Q: Explain why iodine (I2) does not react with ethane, even though I2 is more easily cleaved…
A: Radical or free radical: unpaired valence electron of an atom, molecule, or ion is called as…
Q: Does the presence or absence of a catalyst such as finely divided platinum, palladium, or nickel…
A: The effect of catalyst on equilibrium constant for ethylene-ethane conversion has to be given.
Q: Explain the process of Designing a Multistep Synthesis
A: Chemical synthesis refers to artificial means to carry out reactions in the lab using suitable…
Q: Give the products of the following reactions. Draw the structures. (a) HgCl2 2 MeMgCI +
A: When two or more compounds react together to form a new compound, the process is known as the…
Q: Please enumerate and explain 1. WHAT ARE THE GENERAL RULES IN GRAMS STAINING CLASSIFICATION OF…
A: In microbiology, Gram staining is the most widely utilised, vital, and widely used differential…
Q: Discuss the role of the following in the liquid-liquid extraction of dyes. A. Vinegar B. Baking…
A: Components are partitioned between an aqueous layer and the organic layer in the liquid-liquid…
Q: properties of the catalyst(e.g., shape, size, colour, price). a) aluminosilicate b) trypsin
A: A) aluminosilicate : Properties Aluminosilicate glasses can be formulated to tolerate temperatures…
Q: ) Answer the following questions regarding solvent polarity and reaction rates. i. Suggest how to…
A: Transition state can be defined as the state where new bonds are formed and old bonds are broken.
Q: Give out chemical reactions of butadiene complexs Please answer breifly ASAP
A:
Q: Define the following terms :(i) Kraft temperature(ii) Peptization(iii) Electrokinetic potential
A: Surface chemistry is a phenomenon occurring between two different surfaces.
Q: You are conducting a biochemical experiment with an enzyme that has optimal activity at pH = 6.50.…
A: Buffers are solutions that are used in experiments to resist the change in pH. Carbonic acid is a…
Q: 4) In the lab you are extracting and enzyme called catalase from a potato and want to use this…
A: Enzymes are commonly help in speed up the reaction. Catalase is an enzyme that is found in almost…
Q: Draw the structures of the missing reactants, intermediates, or products in the following mechanism.…
A:
Q: I am studying homolytic and heterolytic cleavage. In the halogenation of methane, a methyl radical…
A: During halogenation of alkane, homolytic cleavage takes place. For the heterolytic cleavage there…
Q: Calculate the specificity constant for an enzyme-catalyzed reaction if the total enzyme…
A: Given Etotal = 15 nMVmax = 9 M/sKm = 150 MSpecificity constant = to be determined
Q: You are conducting a biochemical experiment with an enzyme that has optimal activity at pH = 10.50.…
A: Buffer Solution is a water solvent based solution which consists of a mixture containing a weak acid…
Q: Outline the possible reaction pathways with proper mechanism for the formation of Nylon 6 from…
A: Polymerization: It is a chemical process when a monomer (single unit) combined through various…
Q: Briefly explain about Degradation of azo dyes , include aerobic, anaerobic etc.
A: Let's discuss about the degradation process of azo dyes.
Q: what will happen to the rate constant if the concentrat -fold.
A: As we know in elementary reaction order of reaction is sum of stoichometric coefficients of…
Q: Write the reaction of Polypropylene stereochemistry with Ziegler-Natta catalysts
A: Ziegler natta catalyst
Q: What is IR active and IR inactive?
A: IR spectroscopy corresponds to the transition between the vibrational levels of a molecule. It…
Q: Many enzymes are inhibited irreversibly by heavy metal ions such as Hg²+ , Cu²+ , or Ag*, which can…
A: In a titration between the metal salt and sulfhydryl group, the metal ion replaces the proton of the…
Q: Structure
A:
Q: the advantages and the disadvantages of the catalyst a) aluminosilicate b) trypsin
A: Advantages of catalyst aluminosilicate - aluminum silicate is basically used to dry oozing and…
Q: Briefly highlight the mechanism of hydrogenation of alkenes on Ni or Pt catalyst surface. * What is…
A:
Q: Which group of side chains is most likely to take part in the catalysis during an enzymatic…
A: The solution of the question is given below:
Q: what happens to the pigment of egg yolk during emulsion? Explain the mechanism or the reason why the…
A: An emulsion is thicker or more viscous than its separate components, or the oil and water it…
Q: An enzyme reaction takes place in a 10mL solution that has a total citrate concentration of 120mM…
A: Part (a)Citric acid is a triprotic acid. Since the value of initial pH is higher than pKa3, the…
Q: Explain and describe details for this mechanism.
A: Acid protonated the carbonyl oxygen and makes it electrophilic or increases ots electrophilicity
Q: A. The Cannabis Gummies were illegally shipped into the country but were intercepted at NAIA upon…
A: As per the Bartley expert guidelines, I am allowed to answer first one question at a time. Please…
Q: Write the mechanism for the formation of isopropylbenzene.
A: Nitrobenzene can be formed from benzene by aromatic electrophilic substitution and this we use…
Q: In PLP-catalyzed reactions, the bond broken in the substrate molecule must be perpendicular to the…
A: The PLP is referred to as Pyridoxal 5′-phosphate and it has a function that it acts as a coenzyme in…
Q: State TWO (2) organometallic reactions that involve the concept of dn-pn bonding.
A: The above question describes one of the important reaction in organometallics i. e. the reaction…
Q: Provide a catalytic cycle for the hydrogenation of ethylene using Vaska's Complex. H2 + H2C=CH2…
A: The answer is given as follows
Q: Explain your answer. This is the most important part. Please type on an attached page. A complete…
A: Boiling point direct proportional to intermolecular force.
Q: The substrate dissociation constant, Ks, is k.1/k1. What do large values of this constant mean? That…
A:
Q: Write the factors upon which the catalytic reaction of shape-selectivecatalyst depends?
A: Write the factors upon which the catalytic reaction of shape-selective catalyst depends?
Q: ind the isoelectric and zwitter ion of lysine, show the steps
A: Given the isoelectric and zwitter ion of lysine
Q: Would the continuous assay of Alkaline phosphatase (kinetics lab) with PNPP as a substrate work if…
A:
Q: the end reversible process. What IR bands would you want to follow if you needed to monitor corbonu…
A: Given that, a natural process of in which nicotinamide adenine dinucleotide (NAD+) used to oxidise…
Q: What hazard is associated with using palladium on carbon as a hydrogenation catalyst? and define…
A: ◆ Palladium on carbon (Pd/C) is highly flammable finely divided metal so it may catch fire while…
Q: tautamerism
A: HYPERCONJUGATION It is transfer or we say delocalization of sigma electron. It is generally two…
Q: The initial reaction rate for the reaction in question 23 can be followed spectrophotometrically by…
A:
Q: Compare the column material of GC and LC in terms of the “functional groups” taking place on the…
A: In GC there are two type of coloumn.packed coloumn And capillary Coloumn. In gas liquid…
Q: Give handwritten answer of questions no. 4
A: Here we have to explain above mass spectra of two molecules-
Q: on what factors does the catalytic property of zeolite depends upon?
A: A catalytic reaction which depends upon the pore structure of the catalyst and on the size of the…
Q: Ni(II)-aminopyridine complex was synthesized using a metal-template condensation reaction presented…
A: Answer is given in 4 images, 5th image contain summary of all 4. Thank you.
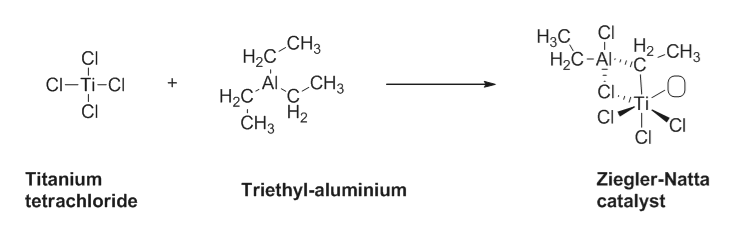
write the Zigler Natta catalyst?
The Zigler Natta catalyst is synthesised from the reaction of titanium tetrachloride with triethyl aluminium.
Mostly it used in the Polymerisation reaction.
Step by step
Solved in 2 steps with 2 images

- Explain how the light naphtha isomerization catalyst is bifunctional.What is the location in the catalyst of the different kinds of sites and what function do they serve?Give an example of ‘shape-selective catalyst’.An example of a heterogeneous catalyst is: a. Wilkinson’s catalyst for the hydrogenation of alkenes b. Grubb’s catalyst with varied substituents for olefin metathesis in THF c. Pt and Pd nanoparticles on ceramic honeycombs in catalytic converters d. Pd(PPh3)4 in toluene solvent used for Miyaura-Suzuki coupling reactions

