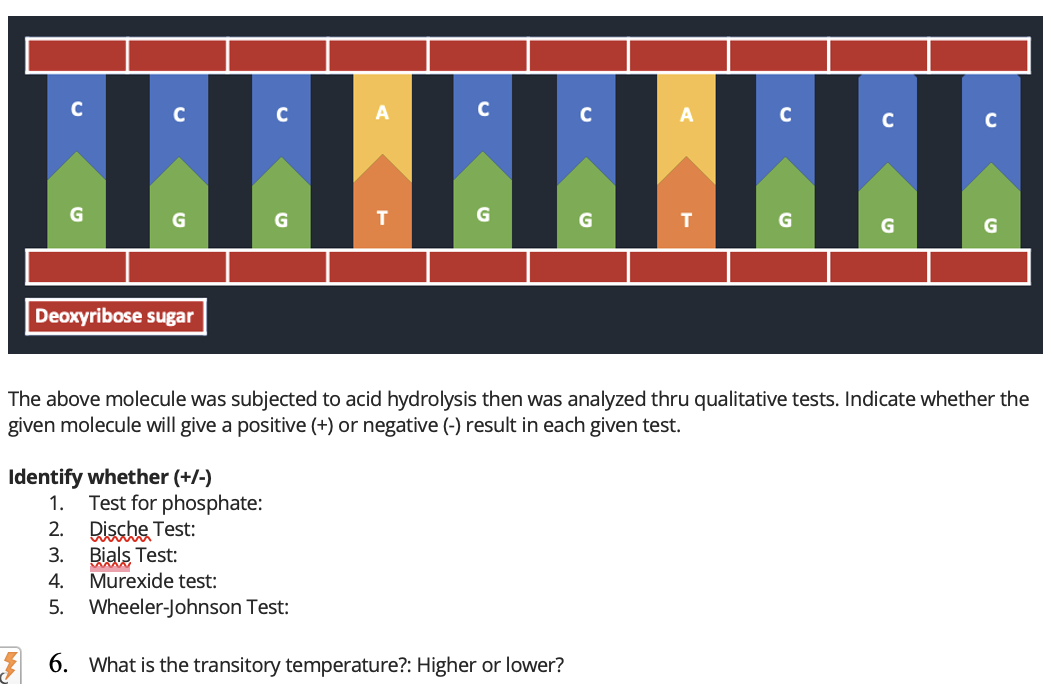
The above molecule was subjected to acid hydrolysis then was analyzed thru qualitative tests. Indicate whether the given molecule will give a positive (+) or negative (-) result in each given test. Identify whether (+/-) Test for phosphate: Dische Test: Bials Test: Murexide test: Wheeler-Johnson Test: 6. What is the transitory temperature?: Higher or lower?
Q: The reaction in Fohl’s test will cause the formation of what compound? What type of reaction happens…
A: Fohl's test is the biochemical test of proteins for the detection of presence of Aminoacids with…
Q: A 100 mL solution of 0.1 M amino acid (AA) at pH 1.0 was titrated with 1.0 M NaOH solution. The pH…
A: Amino acids are composed of a central chiral carbon, hydrogen, an alpha-carboxylic group, an…
Q: Give the color of the water mixed with Lugol's iodine solution in these situations before exposure…
A: LUGOL'S is an aqueous solution containing Iodine 5%, and potassium iodide 10% w/v solution. Lugol's…
Q: The table below summarizes the results for Millon's test. Provide the correct remarks from the…
A: Millon’s test is an analytical test used for the detection of the hydroxy phenol groups. Since the…
Q: Identify HCl. strong electrolyte, weak acid nonelectrolyte weak electrolyte, weak acid weak…
A: Introduction: Those molecules that convey an electric charge when dissolved in liquid are…
Q: DIFFERENCE AMYLOSE AMYLOPECTIN Structure Mol Size Degree of branching IODINE TEST…
A: Polysaccharide is of two types homo and heteropolysaccharides. Homopolysaccharides have a…
Q: A carbohydrate sample reacts positively with all test reagents except for Lugol's. The sample could…
A: The sample could be possibly Sucrose.
Q: Identify what test is being described: Test for the presence of indole group ? Hydrolysis…
A: Indole is an aromatic heterocyclic organic compound which has a bicyclic structure that consists of…
Q: Explain the chemical basis (reaction) of Millon’s test. (Generic reaction and explanation) What is…
A: Proteins are made up of amino acids joined together by peptide bond. Proteins can be estimated by…
Q: What analyte gives a positive result in Molisch Test for Carbohydrates
A: Molisch test is the specific test done to detect the presence of carbohydrates in the solution. It…
Q: Barium sulfate is often used as a medical diagnostic tracer, however, it can only be administered…
A: Barium sulfate is white or yellowish odorless powder or crystals. The Melting point is 1580°C and a…
Q: An amino acid mixture of phenylalanine, glycine and glutamic acid is to be separated by paper…
A: Paper chromatography is the analytical process by which mixtures of complex compounds such as amino…
Q: The test for starch in water is A. silver nitrate. B. iodine. C. barium ions. D.…
A: Starch is a homopolysaccharide molecule that was composed of alpha-D-glucose units in 1-4 and 1-6…
Q: What will you observe when samples of these fats and oils are tested for unsaturation with bromine…
A: Fatty acids are building blocks of fats. Fats are one of the major biomolecules. Fatty acids can be…
Q: Provide the Principles and detailed procedures of the following tests for the "Color Reaction of…
A: Hopkins - cole test Principle -In this test , concentrated H2So4 cause dehydration of tryptophan…
Q: MATCHING TYPE; Match A to B A. Acrolein tesT Benedicts test…
A: biochemical test : the test that are performed on samples of serum, solution, plasma , urine where…
Q: Give and explain the principles of the following tests for lipids: Spot Test Acrolein Test Test for…
A: Your question has multiple subparts. I will answer 1st three of the subparts, as per the guidelines.…
Q: Which of the following below will show a NEGATIVE result in Huble’s test? Group of answer choices…
A: Lipids are carboxylic groups is attached to a long hydrocarbon chain. On the basis of the presence…
Q: What are the possible interferences or complications in detecting and differentiating aldehydes and…
A: Before scientists developed tools such as x-ray crystallography and other spectrophotometric…
Q: A. Biuret Test Mix 1 mL of sample with 1 mL of 5% NaOH. Add 2 – 3 drops of 1% CuSO4. Question…
A: Biuret test is done to check for the presence for proteins. CuSO4 and NaOH are used as reagents.…
Q: When conducting a flame test for sodium iodide, is the result positive or negative?
A: An ionic compound is one that is formed between a metal or metalloid and a non-metal. A Flame test…
Q: Qualitative and Quantitative Tests for Amino Acids and Proteins Complete the table: Test Reagents…
A: Introduction: Amino acids are biomolecules containing an amino and a carboxyl group with a side…
Q: You receive an order for 750 mL of 27% dextrose solution. You have available D50W, 1000 mL. How many…
A: Given, D50W means 50% dextrose solution, 100 ml 750 ml of 27% dextrose is desired Sterile water
Q: Which of the following depicts the mild alkalosis? Select one: a. pH=7.33 Ob. pH=7.45 O c. H=7.65…
A: The collecting duct of kidney participates in reabsorption and secretion of selective ions into…
Q: Give at least two (2) chemical tests for amino acids/ proteins other than what were performed in…
A: There are several other tests to determine properties of proteins and amino acids present in a…
Q: 1. B,010H22 (aq) 2. HB,010H4 (aq) 3. H2B4010H4(aq) Using the above chemicals to match the below…
A: Asked : Chemicals to be matched with given terms
Q: REAGENTS. Match the name of the reagent with the physiological test. Tests may require more than one…
A: A reagent is a substance added to a system to cause a chemical reaction or test if a reaction…
Q: What is the fluid thioglycollate medium? Please explain the testing and its results and the…
A: Microbiology laboratory uses various kinds of culture media to identify the biochemical…
Q: Mention at least two simple chemical tests by which you can distinguish between benzaldehyde and…
A: Benzaldehyde will respond to the Tollen's reagent where as benzoic acid will not respond to it.
Q: 100 mL of 2.5 X NaOH solution from 50X NaOH solution
A: Given Values: Stock solution concentration (NaOH) = 50 X Final Solution concentration (NaOH) = 2.5 X…
Q: Make the following prescription isotonic with lacrimal fluid: 0.06 g 0.30 g Ephedrine sulfate (E =…
A: A drug, often known as a medicine, is a chemical compound that has a biological effect on the human…
Q: Ninhydrin test for amino acids or protein requires that sample test solution should be at initially…
A: Proteins are one of the essential macronutrients for a human body. The building blocks of proteins…
Q: Complete the table below regarding the different laboratory tests done for Carbohydrates. Only for…
A: There are different types of tests to check the presence of carbohydrates in different chemical…
Q: Will the given lipid test positive in the Ninhydrin test?
A: Ninhydrin test is used to check whether a given sample contains amines or alpha-amino acids. The…
Q: Name at least two alternative chemical tests that can be performed to characterize functional…
A: Organic compounds usually can be divided into two parts: a reactive part; functional group and alkyl…
Q: Arrange the following in order of increasing Rf on thin-layer chromatography: acetic acid,…
A: Thin-layer chromatography is a method of separating non-volatile mixtures. Thin-layer chromatography…
Q: Finally, draw the standard curve for BSA. Reagents Blank Tube Tube Tube 2 3 Tube Tube Tube Tube 5 4…
A: BSA is the bovine serum albumin that is a protein concentration used as a reference in the protein…
Q: need to dissolve cholesterol in Chloroform in the Liebermann-Burchard Test
A: The reaction of acetic anhydride in chloroform with unsaturated steroids (e.g. cholesterol) in the…
Q: If 10g of salt was mixed in 1L water. What percent solution will this be.
A: According to the question,Mass of solute = 10 grams = 10gVolume of water = 1L = 1000 mili Liters…
Q: If a patient is prescribed 300 mg of potassium chloride, what is the corresponding milliequivalent?
A: Drug calculation is a important aspect in calculating the dose based on the BMI ,BSA ,age and body…
Q: Given ß- Cyclodextrin Briefly explain its expected reaction (based on their structural formula) to…
A: Cyclodextrin is a class of oligosaccharide that contains a macrocyclic ring of glucose subunits…
Q: How would I calculate the amount of each component in each solution? Stock solutions provided:…
A: In-order to perform dilution, we take a specific volume from stock solution and transfer it to…
Q: Match the qualitative test results to the sample identity in the table below. Give its structure and…
A: Biomolecules are organic compounds produced by living organisms. They play important function as…
Q: If a pharmacist added each of the following to 22-g packages of 2% mupirocin ointment, what would be…
A: Mupirocin is an antibiotic that prevents bacteria from growing on your skin. Mupirocin topical is…
Q: FREE RESIDUAL CHLORINE IS 1. Chloramide compounds 2. hypochlorous or chloric acid 3. haloform…
A: Introduction :- The quantity of chlorine that remains in the water after a given period or contact…
Q: In excess of alkaloidal reagent, did the precipitate formed dissolve or not? What is the evidence…
A: Alkaloid reagents such as tannic acid, potassium iodide, potassium ferrocyanide, or mercuric iodide…
Q: Discuss the results and observations in a regular laboratory experiments the denaturation of: 1.…
A: Denaturation is the process, in which the protein loses its functions due to the change in the…
Q: What is the role of indicator in determining if the substance is acid or base? (Note that the…
A: pH indicators are compounds which are natural dye. Those indicate if the solution is acidic or basic…
Q: Standardization of Hydrochloric Acid Solution Answer the concentrations in 3 significant figure. The…
A: n1M1V1= n2M2V2 n1=2 for Na2CO3, n2=1 for HCl M1 is molarity of Na2CO3 V1 is volume of Na2CO3 M2 is…
Q: What is the iodine indicator test used for? Describe in detail how iodine works in a reaction and…
A: Iodine indicator test is used for detection of starch. The triiodide form of iodine complexes with…
The above molecule was subjected to acid hydrolysis then was analyzed thru qualitative tests. Indicate whether the given molecule will give a positive (+) or negative (-) result in each given test.
Identify whether (+/-)
- Test for phosphate:
- Dische Test:
- Bials Test:
- Murexide test:
- Wheeler-Johnson Test:
6. What is the transitory temperature?: Higher or lower?
Step by step
Solved in 2 steps with 2 images

- Give at least two (2) chemical tests for amino acids/ proteins other than what were performed in this experiment. For each test, cite the respective application and describe the positive result. a.Ninhydrin Test b.Biuret’s Test c.Xanthoproteic Test d.Millon’s Test e.Sakaguchi test f.Hopkins –Cole Test g.Sulfur Reaction or Lead Acetate TestGive at least two (2) chemical tests for amino acids/ proteins other than what were performed in this experiment. For each test, cite the respective application and describe the positive result. test that have been performed a.Ninhydrin Test b.Biuret’s Test c.Xanthoproteic Test d.Millon’s Test e.Sakaguchi test f.Hopkins –Cole Test g.Sulfur Reaction or Lead Acetate TestLipids from an organic sample are extracted separately by column chromatography using the ff eluants: 1st eluant - chloroform: methanol: water (6:3:1) 2nd eluant - methanol: chloroform (9:1) 3rd eluant – petroleum ether: ethyl ether (1:1) Test results for each eluate are tabulated below. Identify the possible lipid present in each eluate. Test Reagents 1. KHSO4 2. HNO3,(NH4)3MoO4 3. CHCl3, acetic anhydride, conc. H2SO4 4. -naphthol, conc. H2SO4 5. Hydroxylamine HCl, FeCl3 6. ninhydrin reagent POSSIBLE LIPIDS: Triacylglycerol, cholesterol, sphingomyelin, lecithin
- Based on this video https://www.youtube.com/watch?v=rKng5-ij6kQ Provide a schematic diagram for the Iodine test methodologies in determining the presence of carbohydrates. Also, give the basic principle for the test.Given ß- Cyclodextrin Briefly explain its expected reaction (based on their structural formula) to the different qualitative tests for carbohydrates which are: (a) Molisch’s test (b)Fehling’s test (c) Bial’s testComplete the table below regarding the different laboratory tests done for Amino Acids and Proteins Tests Procedure (Chemical Reagents Added) Positive Results/ Observations (ex: color of solution, precipitate formation, etc) Positive for (provide details like functional group responsible) Molisch Test Benedict’s Test Barfoed’s Test Moore’s Test Bial’s Test Seliwanoff’s Test Osazone Test Fehling’s Test Tollen’s Test Mucic Acid Test Lugol’s Test/ Iodine Test Hydrolysis w/ Benedict’s Test Hydrolysis w/ Iodine Test Provide the expected results with the samples given below: For negative results just leave it blank, for positive results write “(+) then the expected observation” ex: + blue solution Samples Molisch Test Benedict’s Test Barfoed’s Test Moore’s Test Bial’s Test Seliwanoff’s Test Glucose Galactose Fructose Ribose…
- REAGENTS. Match the name of the reagent with the physiological test. Tests may require more than one answer. a. alpha-naphthol b. dimethyl-alpha-naphthylamine c. ferric chloride d. Gram’s iodine e. hydrogen peroxide f. Kovac’s reagent g. methyl red h. N,N,N′,N′-tetramethyl-p-phenylenediamine dihydrochloride i. potassium hydroxide (KOH) j. sulfanilic acid 2,3-butanediol fermentation catalase test mixed-acid fermentation nitrate reduction oxidase test phenylalanine deamination starch hydrolysis tryptophan degradationYour supervisor asks to make 350 mL of a 0.75% (w/v) starch solution using a 5% (w/v) stock solution. You have calcuated how much stock solution you need. But, how much water (in mL) will you need to add to the stock to achieve the correct final concentration?Qualitative and Quantitative Tests for Amino Acids and ProteinsComplete the table: Test Reagents Positive Result Positive Indication Ninhydrin test Xanthoproteic acid test Millon's test Hopkins Cole test Pauly's diazo Test Lead sulphide test Histidine test Isatin test Folin’s McCarthy Sullivan Test Nitroprusside Test
- MATCHING TYPE; Match A to B A. Acrolein tesT Benedicts test Biuret test Iodine test Barfoed’s test Molisch test Seliwanoff’s test Fehling’s test Salkowski’s test Furter-Meyer test B. A. The test for the presence of cholesterol. B. The test for reducing sugars in acidic solution. C. The test is used to detect cholesterol in a solution D. The test for the presence of starch/amylose. E. The test for presence of reducing sugars in alkali solution. F. The general test for carbohydrates. G. This method is used to detect the presence of tocopherols H. The test to distinguish monosaccharides from disaccharides I. The test for the presence of fats or glycerin. J. The test to determine ketohexose from an aldohexose.Can the iodine test distinguish between amylose and amylopectin? What would you look for?What are the possible interferences or complications in detecting and differentiating aldehydes and ketones using the following tests: 2,4-dinitrophenyl hydrazine test Benedict’s test Tollen’s test Jones’ test Iodoform test pls answer them all thank you!

