
Concept explainers
(a)
Interpretation:
The given diameter 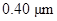 is to be converted to meters and reported in scientific notation.
is to be converted to meters and reported in scientific notation.
Concept introduction:
The scientific notation is the standard method of representation of a long or tedious measurement. The number is expressed as number times ten raised to some power.
For instance mass of a proton is conveniently expressed as 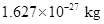 The decimal is shifted to right when the power is positive while it is negative if the decimal has been shifted to left.
The decimal is shifted to right when the power is positive while it is negative if the decimal has been shifted to left.
While a number is written in scientific notations it is scaled down to some digit between 1 to10 ten and the places that previously contain zeroes are changed to ten raised to some power.
For example, 0.00056 is written as 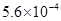
The conversion factor to convert the 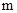 to
to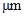 is as follows:
is as follows:
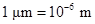
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- Hemoglobin is a protein that transports oxygen from the lungs to the rest of the body. Lab results indicated a patient had a hemoglobin concentration in the blood of 15.5 g/dL, which is in the normal range. (a) Convert the number of grams to milligrams and write the answer in scientifi c notation. (b) Convert the number of grams to micrograms and write the answer in scientifi c notationarrow_forward1(a) Convert the number 0.0000735 to scientific notation. (b) Round off the number 24.15725 to the hundredths place. (c) Convert the number 5.31 x 10³ from scientific notation to regular number. 2a) Take a look at the image, and answer the following questions. 2 3 4 (i) What is the correct length of the red tape? (ii) The number of significant figures in the measurement is (iii) Convert this length to meters(m) and inches(in). Show complete calculations. 2b) The actual mass of a glass marble was recorded as 5.3076 g. When a student measured the mass three times the results were recorded as 5.2987g , 5.3197 g and 5.2078 g Which one of the results can be considered accurate and why? 3(i) The density of copper is 8.92 g/em'. What is the mass(in grams) of a piece of copper which has a length of 3.16 cm, width of 2.74 cm and height of 1.88 cm?arrow_forwardExpress the following numbers in scientific notation:(a) 0.000000027, (b) 356, (c) 47,764, (d) 0.096.arrow_forward
- (a) How many picometers are there in 1 m? (b) Express6.0 * 103 m using a prefix to replace the power of ten.(c) Use exponential notation to express 4.22 mg in grams.(d) Use decimal notation to express 4.22 mg in grams.arrow_forwardThe diameter of Jupiter is 142,984 kilometers. The diameter of Mercury is 4,878 kilometers.(a) Express each of these diameters in scientific notation. Use all the digits given.(b) Which planet has the larger diameter? How many orders of magnitude larger?arrow_forwardA piece of cardboard is 12.0 cm long and 6.3 cm wide. What is the area of this piece of cardboard in square millimeters? Enter your answer in the space provided WITHOUT units. Make sure to round your answer to the correct number of significant figures. Please give the number as a normal number, do not use scientific notation. (In example: a normal number = 4300 instead of the number being in scientific notation as = 4.3 X 103)arrow_forward
- Perform the following conversions. You must use dimensional analysis and put your answer in scientific notation with correct units and significant figures. a) 65.2 mL to uL (Hint: you must go through the base/metric unit) 4 points b) 65.1 miles/hour to m/s 7 points Please only utilize the conversions provided. If calculations use conversions not provided, your answers will not be graded). c) 35.6 cm2 to km² (You must go through the base/metric unit) 5 points (Please write your answer on a piece of paper. No need to type anything here). Edit View Insert Format Tools Table 12pt v Paragraph v BIU O words I| Proctorio is sharing your screen. Stop sharing Hidearrow_forwardCalculate the volume of 42.0 kg of a substance whose density is 8.96 g/mL. Express your answer in milliliters using the correct number of significant figures. Do not enter your answer using scientific notation.arrow_forward1A)convert 6.020x10^-3 to decimal notation 1b) convert 15,000,000 to scientific notation. 2) perform the following calculation show the answer in scientific notation. Round the coefficient to the correct number of Sig figs. (Select the best answer choice) (3.3x10^8)x (6.20x10^-3)=a) 2.05x10^6 b) 2.046x10^6 c) 20.46x10^5 d) 2,046,000 e)2.0x10^6arrow_forward
- Complete the following volume equivalents. (a) 1 L = ? cm³ (b)1 in.³ = ? mLarrow_forwardConvert 97.4 micrometers^3 to liters. Note that 1 mL=1 cm3.arrow_forward2. Carry out the following operations and express the answer with the appropriate number of significant figures and units: (a) (5.231 mm)(6.1 mm), (b) 72.3 g/1.5 mL, (c) 12.21 g+0.0132 g, and (d) 31.03 g+ 12 mg.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





