
Concept explainers
(a)
Interpretation:
Lewis structures have to be drawn for the
Concept introduction:
Lewis structure: The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell. Sometimes the
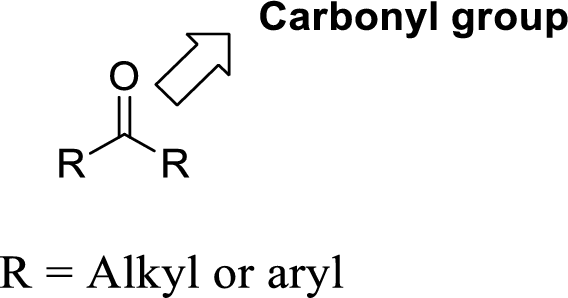
Carbonyl group:
A carbon atom is double-bonded to an oxygen atom
(b)
Interpretation:
Lewis structures have to be drawn for the functional group and valence electrons have to be shown.
Concept introduction:
Lewis structure: The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell. Sometimes the chemical bonding of a molecule cannot be represented using a single Lewis structure. In these cases, the chemical bonding are described by delocalization of electrons and is known as resonance. All the possible resonance structures are imaginary whereas the resonance hybrid is real. These structures will differ only in the arrangement of the electrons not in the relative position of the atomic nuclei.
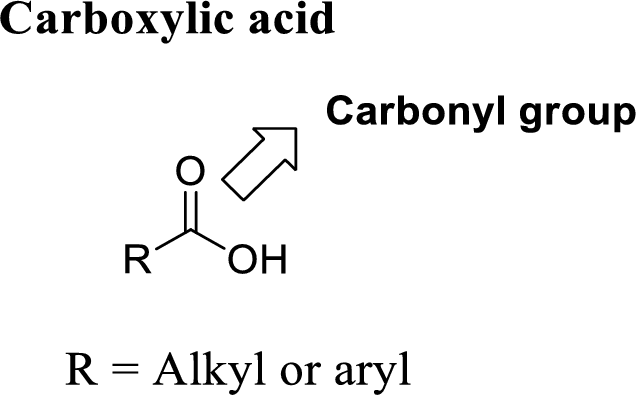
A carbon atom is double-bonded to an oxygen atom
If the carbonyl carbon is attached with hydroxyl group is called as carboxylic acid.
(c)
Interpretation:
Lewis structures have to be drawn for the functional group and valence electrons have to be shown.
Concept introduction:
Lewis structure: The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell. Sometimes the chemical bonding of a molecule cannot be represented using a single Lewis structure. In these cases, the chemical bonding are described by delocalization of electrons and is known as resonance. All the possible resonance structures are imaginary whereas the resonance hybrid is real. These structures will differ only in the arrangement of the electrons not in the relative position of the atomic nuclei.
Hydroxyl group:
Alcohol:
The compound contains hydroxyl
Example is given below
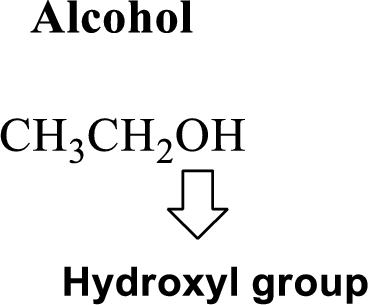
(d)
Interpretation:
Lewis structures have to be drawn for the functional group and valence electrons have to be shown.
Concept introduction:
Lewis structure: The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell. Sometimes the chemical bonding of a molecule cannot be represented using a single Lewis structure. In these cases, the chemical bonding are described by delocalization of electrons and is known as resonance. All the possible resonance structures are imaginary whereas the resonance hybrid is real. These structures will differ only in the arrangement of the electrons not in the relative position of the atomic nuclei.
Ester group:
A carbon atom is double-bonded to an oxygen atom
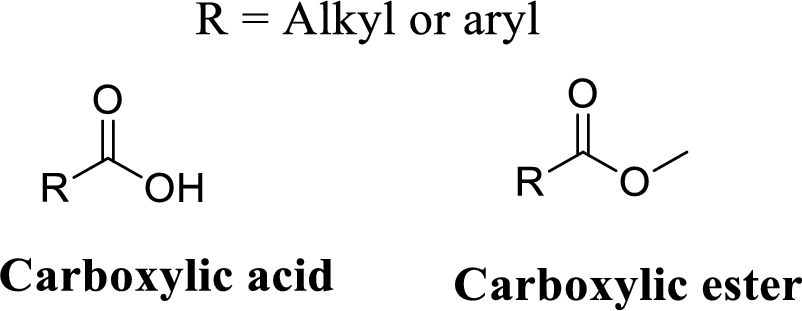
(e)
Interpretation:
Lewis structures have to be drawn for the functional group and valence electrons have to be shown.
Concept introduction:
Lewis structure: The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell. Sometimes the chemical bonding of a molecule cannot be represented using a single Lewis structure. In these cases, the chemical bonding are described by delocalization of electrons and is known as resonance. All the possible resonance structures are imaginary whereas the resonance hybrid is real. These structures will differ only in the arrangement of the electrons not in the relative position of the atomic nuclei.
Amide:
A carbon atom is double-bonded to an oxygen atom
If the carbonyl carbon is attached with nitrogen is called as amide.

Trending nowThis is a popular solution!

Chapter 1 Solutions
Student Study Guide and Solutions Manual for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- 1. Use the image provided to answer the question in detail: a) Two of the earliest drug molecules featured in Chapter 8 are Vicodin and Heroin, whose chemical structures are very similar. Both of these molecules contain an amine functional group and several rings. Indicate the number of aromatic rings in each structure, and identify the amine functional group in each molecule as primary, secondary, or tertiary.arrow_forwardFollowing is the structural formula of acetylsalicylic acid, better known by its common name aspirin. (a) Name the two oxygen-containing functional groups in aspirin. (b) What is the molecular formula of aspirin?arrow_forwardIdentify which of the following statement(s) is/are true. (a) A functional group is a group of atoms in an organic molecule that undergoes a predictable set of chemical reactions. (b) The functional group of an alcohol, an aldehyde, and a ketone have in common the fact that each contains a single oxygen atom. (c) A primary alcohol has one -OH group, a secondary alcohol has two -OH groups, and a tertiary alcohol has three -OH groups. (d) There are two alcohols with the molecular formula C3H8O. (e) There are three amines with the molecular formula C3H9N.arrow_forward
- (b) Draw the structural formula for each of the following compounds. Lukiskan formula struktur bagi setiap sebatian berikut. (i) 2-iodo-4-methylpentan-3-amine (ii) 1,1-dibromo-2-chloro-2-methylbutane (iii) 2-ethyl-6-fluorophenolarrow_forwardBelow is the structure for asprin. CHOOSE ALL THAT APPLY: list all functional groups present in the compound. alcohol, Ester, aldehyde, ketone, Carboxylic acid, Benzene, thiolarrow_forwardAmines and amides both contain nitrogen. What are the differences in the structures of amines and amides in terms of the functional groups?arrow_forward
- Write the chemical formula, bond-line structure, and condensed formula of undecanoic acid, and draw its bond line structure.arrow_forwardWe see that 1-propanol and 2-propanol have the same molecular formula, C3H7OH, but different molecular structures. What is the name for molecules that have the same molecular formula but different structural formulas (different shapes)? Use the specific term.arrow_forward(1) What is the valence electron configuration for the carbon atom? (2) What is the valence electron configuration for the carbon atom?arrow_forward
- Draw Lewis structures for these functional groups. Show all valence electrons also. Q.) Amide grouparrow_forwardPlease refer to the molecule shown below when answering questions: (a) Depending on the reagent used, which functional group is capable of undergoing E1/E2or SN1/SN2 reactions?(b) An aqueous solution of this functional group is likely to turn blue litmus paper red:(c) Identify a functional group that can easily undergo addition reactions with HBr or HCl.(d) Identify a secondary amide.arrow_forwardFor the following, you must draw an appropriate structure that has the chemical formula C5H9NO with the indicated functional group(s) and/or property. In each case, identify any other functional groups in the molecule you draw, that were not indicated in the question. You may use condensed dash or bond-line structure to draw your molecules. c) An acyclic amide that cannot form hydrogen bonds with itselfarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





