QUESTION 8: Metals react with oxygen gas to form metal oxide. For example, magnesium reacts as follows: 2Mg(s) + 0,(g) → 2Mg0(s). What is the mass (in grams) of magnesium oxide that can be prepared from 1.40g of 0,? G: 1.40g 02 мм: 1тol 0z — 32g 02 1 mol Mgo = 40.31g MgO MR: 2mol Mg0 = 1mol 02 СHOICES: А. 5.33g Mgо В. 33.5g MgO С. 5.33g Mgо D. 3.53g MgO
QUESTION 8: Metals react with oxygen gas to form metal oxide. For example, magnesium reacts as follows: 2Mg(s) + 0,(g) → 2Mg0(s). What is the mass (in grams) of magnesium oxide that can be prepared from 1.40g of 0,? G: 1.40g 02 мм: 1тol 0z — 32g 02 1 mol Mgo = 40.31g MgO MR: 2mol Mg0 = 1mol 02 СHOICES: А. 5.33g Mgо В. 33.5g MgO С. 5.33g Mgо D. 3.53g MgO
Chemistry & Chemical Reactivity
9th Edition
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter4: Stoichiometry: Quantitative Information About Chemical Reactions
Section4.1: Mass Relationships In Chemical Reactions: Stoichiometry
Problem 2RC
Related questions
Question
SHOW COMPLETE SOLUTION AND BOX THE FINAL ANSWER.
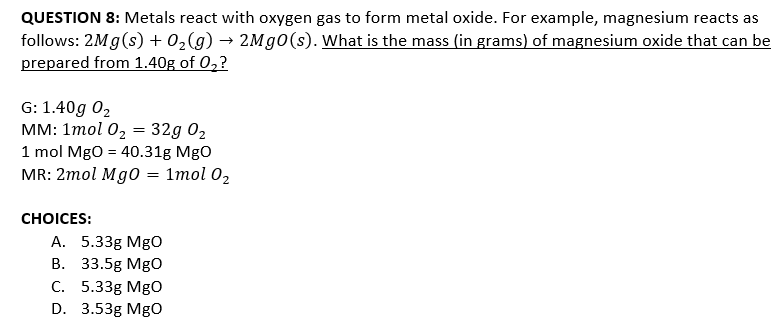
Transcribed Image Text:QUESTION 8: Metals react with oxygen gas to form metal oxide. For example, magnesium reacts as
follows: 2Mg(s) + 0,(g) → 2Mg0(s). What is the mass (in grams) of magnesium oxide that can be
prepared from 1.40g of 0,?
G: 1.40g 02
мм: 1тol 0z — 32g 02
1 mol Mgo = 40.31g MgO
MR: 2mol Mg0 = 1mol 02
СHOICES:
А. 5.33g Mgо
В. 33.5g MgO
С. 5.33g Mgо
D. 3.53g MgO
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning