Q: Draw the products formed when each amine is treated with [1] CH3I (excess); [2] Ag2O; [3] Δ.…
A: Solution: a) part Product formed is given below when treated with [1] CH3I (excess); [2] Ag2O; [3]…
Q: 5. The following aminoketone can undergo an intramolecular reductive amination. Draw the expected…
A:
Q: Rank the amines in each set in order of increasing basicity. H ot (b)bims n N (a) H. NH2 NH2 (c) H H…
A: ***Note: Since you have posted a question with multiple subparts, we will solve the first three…
Q: Draw the products formed when each amine is treated with [1] CH3I (excess); [2] Ag:0; [3] A.…
A: Hofmann elimination is a reaction in which amines are eliminated from the molecule to form alkenes.…
Q: N-Nitroso amines are stabilized by electron delocalization. Write the two most stable resonance…
A: Resonance is the delocalization of electrons to via double bonds.
Q: Oxytocin is a naturally occurring hormone that induces labor and stimulates milk production in…
A: The organic compound is formed by two main elements, carbon, and hydrogen. The specific group of…
Q: Show how you would convert propan-1-ol to the following compounds using tosylateintermediates. You…
A: One of the methods of amination of an alcohol involves the conversion of an alcohol to the tosylate…
Q: Identify all amines below that can NOT be made by reduction of an amide by LAH. -NH2 NH NH2 (A) (B)…
A:
Q: | Rank amines A-E from least basic to most basic. O,N. N-CH, -CH, -CH, E A в Answer: The basicity…
A: Basicity is the ability donate electron Higher the donation ability more basic it is Here We are…
Q: 7. The secondary amine and aldehyde below will react to form HN. a. An imine b. An enamine c. A…
A: The amine is nucleophile and the carbonyl carbon is the electrophile. So nucleophilic attack takes…
Q: Chlorpropamidé håš a lohger NH NHCH,CH,CH,CH, NH NHCH CH,CH,CH, Tobutamide Chlorpropamide Which of…
A: Tolbutamide is a first potassium channel blocker, sulfonylurea oral hypoglycemic medication. When…
Q: 1. Rank the following amines A-D in order of decreasing basicity, where 1 is the most basic. NO2 A…
A: The compounds given are,
Q: 15. What compound will be produced as a result ofaniline with nitrite acid interaction? A. CHs-N…
A:
Q: NH, M IZ IZ
A:
Q: What is the name of the major product of the following synthetic scheme? OH 1. CH,CH,NH, 275 C 2.…
A: In this reaction acid amine coupling takes place which results in formation of amide bond which…
Q: Question attached
A: Amines on reaction with HCl result in the formation of amine salt. Primary amines give primary amine…
Q: a. b. С. (CH3CH2)2NH NH2
A: Amides can be reduced to amines on treatment with reducing agents like LiAlH4. RCONH2 + LiAlH4…
Q: 3. For the following amines, propose a stepwise synthesis for each compound. ? NH2 a) OH b)
A: Step 1 The given amines can be formed using the alkenes as one of the reactants and with the use of…
Q: show how you might prepare each of the following amines through reductive aminations…
A:
Q: Rank the following amines in order of basicity.
A: The given compounds are: A. B.
Q: t)which B the fallowing Statements is true regardirg N- phenylocetamide ? 5aka a cetanilide Lamide…
A: This question is related to general organic chemistry. N-phenyl acetamide has an amide functional…
Q: 1(c) Provide the MAJOR products of the following reactions. CI, NaNH, A H, HO в Br, NaOH D CH,OH,…
A:
Q: The first step in the mechanism for the reaction of an amine with a carboxylic acid to produce an…
A: The first step involved in the reaction between amine and carboxylic acid is given below
Q: H3c CI Which of the following is an intermediate in the mechanism for amide synthesis through…
A:
Q: Which compound is not shown? NHCCH3 A. aspirin OB. saccharin OC. acetanilide O D. acetaminophen O E.…
A: The structure which is not shown is .
Q: 1.Predict the products of the following reactions: ΕΝΗ DMSO 3,4-dibromopyridine h…
A: The reaction in which nucleophile substitutes the leaving group from the aromatic ring is known as…
Q: What results when dimethylsulfide is treated with methyl iodide? 1) CH,I 2)- NH 3) -H A. B. C. D.
A: The reaction taking place is given as,
Q: Draw the structures of the missing substances in each of the following reactions involving amides.…
A: The amide functional group is denoted as follows: R-CO-N(R1)(R2), Where R, R1, and R2 can be alkyl…
Q: 22. Which amide has a line through the bond that is broken during hy 1- H. C D A 23. Give the…
A:
Q: ON 16 Which has the lowest boiling point? OA. CH3(CH2)5CONH2 O B. CH3CH2CON(CH2CH3)2 O C.…
A: The boiling point of a compound is determined based on the interaction among the molecule. The…
Q: The following rearrangement reactions give a primary amine EXCEPT: A. Hoffmann B. Lossen C. Curtius…
A: Except Beckmann all option s give primary amine. Beckmann convert ketone to amide
Q: 5. show two different ways of preparing each of the following compounds via a reductive amination:…
A: In reductive amination reaction, carbonyl compound reacts with ammonia or amine to form an imine…
Q: Which of the following reductive amination reactions will yield a tertiary amine? *NH2 しiy H lime…
A:
Q: Draw the structure that corresponds to each name.a. N-isobutylcyclopentanamineb.…
A: Hey, since there are multiple sub parts questions posted, we will answer first three questions. If…
Q: 3. If the compound shown is subjected to reaction conditions that will hydrolyze amides one of two…
A: Solution of the question is given below:
Q: What is the product of the following reductive amination? CH, CH, NHz mild acd Nafity or Na BH3CN…
A: This is an typical example of reductive amination. The details solution for this reaction is…
Q: Which of the following will have the strongest acid-base interaction with NH3? a. BH3 b. BMe3 c.…
A: Fluorine is more electronegative than hydrogen, so due to inductive effects, the electron density…
Q: Name the following compound: [(NH3)5Co(NH2)Co(NH3)5]Cl5 H-aminobis [tetraamminecobalt(III)] chloride…
A: Co-ordination compounds are formed when the metal ion is bonded to the ligand which donates its…
Q: Rank the following amines in order of basicity from hig
A: Electron pair donar is called " Lewis base " Example : amines In amines N atom has lone pairs…
Q: O Which reagents can reduce amide? 1. LIAIHY 2. H20, H30* NOBH ETOH H2, Pd /CLIAIHO+-Bi); 78C
A:
Q: Which of the following compounds would form Azo-dye from primary aromatic amines? Indicate YES or NO…
A: Azo dye is a test to distinguish between an aromatic amine and aliphatic amine. Only aromatic amines…
Q: Select the best answer that identifies the compound(s) below that would furnish an amine on…
A: Following organic compounds will give amine when reacts with excess LiAlH4.
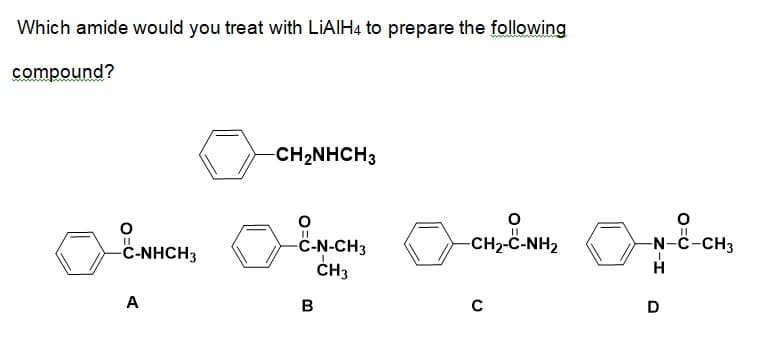
Q: what amides would you treat with LiAlH4 to meke th following compound? benzylmethylamine
A:
Q: Given the following compounds and boiling points: Determine the IMFs Order the compounds from…
A: Boiling point of any substance is proportional to 2 things in the below mentioned order. 1) The…
Q: Rank the following compounds in order of increasing basicity. CH3 H3C° CH3 II 4® // H3C- CH3 CH3 N.…
A: Anion are very strong bases . Anion are present in two option III AND IV but in anion III N2O group…
Q: 3. If the compound shown is subjected to reaction conditions that will hydrolyze amides one of two…
A: This question is related to the stability of products. More stable products will form Faster. Since…
Q: (2) Compound B has a formula CSH100. 1H NMR: 6 3.5 ppm (4H, singlet), & 0.9 ppm (6H, singlet). 13C…
A:
Q: (a) NCH3 (b) NH2 -
A:
Step by step
Solved in 2 steps with 2 images

- Which amine below is an primary aromatic amine? a. N-methyl-2-phenylethanamine b. All of the given c. 3-methylbenzamine d. N-ethylbenzamineRank the following amides from greatest reactivity to least reactivity toward acid-catalyzed hydrolysis:The –NHCOR group of an amide is an activating group, but it is not as strongly activating as NH2. (a) Explain why it is an activating group. (b) Explain why it is less activating than NH2.
- N-Nitroso amines are stabilized by electron delocalization. Write the two most stable resonance contributors of N-nitrosodimethylamine, (CH3)2NNO.Indicate whether the following statement is true or false. Aliphatic amines are more basic than ammonia, whereas aromatic amines are less basic than ammonia. When amines are reacted with bases, they form ammonium salts. Benzenesulfonyl chloride or p-toluenesulfonylchloride give N-substituted sulfonamides with primary and secondary amines. Derivative of primary amines is insoluble in dilute NaOHWhat products are formed when the amide shown below is heated in aqueous NaOH solution for three hours?

