the electrons in the following listed, ir tney were listed Increasing elec ostatic potential Thát is, select the electron with the lowest electrostatic potential energy, select "2" next to the electron with the second lowest electrostatic potential energy, and so on. electrostatic electron location potential energy A 5.7 nm from an anion with a charge of -2 (choose one) e (choose one)e alo в 6.8 nm from an anion with a charge of -1 5.7 nm from an anion with a charge of -1 (choose one) e D 4.3 nm from an anion with a charge of -2 (choose one) 8 Explanation Check O 2021 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility
the electrons in the following listed, ir tney were listed Increasing elec ostatic potential Thát is, select the electron with the lowest electrostatic potential energy, select "2" next to the electron with the second lowest electrostatic potential energy, and so on. electrostatic electron location potential energy A 5.7 nm from an anion with a charge of -2 (choose one) e (choose one)e alo в 6.8 nm from an anion with a charge of -1 5.7 nm from an anion with a charge of -1 (choose one) e D 4.3 nm from an anion with a charge of -2 (choose one) 8 Explanation Check O 2021 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter3: Atomic Shells And Classical Models Of Chemical Bonding
Section: Chapter Questions
Problem 7P
Related questions
Question
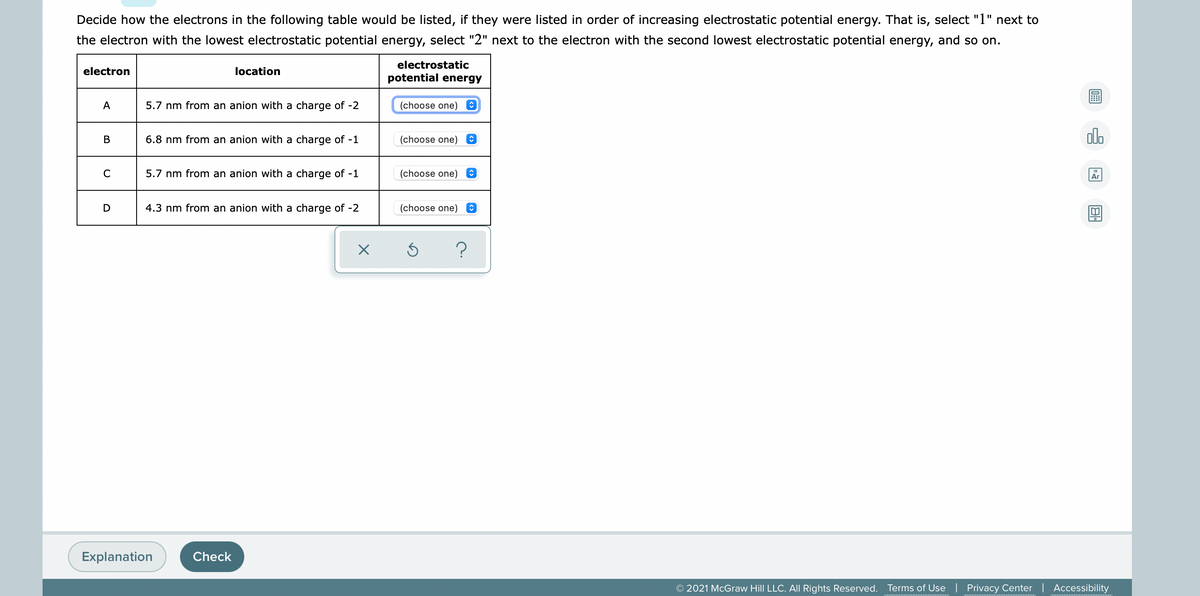
Transcribed Image Text:Decide how the electrons in the following table would be listed, if they were listed in order of increasing electrostatic potential energy. That is, select "1" next to
the electron with the lowest electrostatic potential energy, select "2" next to the electron with the second lowest electrostatic potential energy, and so on.
electrostatic
electron
location
potential energy
A
5.7 nm from an anion with a charge of -2
(choose one) O
olo
В
6.8 nm from an anion with a charge of -1
(choose one)
C
5.7 nm from an anion with a charge of -1
(choose one)
18
Ar
D
4.3 nm from an anion with a charge of -2
(choose one)
Explanation
Check
O 2021 McGraw Hill LLC. All Rights Reserved.
Terms of Use | Privacy Center | Accessibility
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning