Using the Chromatographs containing the unknown mixture of Acetates and the corresponding relative response factors from the standard mixture, calculate the mole percentage of Peak 2 in the unknown mixture.
Using the Chromatographs containing the unknown mixture of Acetates and the corresponding relative response factors from the standard mixture, calculate the mole percentage of Peak 2 in the unknown mixture.
Chapter4: Least-squares And Calibration Methods
Section: Chapter Questions
Problem 3P
Related questions
Question
Using the Chromatographs containing the unknown mixture of Acetates and the corresponding relative response factors from the standard mixture, calculate the mole percentage of Peak 2 in the unknown mixture.
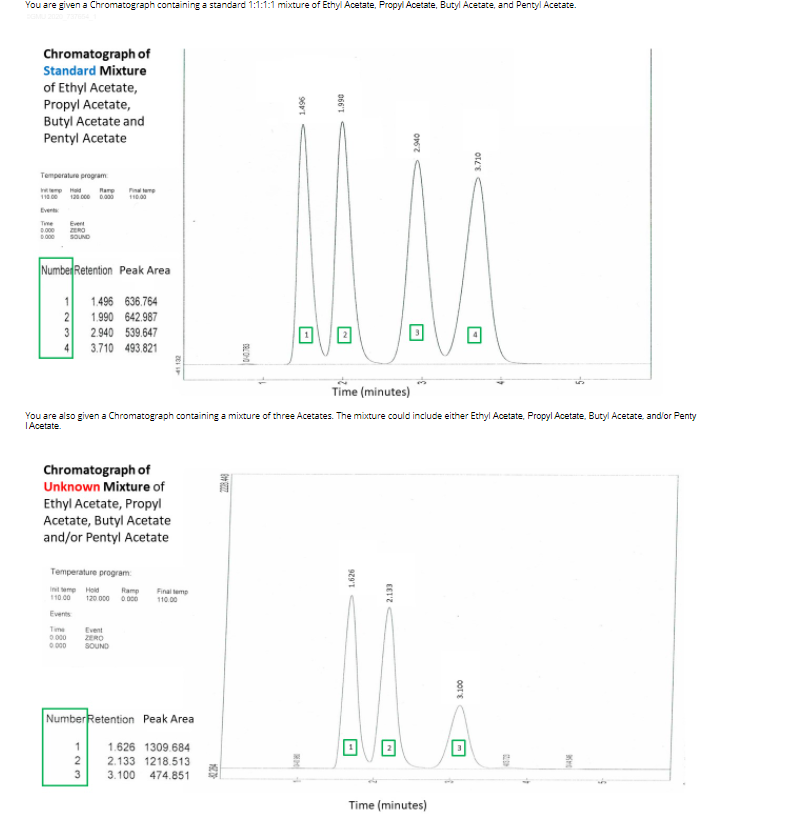
Transcribed Image Text:You are given a Chromatograph containing a standard 1:1:1:1 mixture of Ethyl Acetate, Propyl Acetate, Butyl Acetate, and Pentyl Acetate.
Chromatograph of
Standard Mixture
of Ethyl Acetate,
Propyl Acetate,
Butyl Acetate and
Pentyl Acetate
Temperature program
Rare tem
1100 120 cce 0.000 1D0
Evert
Tme
Evert
D.000 ERO
SOUND
Number Retention Peak Area
1.496 636.764
1.990 642.987
2.940 539.647
3.710 493.821
Time (minutes)
You are also given a Chromatograph containing a mixture of three Acetates. The mixture could include either Ethyl Acetate, Propyl Acetate, Butyl Acetate, and'or Penty
TAcetate.
Chromatograph of
Unknown Mixture of
Ethyl Acetate, Propyl
Acetate, Butyl Acetate
and/or Pentyl Acetate
Temperature program
Int semp Hod
110.00 120 000 o 000
Ramp
Final emp
110.00
Events
Time
0 000
000
Event
ZERO
SOUND
NumberRetention Peak Area
1.626 1309.684
2.133 1218.513
3.100 474.851
Time (minutes)
EET'Z
066'T
1.626
123
123 4
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you



EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT



EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning