You may assume all chemical bonds are single bonds, not double or triple bonds. element pair will form a molecular molecular compound element #1 element ##2 chemical name compound formula carbon iodine hydrogen bromine lithium oxygen
You may assume all chemical bonds are single bonds, not double or triple bonds. element pair will form a molecular molecular compound element #1 element ##2 chemical name compound formula carbon iodine hydrogen bromine lithium oxygen
General, Organic, and Biological Chemistry
7th Edition
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:H. Stephen Stoker
Chapter5: Chemical Bonding: The Covalent Bond Model
Section: Chapter Questions
Problem 5.11EP: The component elements for four binary molecular compounds are shown with different colors on the...
Related questions
Concept explainers
Bond Parameters
Many factors decide the covalent bonding between atoms. Some of the bond parameters are bond angle, bond order, enthalpy, bond length, etc. These parameters decide what kind of bond will form in atoms. Hence it is crucial to understand these parameters in detail and understand how changing these parameters affects the kind of bonding or various characteristics.
Bond Dissociation Energy
The tendency of an atom to attract an electron is known as its electronegativity.
Question
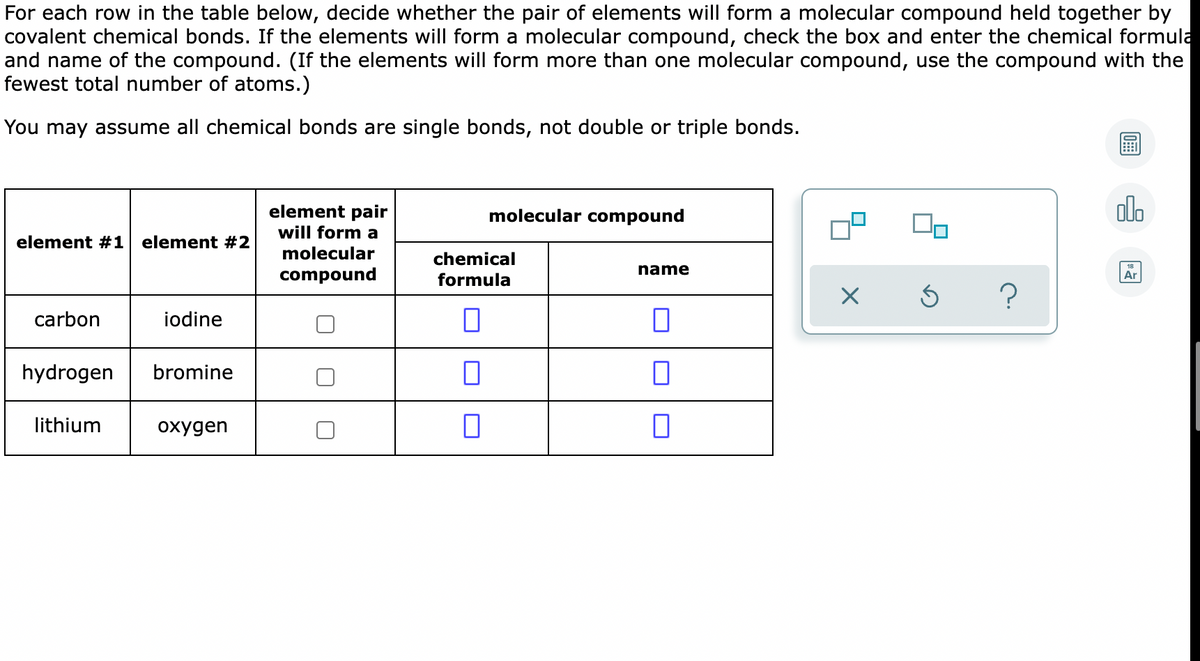
Transcribed Image Text:For each row in the table below, decide whether the pair of elements will form a molecular compound held together by
covalent chemical bonds. If the elements will form a molecular compound, check the box and enter the chemical formula
and name of the compound. (If the elements will form more than one molecular compound, use the compound with the
fewest total number of atoms.)
You may assume all chemical bonds are single bonds, not double or triple bonds.
element pair
alo
molecular compound
will form a
element #1 element #2
molecular
chemical
name
compound
formula
Ar
carbon
iodine
hydrogen
bromine
lithium
охудеn
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning