
Loose Leaf for Organic Chemistry
5th Edition
ISBN: 9781259637025
Author: Janice Smith
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 1.23P
Convert each skeletal structure to a complete structure with all
a. 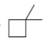 b.
b. 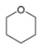 c.
c. 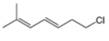 d.
d. 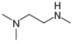
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Convert each skeletal structure to a complete structure with all C’s and H’s drawn in.
Draw an acceptable Lewis structure from each condensed structure, such that all atoms have zero formal charge.
a. diethyl ether, (CH3CH2)2O, the first general anesthetic used in medical procedures
b.acrylonitrile, CH2CHCN, starting material used to manufacture synthetic Orlon fibers
c.dihydroxyacetone, (HOCH2)2CO, an ingredient in sunless tanning products
d.acetic anhydride, (CH3CO)2O, a reagent used to synthesize aspirin
Draw an acceptable Lewis structure from each condensed structure, such that all atoms have zero formal charge.
a diethyl ether, (CH3CH2)2O, the first general anesthetic used in medical procedures
b. acrylonitrile, CH2CHCN, starting material used to manufacture synthetic Orlon fibers
c.dihydroxyacetone, (HOCH2)2CO, an ingredient in sunless tanning products
d.acetic anhydride, (CH3CO)2O, a reagent used to synthesize aspirin
Chapter 1 Solutions
Loose Leaf for Organic Chemistry
Ch. 1 - While the most common isotope of nitrogen has a...Ch. 1 - Label each bond in the following compounds as...Ch. 1 - How many covalent bonds are predicted for each...Ch. 1 - Draw a valid Lewis structure for each species. a....Ch. 1 - Draw an acceptable Lewis structure for each...Ch. 1 - Prob. 1.6PCh. 1 - Draw a Lewis structure for each ion. a. CH3Ob....Ch. 1 - Prob. 1.8PCh. 1 - Draw Lewis structures for each molecular formula....Ch. 1 - Classify each pair of compounds as isomers or...
Ch. 1 - Prob. 1.11PCh. 1 - Prob. 1.12PCh. 1 - Prob. 1.13PCh. 1 - Draw a second resonance structure for each species...Ch. 1 - Prob. 1.15PCh. 1 - Prob. 1.16PCh. 1 - Prob. 1.17PCh. 1 - Prob. 1.18PCh. 1 - Using the principles of VSEPR theory, you can...Ch. 1 - Convert each condensed formula to a Lewis...Ch. 1 - Prob. 1.21PCh. 1 - Prob. 1.22PCh. 1 - Convert each skeletal structure to a complete...Ch. 1 - What is the molecular formula of quinine, the...Ch. 1 - Draw in all hydrogens and lone pairs on the...Ch. 1 - Prob. 1.26PCh. 1 - What orbitals are used to form each of the CC, and...Ch. 1 - What orbitals are used to form each bond in the...Ch. 1 - Determine the hybridization around the highlighted...Ch. 1 - The unmistakable odor of a freshly cut cucumber is...Ch. 1 - Prob. 1.31PCh. 1 - Rank the following atoms in order of increasing...Ch. 1 - Prob. 1.33PCh. 1 - Prob. 1.34PCh. 1 - Provide the following information about...Ch. 1 - Use the ball-and-stick model to answer each...Ch. 1 - Citric acid is responsible for the tartness of...Ch. 1 - Zingerone gives ginger its pungent taste. a.What...Ch. 1 - Assign formal charges to each carbon atom in the...Ch. 1 - Assign formal charges to each and atom in the...Ch. 1 - Prob. 1.41PCh. 1 - Prob. 1.42PCh. 1 - Prob. 1.43PCh. 1 - Draw all possible isomers for each molecular...Ch. 1 - 1.45 Draw Lewis structures for the nine isomers...Ch. 1 - Prob. 1.46PCh. 1 - Prob. 1.47PCh. 1 - Prob. 1.48PCh. 1 - Prob. 1.49PCh. 1 - Prob. 1.50PCh. 1 - Prob. 1.51PCh. 1 - Prob. 1.52PCh. 1 - Consider compounds A-D, which contain both a...Ch. 1 - Prob. 1.54PCh. 1 - Prob. 1.55PCh. 1 - 1.56 Consider the compounds and ions with curved...Ch. 1 - 1.57 Predict all bond angles in each...Ch. 1 - 1.58 Predict the geometry around each highlighted...Ch. 1 - Prob. 1.59PCh. 1 - Draw in all the carbon and hydrogen atoms in each...Ch. 1 - 1.61 Convert each molecule into a skeletal...Ch. 1 - Prob. 1.62PCh. 1 - Prob. 1.63PCh. 1 - Predict the hybridization and geometry around each...Ch. 1 - Prob. 1.65PCh. 1 - Ketene, , is an unusual organic molecule that has...Ch. 1 - Rank the following bonds in order of increasing...Ch. 1 - Prob. 1.68PCh. 1 - Two useful organic compounds that contain Cl atoms...Ch. 1 - Use the symbols + and to indicate the polarity of...Ch. 1 - Prob. 1.71PCh. 1 - Anacin is an over-the-counter pain reliever that...Ch. 1 - Answer the following questions about acetonitrile...Ch. 1 - Prob. 1.74PCh. 1 - 1.75 The principles of this chapter can be...Ch. 1 -
a. What is the hybridization of each N atom in...Ch. 1 - 1.77 Stalevo is the trade name for a medication...Ch. 1 - 1.78 and are two highly reactive carbon...Ch. 1 - 1.79 The N atom in (acetamide) is hybridized,...Ch. 1 - Prob. 1.80PCh. 1 - Prob. 1.81PCh. 1 - Prob. 1.82PCh. 1 - Prob. 1.83PCh. 1 - Prob. 1.84PCh. 1 - Prob. 1.85P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Functionalized Hydrocarbons Identify each compound according to its functional group (e.g.,amine,ester,etc.):arrow_forwardDraw a skeletal structure for each condensed structure given and draw a condensed structure for each skeletal structure.arrow_forwarddraw expanded,condensed and skeletal structure of atenololarrow_forward
- Draw an acceptable Lewis structure from each condensed structure, such that all atoms have zero formal charge. a. diethyl ether, (CH3CH2)2O, the rst general anesthetic used in medical proceduresb. acrylonitrile, CH2CHCN, starting material used to manufacture synthetic Orlon bersc. dihydroxyacetone, (HOCH2)2CO, an ingredient in sunless tanning productsd. acetic anhydride, (CH3CO)2O, a reagent used to synthesize aspirinarrow_forwardConvert each condensed formula to a Lewis structure. a.CH3(CH2)4CH(CH3)2 b. (CH3)3CCH(OH)CH2CH3 c. (CH3)2CHCHO d.(HOCH2)2CH(CH2)3C(CH3)2CH2CH3arrow_forwardDraw the skeletal formula of A, B and Carrow_forward
- NAME AND CLASSIFY MOLECULES ACCORDING TO THEIR FUNCTIONAL GROUP(S)arrow_forwarda) what two types of bonds are present in alkynes but not alkanes? b) draw the structure of an alkyne that would not have one of the two bonds. Explain.arrow_forwardplease help with all parts Convert the following condensed structures to Kekule´Kekule´ structures, showing the covalent bonds. Draw the molecule by placing atoms on the canvas and connecting them with bonds. Include all hydrogen atoms and nonbonding electrons.: part 1) (CH3)2CHCl part2) (CH3)3CBr part 3) (CH3)3C(CH2)3CHOarrow_forward
- How would I convert each condensed formula to a lewis structure 1a. (CH3)2CHOCH2CH2CH2OH 1b. CH3(CH2)2CO2C(CH3)3arrow_forwardConvert each condensed formula to a Lewis structure. CH3(CH2)4CH(CH3)2 (CH3)3CCH(OH)CH2CH3 (CH3)2CHCHO (HOCH2)2CH(CH2)3C(CH3)2CH2CH3arrow_forwardConvert the following condensed formulas into skeletal structures. a. CH3CONHCH3 b. CH3COCH2Br c. (CH3)COH d. CH3COCl e. CH3COCH2CO2H f. HO2CCH(OH)CO2Harrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co


Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License