50 L of water. Final addition - (74 - 50) =…
Q: In the distillation of the mixture of acetic acid (MW=60.05 g/mole; d=1.05g/ml) and water (MW= 18.00…
A: Density of acetic acid = 1.05 g/ml Density of water = 1.00 gm/ml Molarity of NaOH = 1.00 M Volume…
Q: A. Standardization of NaOH solution with KHP Trial 1 Trial 2 Weight of KHC,H,o. 1.596 g 1.819 g…
A:
Q: Need help with question #24 Results Trial 1 : Initial Volume = 0 0.5143g=KHP 14.3 = Buret…
A: The stoichiometric reaction of KHP with NaOH is: NaOH + C8H5O4K → C8H4O4KNa + H2O…
Q: Trial 1 Trial 2 1-Mass of empty evaporating dish 55.312 g 55.315 g 56.619 g 56.018 g 56.448 g 2-…
A: The mass of MgSO4 can be calculated by subtracting the mass of empty evaporating dish from the mass…
Q: Name: Date lab performed: Partner(s) name: Date due: Measurements: Density of a Salime Solutiom…
A: Density = mass/ volume
Q: Coming homo after the end of the winter semostor, you find that your family's swimming pool has not…
A: Solution - According to the question - Given - take whole swimming pool as a control volume. given…
Q: Part B (Trial #1) Part C (Trial #2) Mass of empty evaporation dish 154.48 g 158.34 g Volume of…
A:
Q: Analysis of product: Calculate the percent recovery based on the amount of tea originally used and…
A:
Q: maining in cylinder, (mL) 1.09 Volume of NaCl solution transferred to dish, (mL) Mass of dish,…
A:
Q: Trial-1 Volume of NaOH in Liters. Moles of NaOH (MxV) Moles of CH,COOH Molar Mass Of CH,COOH (g/mol)…
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question…
Q: Complete the following table: Subst. weighed Subst. sought Grav. Factor 1.AgCI NaCl 2.Baso, SO3…
A: The gravimetric factor is the ratio between the grams of a compound and the grams of a single…
Q: 50.0 g solute in 0.425 MK2CO3 Express your answer with the appropriate units. HÁ V = Value Units…
A: Concentration of K2CO3=0.425M Mass of K2CO3 =50.0g
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- Calculate the Constant Weight (in grams) of the Empty Crucibles. Show your solution. Crucible No. 1 2 3 Weighing 1 22.6035 22.0223 24.1535 Weighing 2 22.6017 22.0204 24.1533 Weighing 3 22.5994 22.0199 - Weighing 4 22.5992 - - Constant Weight, g ??? ??? ???The gravimetric factor used to express CoCBr6·H20 in a sample that is finally weighed as PbClBr is choose below: FW PbClBr / 6 x FW CoCBr6·H20 FW CoCBr6·H20 / FW PbClBr FW CoCBr6·H20 / 6 x FW PbClBr 6 x FW PbClBr / FW CoCBr6·H20Express 20,000ppb as a percentage strength A. 2% B. 0.02% C. 0.00002% D. 0.002%
- There's 1 drink (and you are asked to determine the glucose concentration in the drink in the units of g/100mL. (Why these units? Well, once you have the concentrations in g/100mL you will be able to compare your values with the nutritional values given on the drink bottles’ labels). The sample of the drink was diluted 1/100 (i.e. by a factor of 100). This was an essential step in the method because, without it, the machine used to analyse the glucose concentration (spectrophotometer) would have given an error as the concentration would have been too high for accurate detection. What this means for you is that the dilution factor will need to be taken into consideration in your calculations (remember the aim is to calculate the concentration in the original drink and not in the diluted drink). You measured the concentration of their diluted drink using the spectrophotometer and their results were provided to them in the units mM (millimolar). Glucose Concentration in mM of drink =…There's 1 drink (and you are asked to determine the glucose concentration in the drink in the units of g/100mL. (Why these units? Well, once you have the concentrations in g/100mL you will be able to compare your values with the nutritional values given on the drink bottles’ labels). The sample of the drink was diluted 1/100 (i.e. by a factor of 100). This was an essential step in the method because, without it, the machine used to analyse the glucose concentration (spectrophotometer) would have given an error as the concentration would have been too high for accurate detection. What this means for you is that the dilution factor will need to be taken into consideration in your calculations (remember the aim is to calculate the concentration in the original drink and not in the diluted drink). You measured the concentration of their diluted drink using the spectrophotometer and their results were provided to them in the units mM (millimolar). Glucose Concentration in mM of drink =…If 35,000 kg of whole milk containing 4% fat is to be separated in a 6-hour period into skim milk with 0.45% fat and cream with 45% fat, what are the mass flow rates of the two output streams from a continuous centrifuge which accomplishes this separation? (Ans; Cream=464.8335kg/h, Skim milk= 5368.4998kg/h)
- 1. Calculate the experimental density of a salt solution and the percent error (same as relative error percent) using some or all the data given below. solubility of NaCl salt in water: 0.357 g/mLmass of empty graduated cylinder: 25.19g mass of graduated cylinder + salt solution: 30.47g total volume of salt solution: 4.98 mLtrue density of salt solution: 1.07 g/mLSum of coefficients C7H8 + O2 --> CO2 + H2O after balancingA student obtained 2.111 g of a mixture containing four components. After performing separation by chemical and physical methods, the student obtained the following mass data: Component 1: 0.486 g Component 2: 0.317 g Component 3: 0.127 g Component 4: 0.929 g Calculate the total percent recovery of the mixture. (Please show work)
- An orange juice processing plant now produces essential oil from orange peels. one It is known that 250 kg of peel comes out of 1 ton of oranges and 2.5 g of essential oil comes out of 1 kg of peel. In a laboratory study, 250 g of bark was treated with hexane solvent and 0.548 g of essential oil was obtained in the sample cup of the rotary evaporator. Accordingly, the rotary Calculate the separation efficiency obtained in the evaporator?(6.65x10^5)/(6.60x10^-6) (3.73x10^8) Please Calaculition. Only typed solutionUsing the percent purity calculations, determine the percent yield of synthesis of aspirin. Part I Synthesis of Aspirin Mass of salicylic acid used (g) 2.029g Volume of acetic anhydride used (mL) 5ml Mass of acetic anhydride used (vol. × 1.08 g/mL) 5.4g Mass of aspirin synthesized (g) 3.256g Part II Melting Temperature Data Melting temperature (°C) 133°C Part III Salicylic Acid Standard Stock Solution Initial mass of salicylic acid (g) 0.210g Moles of salicylic acid (mol) 0.0147 mol Initial molarity of salicylic acid (M) 0.724 M Part III Beer’s Law Data for Salicylic Acid Standard Solutions Trial Concentration (M) Absorbance Water (mL) 1 10 0.301 0 2 7.5 0.219 2.5 3 5.0 0.163 5.0 4 2.5 0.074 7.5 Best-fit line equation for the salicylic acid standards Test of the Purity of the Synthesized Aspirin Initial mass of aliquot of product (g)…

