Use the solubility graph to answer the following question. Which of the listed salts is the most soluble at 20 °C? Solubility (g/L of solvent) OKCI NaCl OKNO3 O KI NH4CI 1400 1300 1200 1100 1000 900 800 700 600 500 400 300 200 100 0 Solubility Curves ZKI -HCI SO₂ NaNO₂ -KNO3 NHẠCH KCI -KCIO3 NaCl NH₂ 10 20 30 40 50 60 70 80 90 100 Temperature (°C)
Use the solubility graph to answer the following question. Which of the listed salts is the most soluble at 20 °C? Solubility (g/L of solvent) OKCI NaCl OKNO3 O KI NH4CI 1400 1300 1200 1100 1000 900 800 700 600 500 400 300 200 100 0 Solubility Curves ZKI -HCI SO₂ NaNO₂ -KNO3 NHẠCH KCI -KCIO3 NaCl NH₂ 10 20 30 40 50 60 70 80 90 100 Temperature (°C)
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter16: Solutions
Section: Chapter Questions
Problem 11E
Related questions
Question
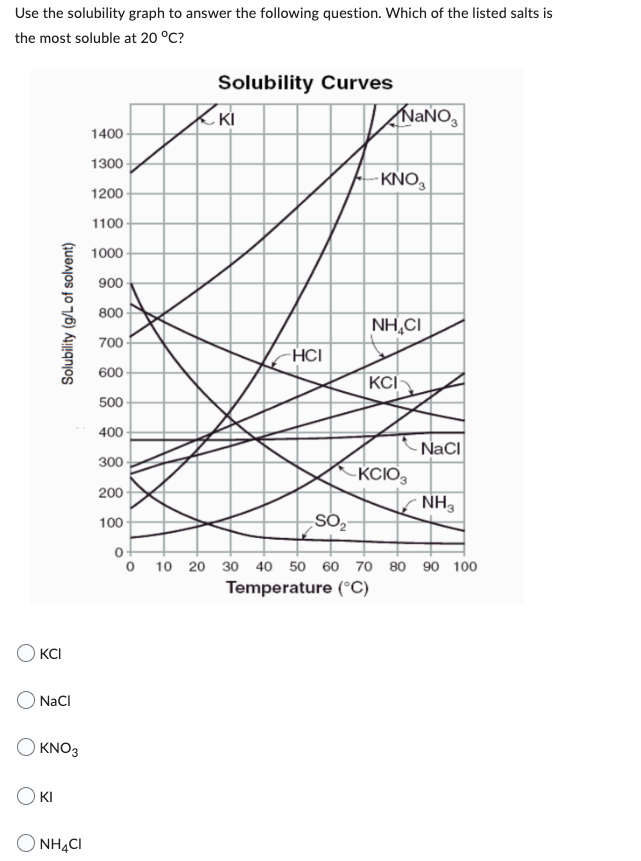
Transcribed Image Text:Use the solubility graph to answer the following question. Which of the listed salts is
the most soluble at 20 °C?
Solubility (g/L of solvent)
OKCI
NaCl
OKNO3
O KI
NH4CI
1400
1300
1200
1100
1000
900
800
700
600
500
400
300
200
100
0
Solubility Curves
ZKI
-HCI
SO₂
NaNO3
-KNO3
NHẠCH
KCI
-KCIO3
NaCl
NH₂
10 20 30 40 50 60 70 80 90 100
Temperature (°C)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning


General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning


General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax
