You need to make 120.0 mL of a 0.40 M NaCl solution. You have solid NaCl and your lab partner has a 1.5 MNaCl solution. Explain how you each make the 0.40 M NaCl solution. (a) What mass of the solid NaCl should you dissolve in pure water in order to prepare 120.0 mL of 0.40 M solution? Mass of NaCl= (b) What volume of 1.5 MNaCl solution should your partner mix with pure water in order to prepare 120.0 mL of 0.40 M solution? Volume = mL
You need to make 120.0 mL of a 0.40 M NaCl solution. You have solid NaCl and your lab partner has a 1.5 MNaCl solution. Explain how you each make the 0.40 M NaCl solution. (a) What mass of the solid NaCl should you dissolve in pure water in order to prepare 120.0 mL of 0.40 M solution? Mass of NaCl= (b) What volume of 1.5 MNaCl solution should your partner mix with pure water in order to prepare 120.0 mL of 0.40 M solution? Volume = mL
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter15: Acid–base Equilibria
Section: Chapter Questions
Problem 98AP
Related questions
Question
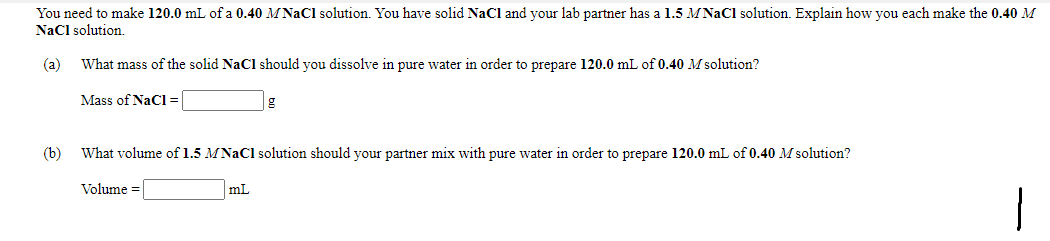
Transcribed Image Text:You need to make 120.0 mL of a 0.40 MNAC1 solution. You have solid NaCl and your lab partner has a 1.5 MNaCl solution. Explain how you each make the 0.40 M
Nacl solution.
(a)
What mass of the solid NaCl should you dissolve in pure water in order to prepare 120.0 mL of 0.40 M solution?
Mass of NaCl=
(b)
What volume of 1.5 MNAC1 solution should your partner mix with pure water in order to prepare 120.0 mL of 0.40 M solution?
Volume =
mL
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning