
Concept explainers
(a)
Interpretation:
The eight alcohols have to be written with the molecular formula of
Concept introduction:
Isomer: A molecule having same molecular formula with different chemical formula is called isomer.
Functional group: They are certain substitutes in the organic molecules which are determine the characteristic reactions taking place in it.
Alcohol:
The compound contains hydroxyl
Example is given below,
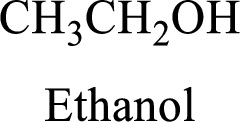
(b)
Interpretation:
Eight aldehydeswith the molecular formula of
Concept introduction:
Isomer: A molecule having same molecular formula with different chemical formula is called isomer.
Functional group: They are certain substitutes in the organic molecules which are determine the characteristic reactions taking place in it.
Carbonyl group:
A carbon atom is double-bonded to an oxygen atom
If the carbonyl carbon is attached with two alkyl or aryl group is called as
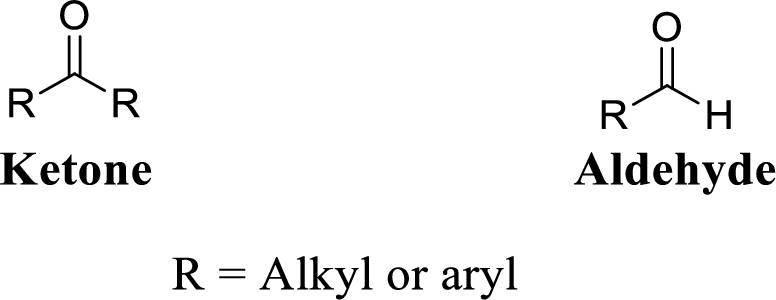
(c)
Interpretation:
Six ketones have to be written with the molecular formula of
Concept introduction:
Isomer: A molecule having same molecular formula with different chemical formula is called isomer.
Functional group: They are certain substitutes in the organic molecules which are determine the characteristic reactions taking place in it.
Carbonyl group:
A carbon atom is double-bonded to an oxygen atom
If the carbonyl carbon is attached with two alkyl or aryl group is called as ketone, if the carbonyl carbon is attached with one hydrogen atom and one alkyl or aryl group is called as aldehyde.
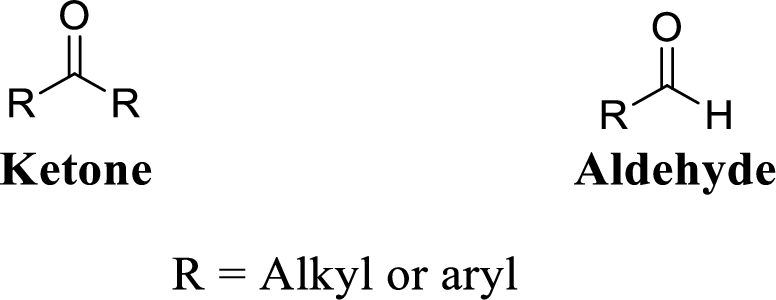
(d)
Interpretation:
Eight carboxylic acid have to be written with the molecular formula of
Concept introduction:
Isomer: A molecule having same molecular formula with different chemical formula is called isomer.
Functional group: They are certain substitutes in the organic molecules which are determine the characteristic reactions taking place in it.
Carboxylic acid:
A carbon atom is double-bonded to an oxygen atom
If the carbonyl carbon is attached with hydroxyl group is called as carboxylic acid.
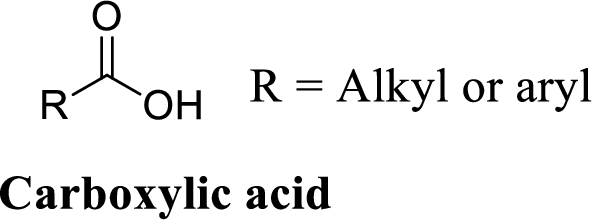
(e)
Interpretation:
Nine carboxylic estershave to be written with the molecular formula of
Concept introduction:
Isomer: A molecule having same molecular formula with different chemical formula is called isomer.
Functional group: They are certain substitutes in the organic molecules which are determine the characteristic reactions taking place in it.
Carboxylic ester:
A carbon atom is double-bonded to an oxygen atom
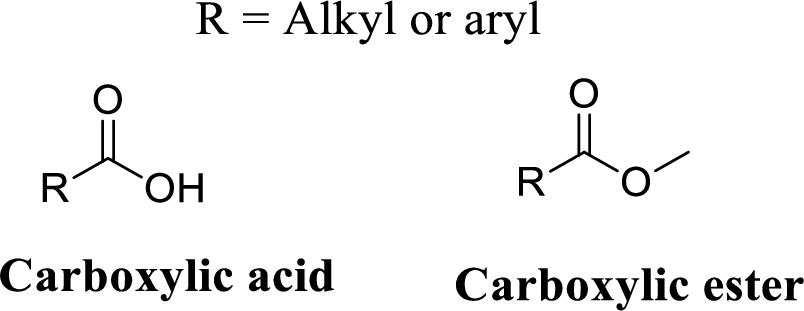
Trending nowThis is a popular solution!

Chapter 1 Solutions
Organic Chemistry
- Write a condensed structural formula for a dihydroxy compound with the formula C3H8O2.arrow_forwardEnter the molecular formula for butane, C4H10?arrow_forwardGlucose, C6H12O6, contains an aldehyde group but exist predominantly in the form of the cyclic hemiacetal show below. A cyclic hemiacetal is formed when the —OH group of one carbon bonds to the carbonyl group of another carbon. Identify which carbon provides the —OH group and which provides the —CHO? Give a functional isomer of glucose and draw its structure.arrow_forward
- Draw the expanded structural formula for a carboxylic acid that has the formula C5H10O2C5H10O2, with two methyl substituents.arrow_forwardDraw and name the five cycloalkane structures of formula C5H10. Can any of these structures give rise to geometric (cis-trans) isomerism? If so, show the cis and trans stereoisomersarrow_forwardGive the structural formulae of all the chain isomers of C5H12.arrow_forward
- Write the chemical equation for the reaction of propanoic acid with 1-butanol (an alcohol). The formula of 1-butanol is CH-CH-CH2-CH2-OH. Which is formula of the this rule of reaction?arrow_forwardWrite structural formulas for three ketones that have the formula C5H10O.arrow_forwardDraw all the structural and geometric isomers of pentene, C5H10, that have an unbranched hydrocarbon chain.arrow_forward
- 1. the three secondary alcohols with the molecular formula C5H12O. 2. the Three secondary amine with the molecular formula C4H11N. 3. Draw the structural formulas for the six ketones with the molecular formula C6H12O. 4. Draw Structural formulas to the eight carboxylic acids with the molecular formula C6H12O2.arrow_forwardDraw all the possible structural isomers with the molecular formula C6H14, Name themarrow_forwardDraw the structures of all monobromo derivatives of pentane, C5H11Br, which contain a 4-carbon chain.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning




