
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
8th Edition
ISBN: 8220102895805
Author: Peterson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 1, Problem 1.32UKC
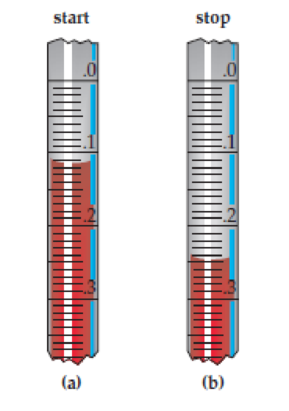
Assume that you are delivering a solution sample from a pipette. Figures (a) and (b) show the volume level before and after dispensing the sample, respectively. State the liquid level (in mL) before and after dispensing the sample, and calculate the volume of the sample.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A student needs 240 mL of a 1/20 dilution of an undiluted stock solution. What volume
(mL) of the stock solution will be required?
Note: Your answer should be in numerical format to the nearest whole number (with a
decimal).
If you transferred 15 mL of an undiluted sample into 35 mL of diluting fluid, what dilution have you created?
(Note: provide your answer in decimal format to three decimal places.)
You inject a patient with 4 mg of inulin and find their concentration to be 1.4 mg/L. You then inject a patient with 10 mg of D20 and later find their concentration to be .23 mg/L. What would you estimate the volume of their ICF to be?
Chapter 1 Solutions
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
Ch. 1.2 - Pure acetic acid, which gives the sour taste to...Ch. 1.3 - Classify each of the following as a mixture or a...Ch. 1.3 - Classify each of the following as a physical...Ch. 1.3 - In the next image, red spheres represent element A...Ch. 1.3 - The active ingredient in aspirin, ASA, melts at...Ch. 1.3 - Prob. 1.2CIAPCh. 1.4 - Match the names of the elements described below...Ch. 1.4 - Identify the elements represented in each of the...Ch. 1.6 - Calomel (Hg2Cl2) is not toxic but methyl mercury...Ch. 1.6 - Give the full name of the following units and...
Ch. 1.8 - Prob. 1.8PCh. 1.8 - How would you record the temperature reading on...Ch. 1.8 - Prob. 1.10PCh. 1.8 - Convert the following values from scientific...Ch. 1.8 - Prob. 1.12PCh. 1.9 - Round off the following quantities to the...Ch. 1.9 - Carry out the following calculations, rounding...Ch. 1.10 - Prob. 1.15PCh. 1.10 - Convert 0.840 qt to milliliters in a single...Ch. 1.10 - A patient is to receive 20 mg of methimazole, a...Ch. 1.10 - Calculate the dosage in milligrams per kilogram...Ch. 1.11 - A thermochromic plastic chip included in a...Ch. 1.11 - A temperature-sensitive bath toy undergoes several...Ch. 1.11 - The highest land temperature ever recorded was 136...Ch. 1.11 - Prob. 1.20PCh. 1.11 - Prob. 1.21PCh. 1.11 - What is the specific heat of aluminum if it takes...Ch. 1.12 - A sample of pumice, a porous volcanic rock, weighs...Ch. 1.12 - Chloroform, once used as an anesthetic agent, has...Ch. 1.12 - The sulfuric acid solution in an automobile...Ch. 1.12 - Prob. 1.6CIAPCh. 1.12 - Prob. 1.7CIAPCh. 1 - The six elements in blue at the far right of the...Ch. 1 - Identify the three elements indicated on the...Ch. 1 - The radioactive element indicated on the following...Ch. 1 - (a)What is the specific gravity of the following...Ch. 1 - Assume that you have two graduated cylinders, one...Ch. 1 - State the length of the pencil depicted in the...Ch. 1 - Assume that you are delivering a solution sample...Ch. 1 - Assume that identical hydrometers are placed in...Ch. 1 - What is the difference between a physical change...Ch. 1 - Which of the following is a physical change and...Ch. 1 - Which of the following is a physical change and...Ch. 1 - Name and describe the three states of matter.Ch. 1 - Prob. 1.38ASPCh. 1 - Sulfur dioxide is a compound produced when sulfur...Ch. 1 - Butane (C4H8) is an easily compressible gas used...Ch. 1 - Classify each of the following as a mixture or a...Ch. 1 - Which of these terms, (i) mixture, (ii) solid,...Ch. 1 - Hydrogen peroxide, often used in solutions to...Ch. 1 - Prob. 1.44ASPCh. 1 - What is the most abundant element in the earths...Ch. 1 - Prob. 1.46ASPCh. 1 - Supply the missing names or symbols for the...Ch. 1 - Prob. 1.48ASPCh. 1 - Prob. 1.49ASPCh. 1 - Prob. 1.50ASPCh. 1 - Glucose, a form of sugar, has the formula C6H12O6....Ch. 1 - Prob. 1.52ASPCh. 1 - Prob. 1.53ASPCh. 1 - Prob. 1.54ASPCh. 1 - Prob. 1.55ASPCh. 1 - Prob. 1.56ASPCh. 1 - Prob. 1.57ASPCh. 1 - How many pictograms are in 1 mg? In 35 ng?Ch. 1 - How many microliters are in 1 L? In 20 mL?Ch. 1 - Prob. 1.60ASPCh. 1 - Prob. 1.61ASPCh. 1 - Prob. 1.62ASPCh. 1 - Prob. 1.63ASPCh. 1 - Prob. 1.64ASPCh. 1 - Round off each of the numbers in Problem 1.63 to...Ch. 1 - Carry out the following calculations, express each...Ch. 1 - Prob. 1.67ASPCh. 1 - Carry out the following conversions: (a) 3.614 mg...Ch. 1 - Carry out the following conversions. Consult...Ch. 1 - Express the following quantities in more...Ch. 1 - Fill in the blanks to complete the equivalencies...Ch. 1 - Prob. 1.72ASPCh. 1 - The muzzle velocity of a projectile fired from a 9...Ch. 1 - Prob. 1.74ASPCh. 1 - The Willis Tower in Chicago has an approximate...Ch. 1 - Prob. 1.76ASPCh. 1 - Prob. 1.77ASPCh. 1 - The white blood cell concentration in normal blood...Ch. 1 - Prob. 1.79ASPCh. 1 - Diethyl ether, a substance once used as a general...Ch. 1 - Prob. 1.81ASPCh. 1 - Calculate the specific heat of copper if it takes...Ch. 1 - Prob. 1.83ASPCh. 1 - A 150 g sample of mercury and a 150 g sample of...Ch. 1 - When 100 cal (418 J) of heat is applied to a 125 g...Ch. 1 - Prob. 1.86ASPCh. 1 - Prob. 1.87ASPCh. 1 - Prob. 1.88ASPCh. 1 - Prob. 1.89ASPCh. 1 - Prob. 1.90ASPCh. 1 - Ethylene glycol, commonly used as automobile...Ch. 1 - Prob. 1.92ASPCh. 1 - Prob. 1.93ASPCh. 1 - Prob. 1.94ASPCh. 1 - Prob. 1.95ASPCh. 1 - Prob. 1.96ASPCh. 1 - Prob. 1.97ASPCh. 1 - Prob. 1.98ASPCh. 1 - Prob. 1.99ASPCh. 1 - Approximately 75 mL of blood is pumped by a normal...Ch. 1 - Prob. 1.101CPCh. 1 - Prob. 1.102CPCh. 1 - Prob. 1.103CPCh. 1 - Prob. 1.104CPCh. 1 - Prob. 1.105CPCh. 1 - Prob. 1.106CPCh. 1 - Prob. 1.107CPCh. 1 - When 1.0 tablespoon of butter is burned or used by...Ch. 1 - Prob. 1.109CPCh. 1 - Prob. 1.110CPCh. 1 - Prob. 1.111CPCh. 1 - At a certain point, the Celsius and Fahrenheit...Ch. 1 - Prob. 1.113GPCh. 1 - Prob. 1.114GPCh. 1 - Sulfuric acid (H2SO4, density 1.83 g/mL) is...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- For investigating the effect of pressure (height of a column of liquid), a 50-mL burette was used with a small length of rubber tubing fixed to the bottom on which a clamp could be fitted to start/stop the flow of water (Figure 1). Effluent was collected in a beaker and mean flow rate was calculated (mL/s), and a stopwatch was used to measure how long it took for 5 mL of liquid to flow out. For the subsequent sections of the practical, a 25-mL burette was used, and the top was connected via rubber tubing to a 5-liter reservoir placed on the shelving above the bench. The tubing at the base was connected to the different flow modules that comprised the different configurations of tubes to be tested. a. Interpret the graph. b. What does this relate to in the cardiovascular system, and is it something that can change? How? c. What would be the implications for our ability to control blood flow to different tissues, if this were the only control mechanism available? d. Based on the…arrow_forwardTwo students independently determine the volume of water delivered by a 10.00-mL pipet. Each student takes 8 measurements, then computes the average volume delivered and the standard deviation. The results are tabulated below. Average St. Dev. Student A 10.4 mL ±0.7 mL Student B 10.00 mL ±0.02 mL Which statement best describes the results? 1)A: good precision, poor accuracy. B: poor precision, good accuracy. 2)A: poor precision, good accuracy. B: good precision, poor accuracy. 3)A: poor precision, poor accuracy. B: good precision, good accuracy. 4)A: good precision, good accuracy. B: good precision, good accuracy. 5)A: poor precision, poor accuracy. B: poor precision, poor accuracy.arrow_forwardHow many CFU per ml are there in the original sample using the following dilution and platingseries? Make sure to label all dilutions that were done.arrow_forward
- A patient is prescribed 15mmol of potassium chloride injection. The ampoule contains 20mmol in 10ml. What volume of potassium chloride (ml) is required? units – mlarrow_forwardFor investigating the effect of pressure (height of a column of liquid), a 50-mL burette wasused with a small length of rubber tubing fixed to the bottom on which a clamp could befitted to start/stop the flow of water (Figure 1). Effluent was collected in a beaker and meanflow rate was calculated (mL/s), and a stopwatch was used to measure how long it took for5 mL of liquid to flow out. For the subsequent sections of the practical, a 25-mL burette wasused, and the top was connected via rubber tubing to a 5-liter reservoir placed on theshelving above the bench. The tubing at the base was connected to the different flowmodules that comprised the different configurations of tubes to be tested. Figure 1: A. Theoretical set-up of the experiment. B. Flow rate data with increasing pressure a. Interpret the graph.b. What does this relate to in the cardiovascular system, and is it something that canchange? How?c. What would be the implications for our ability to control blood flow to different…arrow_forwardYou need to give your patient a dose of 20mg and the medication comes in a dry form with a dry weight of 200mg. To reconstitute, you must add 10mL to the dry powder to get a final concentration of 16.7mg/mL. How much volume (in mL) should you give your patient for a single injection based on this order (20mg)?arrow_forward
- Show the formula and steps that you will use to complete these calculations. The physician has prescribed a dose of 100 mg of intramuscular morphine. The available stock has a strength of 10 mg/mL. How much of this solution should you draw up into the syringe? The prescribed dose of a drug is 120 mcg. The available stock has a strength of 20 mcg/mL. How much should you give the patient? Substitute the known quantities in the formula and perform a calculation to determine the volume. Show your work).arrow_forwardThe answer key is 24.5 mL/hr. Please show step by step solution.arrow_forwardOne hundred (100) ml of raw goat's milk bought from a street vendor is added in bottle 1 which initially contains 900 ml diluent. The sample was serially diluted by transferring 10 ml each to the succeeding bottles 2, 3 and 4 all of which contain 90ml diluent. Calculate the final dilution in bottle 4 expressed in decimal number. (Attach a screenshot of your computations at the end of the assessment). 0.01 0.001 0.0001 0.00001 0.000001arrow_forward
- You record the absorbance of your unknown BSA sample at 0.276. What is the concentration in ug/mL of the unknown assuming you have a standard curve with the line of best fit with y=0.0001x+0.0038 as its equationarrow_forwardAn order was received for 750 mL of 7.5% dextrose solution using 5% D5W and 20% D20W. How many parts of each dextrose solution is needed?(round answers to the nearest tenth)arrow_forwardThe physician ordered 700ml of a special electrolyte solution to flow at 75 gtt/min. The drop factor is 15 gtt/ml. How long will it take in hours and minutes will it take to infuse? using dimensional analysis and show workarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON

Biochemistry
Biochemistry
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:9781464126116
Author:David L. Nelson, Michael M. Cox
Publisher:W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul...
Biochemistry
ISBN:9781118918401
Author:Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:WILEY

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Fundamentals of General, Organic, and Biological ...
Biochemistry
ISBN:9780134015187
Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:PEARSON
GCSE Chemistry - Acids and Bases #34; Author: Cognito;https://www.youtube.com/watch?v=vt8fB3MFzLk;License: Standard youtube license