The addition of dimethylglyoxime, H2C4H602N2, to a solution containing nickel(II) ion gives rise to a precipitate: Nickel dimethylglyoxime is a bulky precipitate that is inconvenient to manipulate in amounts greater than 135 mg. The amount of nickel in a type of permanent-magnet alloy ranges between 22% and 38%. Calculate the sample size that should not be exceeded when analyzing these alloys for nickel.
The addition of dimethylglyoxime, H2C4H602N2, to a solution containing nickel(II) ion gives rise to a precipitate: Nickel dimethylglyoxime is a bulky precipitate that is inconvenient to manipulate in amounts greater than 135 mg. The amount of nickel in a type of permanent-magnet alloy ranges between 22% and 38%. Calculate the sample size that should not be exceeded when analyzing these alloys for nickel.
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter23: Polymeric Materials And Soft Condensed Matter
Section: Chapter Questions
Problem 14P
Related questions
Question
Please show complete solution
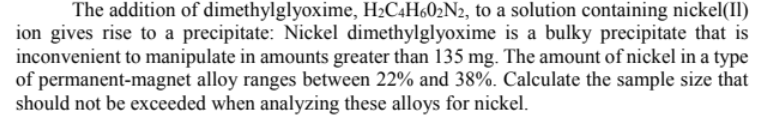
Transcribed Image Text:The addition of dimethylglyoxime, H2C4H602N2, to a solution containing nickel(II)
ion gives rise to a precipitate: Nickel dimethylglyoxime is a bulky precipitate that is
inconvenient to manipulate in amounts greater than 135 mg. The amount of nickel in a type
of permanent-magnet alloy ranges between 22% and 38%. Calculate the sample size that
should not be exceeded when analyzing these alloys for nickel.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,