YOUR WORK!!!!! 13. A car traveling south at a velocity of 30 m/sec slows down to a velocity of 10 m/sec in 10 sec. Calculate the deceleration. 14. A car accelerates from 0 to 72 m/sec in 8 sec. What is the car's acceleration?
YOUR WORK!!!!! 13. A car traveling south at a velocity of 30 m/sec slows down to a velocity of 10 m/sec in 10 sec. Calculate the deceleration. 14. A car accelerates from 0 to 72 m/sec in 8 sec. What is the car's acceleration?
Chemical Principles in the Laboratory
11th Edition
ISBN:9781305264434
Author:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Chapter21: Rates Of Chemical Reactions, Ii. A Clock Reaction
Section: Chapter Questions
Problem 2ASA
Related questions
Question
Can you help me with some of these it doesn’t have to be all of them I would appreciate it
Transcribed Image Text:Submit Assignr
nez - Acceleration Worksheet 6767218 ☆
Format Tools Add-ons Help
Last edit was 19 minutes ago
Normal text
Arial
10
+
IUA
| 1 2 | 3 4 I 5
I.. 6 I 7
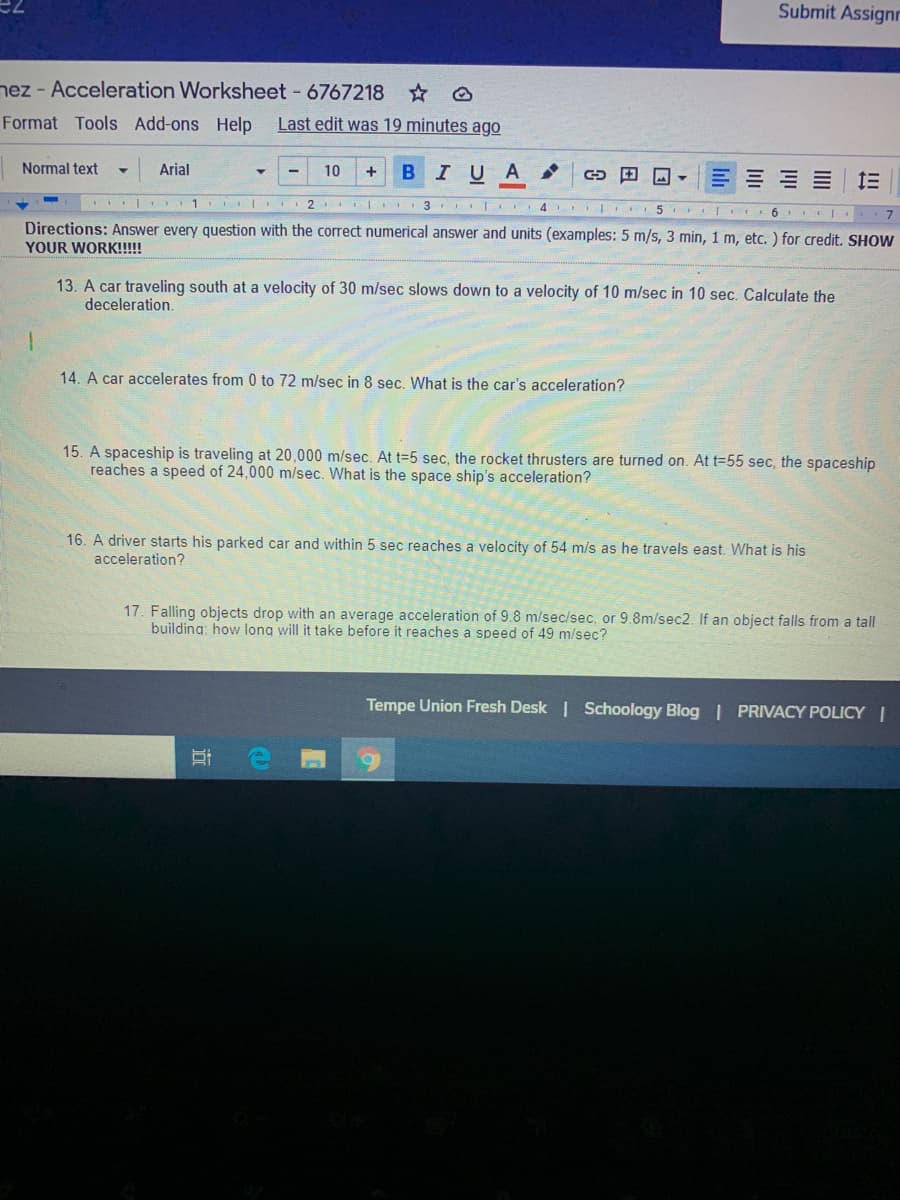
Directions: Answer every question with the correct numerical answer and units (examples: 5 m/s, 3 min, 1 m, etc. ) for credit. SHOW
YOUR WORK!!!
13. A car traveling south at a velocity of 30 m/sec slows down to a velocity of 10 m/sec in 10 sec. Calculate the
deceleration.
14. A car accelerates from 0 to 72 m/sec in 8 sec. What is the car's acceleration?
15. A spaceship is traveling at 20,000 m/sec. At t=5 sec, the rocket thrusters are turned on. At t=55 sec, the spaceship
reaches a speed of 24,000 m/sec. What is the space ship's acceleration?
16. A driver starts his parked car and within 5 sec reaches a velocity of 54 m/s as he travels east. What is his
acceleration?
17. Falling objects drop with an average acceleration of 9.8 m/sec/sec, or 9.8m/sec2. If an object falls from a tall
building: how long will it take before it reaches a speed of 49 m/sec?
Tempe Union Fresh Desk | Schoology Blog | PRIVACY POLICY |
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole