
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
8th Edition
ISBN: 8220102895805
Author: Peterson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 1, Problem 1.30UKC
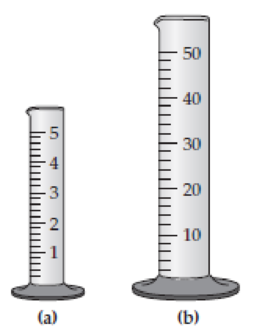
Assume that you have two graduated cylinders, one with a capacity of 5 mL (a) and the other with a capacity of 50 mL (b). Draw a line in each showing how much liquid you would add if you needed to measure 2.64 mL of water. Which cylinder do you think is more precise? Explain.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
How many grams of NaOH would you need to make 3,982 mL of a 3 M NAOH solution (m.w. = 40.00 g/mol)?
Report your answer in the requested units to the nearest 2 decimal places (Example "40.02"). Do not report the units
in your answer; enter the numbers only. It is recommended that you do not round between calculation steps.
How many grams of NaOH would you need to make 7,252 mL of a 230 mM NaOH solution (m.w. = 40.00 g/mol)?
Do not include units in your answer; report your answer in the requested units.
What volume (in mL) is 60% of 1.25 litres?
Chapter 1 Solutions
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
Ch. 1.2 - Pure acetic acid, which gives the sour taste to...Ch. 1.3 - Classify each of the following as a mixture or a...Ch. 1.3 - Classify each of the following as a physical...Ch. 1.3 - In the next image, red spheres represent element A...Ch. 1.3 - The active ingredient in aspirin, ASA, melts at...Ch. 1.3 - Prob. 1.2CIAPCh. 1.4 - Match the names of the elements described below...Ch. 1.4 - Identify the elements represented in each of the...Ch. 1.6 - Calomel (Hg2Cl2) is not toxic but methyl mercury...Ch. 1.6 - Give the full name of the following units and...
Ch. 1.8 - Prob. 1.8PCh. 1.8 - How would you record the temperature reading on...Ch. 1.8 - Prob. 1.10PCh. 1.8 - Convert the following values from scientific...Ch. 1.8 - Prob. 1.12PCh. 1.9 - Round off the following quantities to the...Ch. 1.9 - Carry out the following calculations, rounding...Ch. 1.10 - Prob. 1.15PCh. 1.10 - Convert 0.840 qt to milliliters in a single...Ch. 1.10 - A patient is to receive 20 mg of methimazole, a...Ch. 1.10 - Calculate the dosage in milligrams per kilogram...Ch. 1.11 - A thermochromic plastic chip included in a...Ch. 1.11 - A temperature-sensitive bath toy undergoes several...Ch. 1.11 - The highest land temperature ever recorded was 136...Ch. 1.11 - Prob. 1.20PCh. 1.11 - Prob. 1.21PCh. 1.11 - What is the specific heat of aluminum if it takes...Ch. 1.12 - A sample of pumice, a porous volcanic rock, weighs...Ch. 1.12 - Chloroform, once used as an anesthetic agent, has...Ch. 1.12 - The sulfuric acid solution in an automobile...Ch. 1.12 - Prob. 1.6CIAPCh. 1.12 - Prob. 1.7CIAPCh. 1 - The six elements in blue at the far right of the...Ch. 1 - Identify the three elements indicated on the...Ch. 1 - The radioactive element indicated on the following...Ch. 1 - (a)What is the specific gravity of the following...Ch. 1 - Assume that you have two graduated cylinders, one...Ch. 1 - State the length of the pencil depicted in the...Ch. 1 - Assume that you are delivering a solution sample...Ch. 1 - Assume that identical hydrometers are placed in...Ch. 1 - What is the difference between a physical change...Ch. 1 - Which of the following is a physical change and...Ch. 1 - Which of the following is a physical change and...Ch. 1 - Name and describe the three states of matter.Ch. 1 - Prob. 1.38ASPCh. 1 - Sulfur dioxide is a compound produced when sulfur...Ch. 1 - Butane (C4H8) is an easily compressible gas used...Ch. 1 - Classify each of the following as a mixture or a...Ch. 1 - Which of these terms, (i) mixture, (ii) solid,...Ch. 1 - Hydrogen peroxide, often used in solutions to...Ch. 1 - Prob. 1.44ASPCh. 1 - What is the most abundant element in the earths...Ch. 1 - Prob. 1.46ASPCh. 1 - Supply the missing names or symbols for the...Ch. 1 - Prob. 1.48ASPCh. 1 - Prob. 1.49ASPCh. 1 - Prob. 1.50ASPCh. 1 - Glucose, a form of sugar, has the formula C6H12O6....Ch. 1 - Prob. 1.52ASPCh. 1 - Prob. 1.53ASPCh. 1 - Prob. 1.54ASPCh. 1 - Prob. 1.55ASPCh. 1 - Prob. 1.56ASPCh. 1 - Prob. 1.57ASPCh. 1 - How many pictograms are in 1 mg? In 35 ng?Ch. 1 - How many microliters are in 1 L? In 20 mL?Ch. 1 - Prob. 1.60ASPCh. 1 - Prob. 1.61ASPCh. 1 - Prob. 1.62ASPCh. 1 - Prob. 1.63ASPCh. 1 - Prob. 1.64ASPCh. 1 - Round off each of the numbers in Problem 1.63 to...Ch. 1 - Carry out the following calculations, express each...Ch. 1 - Prob. 1.67ASPCh. 1 - Carry out the following conversions: (a) 3.614 mg...Ch. 1 - Carry out the following conversions. Consult...Ch. 1 - Express the following quantities in more...Ch. 1 - Fill in the blanks to complete the equivalencies...Ch. 1 - Prob. 1.72ASPCh. 1 - The muzzle velocity of a projectile fired from a 9...Ch. 1 - Prob. 1.74ASPCh. 1 - The Willis Tower in Chicago has an approximate...Ch. 1 - Prob. 1.76ASPCh. 1 - Prob. 1.77ASPCh. 1 - The white blood cell concentration in normal blood...Ch. 1 - Prob. 1.79ASPCh. 1 - Diethyl ether, a substance once used as a general...Ch. 1 - Prob. 1.81ASPCh. 1 - Calculate the specific heat of copper if it takes...Ch. 1 - Prob. 1.83ASPCh. 1 - A 150 g sample of mercury and a 150 g sample of...Ch. 1 - When 100 cal (418 J) of heat is applied to a 125 g...Ch. 1 - Prob. 1.86ASPCh. 1 - Prob. 1.87ASPCh. 1 - Prob. 1.88ASPCh. 1 - Prob. 1.89ASPCh. 1 - Prob. 1.90ASPCh. 1 - Ethylene glycol, commonly used as automobile...Ch. 1 - Prob. 1.92ASPCh. 1 - Prob. 1.93ASPCh. 1 - Prob. 1.94ASPCh. 1 - Prob. 1.95ASPCh. 1 - Prob. 1.96ASPCh. 1 - Prob. 1.97ASPCh. 1 - Prob. 1.98ASPCh. 1 - Prob. 1.99ASPCh. 1 - Approximately 75 mL of blood is pumped by a normal...Ch. 1 - Prob. 1.101CPCh. 1 - Prob. 1.102CPCh. 1 - Prob. 1.103CPCh. 1 - Prob. 1.104CPCh. 1 - Prob. 1.105CPCh. 1 - Prob. 1.106CPCh. 1 - Prob. 1.107CPCh. 1 - When 1.0 tablespoon of butter is burned or used by...Ch. 1 - Prob. 1.109CPCh. 1 - Prob. 1.110CPCh. 1 - Prob. 1.111CPCh. 1 - At a certain point, the Celsius and Fahrenheit...Ch. 1 - Prob. 1.113GPCh. 1 - Prob. 1.114GPCh. 1 - Sulfuric acid (H2SO4, density 1.83 g/mL) is...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- A helium gas cylinder of the sort used to fill balloons has a volume of 0.180 m3 and a pressure of 150 X 105 Pa (150 atm) at 298 K (25 °C). How many moles of helium are in the tank? How many grams?arrow_forwardHow many moles of HCl are there in 75.0 mL of 0.160 M HCl?arrow_forwardCalculate the mass of a liquid with a density of 3.2 g/mL and a volume of 25 mLarrow_forward
- The density of a substance is 3.596 g/ml and has a weight of 32g. What is the volume?arrow_forwardA sample of steel weighing 2.00 g is analyzed for Cr (AW 52.0). The Cr is oxidized into chromate with alkaline permanganate and the excess permanganate is destroyed. A certain volume of 0.120 M FeSO4 is added to the acid solution and the excess is titrated with 0.0220 M KMnO4, requiring 31.0 mL. If the sample contained 0.50 % Cr, what volume (in mL) of FeSO4 was added?arrow_forwardAn aerosol spray can of deodorant with a volume of 350 mL contains 3.2 g of propane gas (C3H8) as propellant. What is the pressure (in Pa) in the can at 20 °C?arrow_forward
- Let's say you had a 10mL, 25mL,50mL and a 100mL graduated cylinder.Which one would you use if you needed to measure a volume of 5mL? Why?arrow_forwardA patient is prescribed 15mmol of potassium chloride injection. The ampoule contains 20mmol in 10ml. What volume of potassium chloride (ml) is required? units – mlarrow_forwardA vial containing 200 mg hydrocortisone sodium succinate powder for injection is to be reconstituted to produce 4 ml of injection. Which of the following is the amount of Water for Injections, BP that should be added to the powder? The displacement volume of hydrocortisone sodium succinate is 0.05 ml/50 mg. 2 ml 3 ml 2.5 ml 3.8 ml O 3.9 mlarrow_forward
- 1) What is the prefix that is used when we are expressing 1/1000 of the unit? 2) What is the unit of measurement for volume? 3) How many grams (g) are in 200 micrograms (ug)? 4) There are 2.54 cm in an inch. There are 12 inches in a foot. Calculate the height of a 6-foot 11-inch person in meters. 5) How many micrograms (ug) does 1 ul of water weigh? 6) Convert the following numbers: a. 1 ul = ml b. 1.534 cm = mm c. 46.8 ul = dl d. 6.9732 ug = kg 7) A drug is given at a dosage of 25 mg / kg body weight. a. Convert this dosage to ug per kg (ug/kg). b. How many ug of the drug in question a should be administered to a person who weighs 77 kg? 8) At rest the left ventricle of the heart pumps 5.0 liters of blood per minute (this is known as the cardiac output). Blood flow to the kidneys is approximately 1,220 ml per min at rest. a. What percentage (%) of the cardiac output do the kidneys receive at rest? b. Assume that the kidneys will receive the same percent of the cardiac output that was…arrow_forwardAn acidified and reduced iron sample required 40.2 ml of 0.0206 M KMN04. Find mg Fe (at wt = 55.8) and mg Fe203 (FW = 159.7 mg/mmol).arrow_forwardYou need to give your patient a dose of 20mg and the medication comes in a dry form with a dry weight of 200mg. To reconstitute, you must add 10mL to the dry powder to get a final concentration of 16.7mg/mL. How much volume (in mL) should you give your patient for a single injection based on this order (20mg)?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning




Principles Of Radiographic Imaging: An Art And A ...
Health & Nutrition
ISBN:9781337711067
Author:Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:Cengage Learning
GCSE Chemistry - Acids and Bases #34; Author: Cognito;https://www.youtube.com/watch?v=vt8fB3MFzLk;License: Standard youtube license