Which set of atoms below are metals that have only one electron in their outermost "s" orbitals? H Не Li Be N F Ne 11 12 13 14 15 16 17 18 Na Mg Al Si cI Ar 19 20 21 22 23 24 25 26 27 28 29 31 32 33 34 35 36 к Са Sc Ti v Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 37 39 41 42 43 44 45 47 53 54 Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te Xe 57-70 71 72 73 74 75 76 77 79 82 83 84 85 Cs Ba Lu Hf Ta w Re Os Ir Pt Au Hg TI Pb Bi Po At Rn 183 104 105 110 111 112 114 Fr Ra** Lr Rf Db Sg Bh Hs Mt Uun Uuu Uub Uuq 65 70 "Lanthanide series S7 64 La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb ** Actinide series MARA 54 91 92 33 97 99 100 101 12 Ac Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Select one: O a Zn, Cd, Hg O b. K, Rb, Cs O. Ni, Pd, Pt Od. Mn, Tc, Re O e. Sc, Y, Lu
Which set of atoms below are metals that have only one electron in their outermost "s" orbitals? H Не Li Be N F Ne 11 12 13 14 15 16 17 18 Na Mg Al Si cI Ar 19 20 21 22 23 24 25 26 27 28 29 31 32 33 34 35 36 к Са Sc Ti v Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 37 39 41 42 43 44 45 47 53 54 Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te Xe 57-70 71 72 73 74 75 76 77 79 82 83 84 85 Cs Ba Lu Hf Ta w Re Os Ir Pt Au Hg TI Pb Bi Po At Rn 183 104 105 110 111 112 114 Fr Ra** Lr Rf Db Sg Bh Hs Mt Uun Uuu Uub Uuq 65 70 "Lanthanide series S7 64 La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb ** Actinide series MARA 54 91 92 33 97 99 100 101 12 Ac Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Select one: O a Zn, Cd, Hg O b. K, Rb, Cs O. Ni, Pd, Pt Od. Mn, Tc, Re O e. Sc, Y, Lu
General Chemistry - Standalone book (MindTap Course List)
11th Edition
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Chapter8: Electron Configurations And Periodicity
Section: Chapter Questions
Problem 8.110QP: The electron affinity of the lutetium atom (element 71) was measured using the technique of...
Related questions
Question
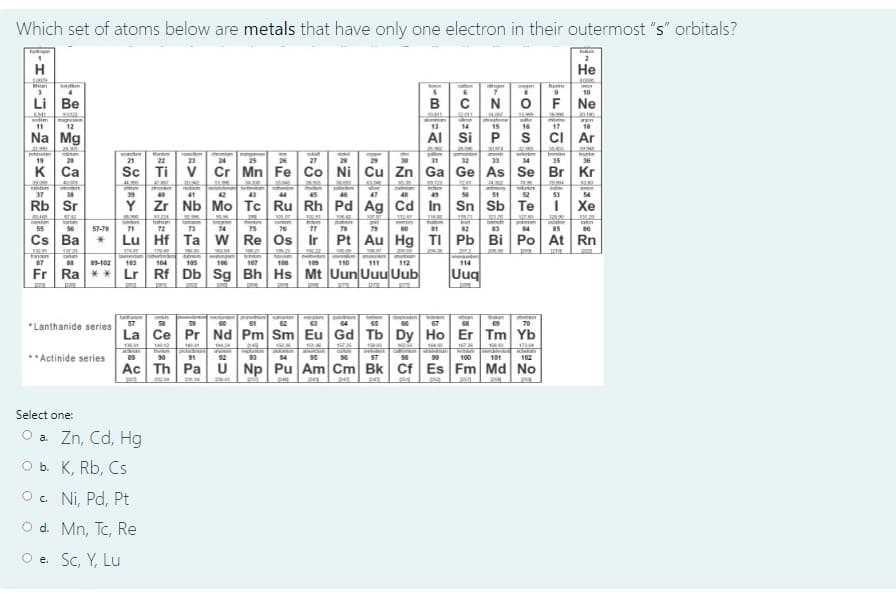
Transcribed Image Text:Which set of atoms below are metals that have only one electron in their outermost "s" orbitals?
H
Не
Rere
10
Li Be
B
N OFN.
2010
rgon
18
11
12
14
15
16
17
Na Mg
AI Si
ci Ar
ptm
19
vam
23
mongon
25
26
30
Inomie
35
kpte
36
20
21
22
24
27
28
29
31
32
33
34
K Ca
Sc Ti v Cr Mn Fe Co Ni Cu Zn Ga Ge As Se
Br Kr
33000
2052
otne
37
38
39
40
41
42
43
44
45
46
47
48
49
50
51
52
53
54
Rb Sr
Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In
Sn Sb Te
Хе
12
ca
55
fomum
72
tolkm
81
RLA
bamh
83
hortam
ad
56
57-70
71
73
74
75
76
77
78
79
80
82
84
85
86
Cs Ba
Lu Hf Ta
W Re Os Ir
Pt Au Hg TI Pb Bi Po At Rn
STA
tann
boh
107
umatan
112
87
83-102
103
104
105
106
108
109
110
111
114
Fr Ra ** Lr
Rf Db Sg Bh Hs Mt Uun Uuu Uub
Uuq
57
61
62
63
64
65
67
68
69
70
"Lanthanide series
La Ce Pr
Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb
174
** Actinide series
90
92
93
84
95
96
97
99
100
101
102
Ac Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No
Select one:
O a Zn, Cd, Hg
O b. K, Rb, Cs
O . Ni, Pd, Pt
O d. Mn, Tc, Re
O e. Sc, Y, Lu
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning