Chapter26: Biomolecules: Amino Acids, Peptides, And Proteins
Section26.SE: Something Extra
Problem 28MP
Related questions
Question
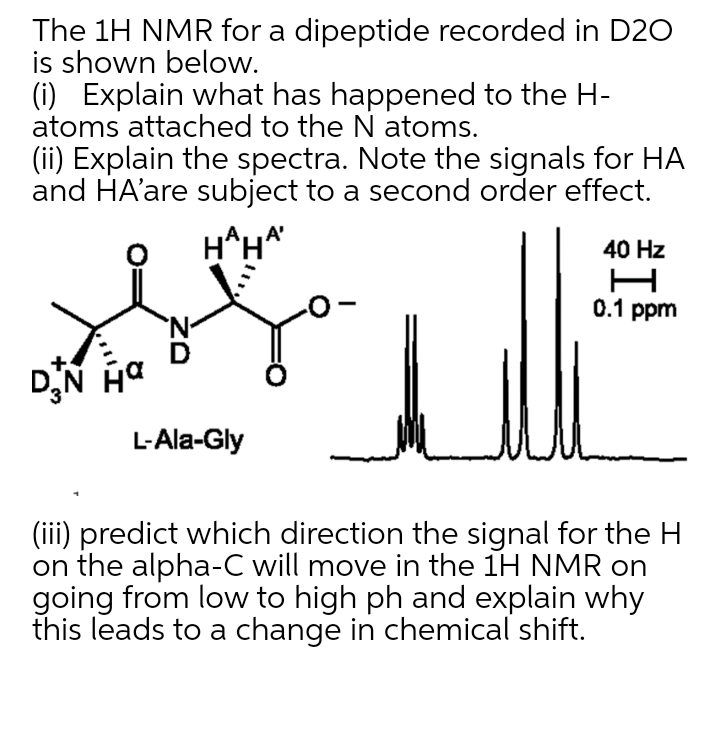
Transcribed Image Text:The 1H NMR for a dipeptide recorded in D20
is shown below.
(i) Explain what has happened to the H-
atoms attached to the N atoms.
(ii) Explain the spectra. Note the signals for HA
and HA'are subject to a second order effect.
H^H*
40 Hz
0.1 ppm
'N-
DN Ha
L-Ala-Gly
(iii) predict which direction the signal for the H
on the alpha-C will move in the 1H NMR on
going from low to high ph and explain why
this leads to a change in chemical shift.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



